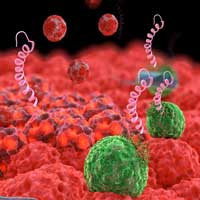
 For synthetic biologists these concepts aren't some far-off ideal. They are projects already in progress, and they have huge real-world implications should they succeed.
For synthetic biologists these concepts aren't some far-off ideal. They are projects already in progress, and they have huge real-world implications should they succeed.
Dec 6th, 2019
Read more
 You cannot make glue out of a ham sandwich - but you may be able to use the components of that food to create a strong adhesive.
You cannot make glue out of a ham sandwich - but you may be able to use the components of that food to create a strong adhesive.
Dec 5th, 2019
Read more
 Cell-free systems stand to dramatically impact society, from the environment to medicine to education.
Cell-free systems stand to dramatically impact society, from the environment to medicine to education.
Dec 2nd, 2019
Read more
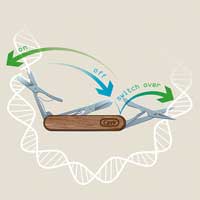 Scientists have developed a revolutionary CRISPR technology called 'CRISPR-Switch', which enables unprecedented control of the CRISPR technique in both space and time.
Scientists have developed a revolutionary CRISPR technology called 'CRISPR-Switch', which enables unprecedented control of the CRISPR technique in both space and time.
Nov 29th, 2019
Read more
 New method gives protein-based hydrogels shape-memory capabilities.
New method gives protein-based hydrogels shape-memory capabilities.
Nov 29th, 2019
Read more
 Researchers have created a new coating to prevent clotting and infection in synthetic vascular grafts, while also accelerating the body?s own process for integrating the grafted vessels.
Researchers have created a new coating to prevent clotting and infection in synthetic vascular grafts, while also accelerating the body?s own process for integrating the grafted vessels.
Nov 27th, 2019
Read more
A move towards a more sustainable bio-based economy has been given a new boost by researchers who have been able to simplify a process to transform waste materials into high value chemicals.
Nov 26th, 2019
Read more
 Researchers present sensor prototype that can rapidly, precisely, and cost-effectively measure molecular signals for cancer.
Researchers present sensor prototype that can rapidly, precisely, and cost-effectively measure molecular signals for cancer.
Nov 25th, 2019
Read more
 Scientists have developed new delivery vehicles for future gene therapies. They are using artificial fats to transport DNA into cells.
Scientists have developed new delivery vehicles for future gene therapies. They are using artificial fats to transport DNA into cells.
Nov 18th, 2019
Read more
 New mechanism to explain how the cell organelle that sorts and distributes substances entering a cell is formed and maintained.
New mechanism to explain how the cell organelle that sorts and distributes substances entering a cell is formed and maintained.
Nov 15th, 2019
Read more
 New bio-inspired hydrogels can act like superglue in highly ionic environments such as seawater, overcoming issues in currently available marine adhesives.
New bio-inspired hydrogels can act like superglue in highly ionic environments such as seawater, overcoming issues in currently available marine adhesives.
Nov 12th, 2019
Read more
 Scientists computed a zoo of millions of alternate genetic polymer molecular structures, giving context for why biology encodes information how it does, and providing potential leads for new drugs and a guide to searches for extraterrestrial biology.
Scientists computed a zoo of millions of alternate genetic polymer molecular structures, giving context for why biology encodes information how it does, and providing potential leads for new drugs and a guide to searches for extraterrestrial biology.
Nov 11th, 2019
Read more
 Synthetic DNA-encoded monoclonal antibodies (DMAbs) allow for in vivo production of broadly neutralizing antibodies in preclinical studies.
Synthetic DNA-encoded monoclonal antibodies (DMAbs) allow for in vivo production of broadly neutralizing antibodies in preclinical studies.
Nov 10th, 2019
Read more
 A team of researchers have developed a dielectrophoresis enabled MicroelectroFluidic Probe (MeFP) that has the ability to sequentially separate and pattern mammalian cells in an open microfluidic system.
A team of researchers have developed a dielectrophoresis enabled MicroelectroFluidic Probe (MeFP) that has the ability to sequentially separate and pattern mammalian cells in an open microfluidic system.
Nov 4th, 2019
Read more
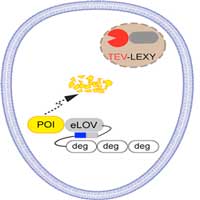 Researchers report they have developed a new technique that uses light to control the lifetime of a protein inside the cell. This method will allow scientists to better observe how specific proteins contribute to health, development and disease.
Researchers report they have developed a new technique that uses light to control the lifetime of a protein inside the cell. This method will allow scientists to better observe how specific proteins contribute to health, development and disease.
Nov 4th, 2019
Read more
 Finding a handful of cancer cells hiding among billions of blood cells in a patient sample can be like finding a needle in a haystack. In a new approach enabled by 3D-printed cell traps, researchers are removing the hay to expose the cancer cells.
Finding a handful of cancer cells hiding among billions of blood cells in a patient sample can be like finding a needle in a haystack. In a new approach enabled by 3D-printed cell traps, researchers are removing the hay to expose the cancer cells.
Oct 29th, 2019
Read more
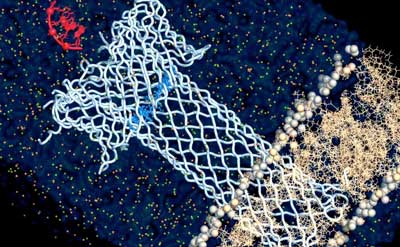 Some bacteria release a toxin that forms pores on other cells. Scientists have studied the pore-forming toxin aerolysin and genetically engineered it to be used as a high-resolution sensor for biological molecules like DNA and proteins.
Some bacteria release a toxin that forms pores on other cells. Scientists have studied the pore-forming toxin aerolysin and genetically engineered it to be used as a high-resolution sensor for biological molecules like DNA and proteins.
Oct 29th, 2019
Read more
 Envisioned technologies would integrate injury stabilization, regenerative therapy, and functional restoration to support new standard of care for spinal cord injury.
Envisioned technologies would integrate injury stabilization, regenerative therapy, and functional restoration to support new standard of care for spinal cord injury.
Oct 21st, 2019
Read more
 For synthetic biologists these concepts aren't some far-off ideal. They are projects already in progress, and they have huge real-world implications should they succeed.
For synthetic biologists these concepts aren't some far-off ideal. They are projects already in progress, and they have huge real-world implications should they succeed.
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed














