Revealing the rules behind virus scaffold construction
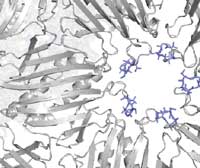 New insights into epistasis could impact future drug delivery and therapeutic strategies.
New insights into epistasis could impact future drug delivery and therapeutic strategies.
Mar 19th, 2019
Read more
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed
 New insights into epistasis could impact future drug delivery and therapeutic strategies.
New insights into epistasis could impact future drug delivery and therapeutic strategies.
Mar 19th, 2019
Read more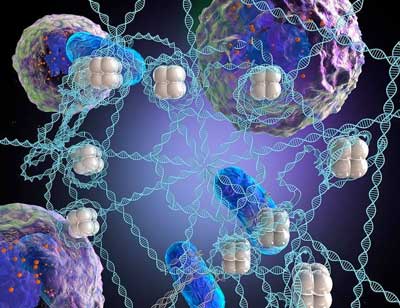 Today's antibiotics are not particularly engineered to coordinate their fight against bacteria with white blood cells, the body's own first line of defense against infectors, but a new study gives hope that that could change.
Today's antibiotics are not particularly engineered to coordinate their fight against bacteria with white blood cells, the body's own first line of defense against infectors, but a new study gives hope that that could change.
Mar 18th, 2019
Read more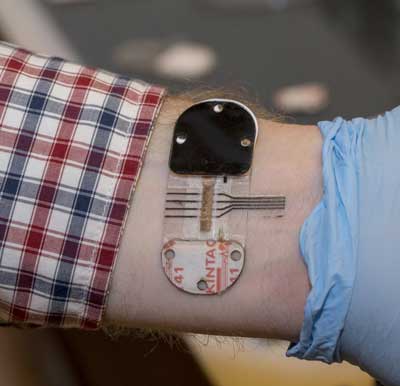 Researchers created the world's first continuous-monitoring device that tests sweat as effectively as blood but in a noninvasive way and over many hours.
Researchers created the world's first continuous-monitoring device that tests sweat as effectively as blood but in a noninvasive way and over many hours.
Mar 15th, 2019
Read more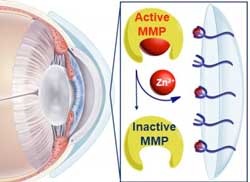 Researchers have created a hydrogel that could one day be made into a contact lens to more effectively treat corneal melting, a condition that is a significant cause for blindness world-wide.
Researchers have created a hydrogel that could one day be made into a contact lens to more effectively treat corneal melting, a condition that is a significant cause for blindness world-wide.
Mar 12th, 2019
Read more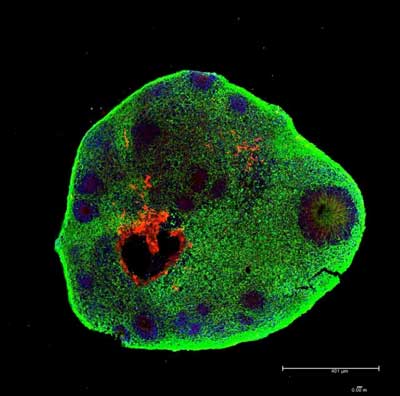 Scientists produce brain organoids with primitive eyes using computational fluid dynamics.
Scientists produce brain organoids with primitive eyes using computational fluid dynamics.
Mar 7th, 2019
Read more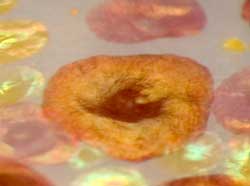 Marine biologists report a paradigm-shifting discovery in how specialized organs in squid skin, called chromatophores, contribute to the squid's camouflage via an elegant interplay of pigmentary action and structural coloration.
Marine biologists report a paradigm-shifting discovery in how specialized organs in squid skin, called chromatophores, contribute to the squid's camouflage via an elegant interplay of pigmentary action and structural coloration.
Mar 7th, 2019
Read more With a few genetic tweaks, a type of soil bacteria with an appetite for hydrocarbons shows promise as a biological factory for converting a renewable -- but frustratingly untapped -- bounty into a replacement for ubiquitous plastics.
With a few genetic tweaks, a type of soil bacteria with an appetite for hydrocarbons shows promise as a biological factory for converting a renewable -- but frustratingly untapped -- bounty into a replacement for ubiquitous plastics.
Mar 6th, 2019
Read more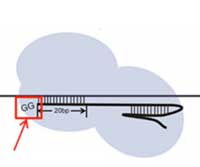 Digenome-seq technique reveals the accuracy of CRISPR-based editing tool that changes DNA letter A to G.
Digenome-seq technique reveals the accuracy of CRISPR-based editing tool that changes DNA letter A to G.
Mar 4th, 2019
Read more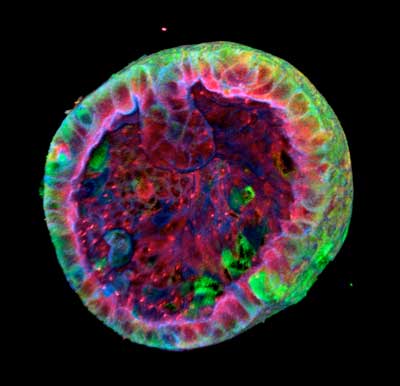 Scientists have successfully created kidney organoids from urine cells. This could lead to a wide range of new treatments that are less onerous for kidney patients.
Scientists have successfully created kidney organoids from urine cells. This could lead to a wide range of new treatments that are less onerous for kidney patients.
Mar 4th, 2019
Read more Researchers show that they can quickly measure a protein's microscopic movements, opening new possibilities in medical research.
Researchers show that they can quickly measure a protein's microscopic movements, opening new possibilities in medical research.
Mar 4th, 2019
Read more Researchers have employed a plasma protein found in blood to develop a new method for making wound-healing tissue scaffolds.
Researchers have employed a plasma protein found in blood to develop a new method for making wound-healing tissue scaffolds.
Mar 4th, 2019
Read more Scientists have invented technology to watch bacteria build cell walls in real time.
Scientists have invented technology to watch bacteria build cell walls in real time.
Mar 1st, 2019
Read more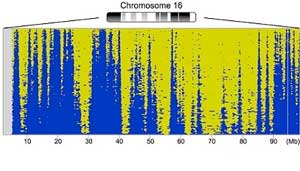 Researchers have established a novel method to scrutinize DNA replication in individual cells.
Researchers have established a novel method to scrutinize DNA replication in individual cells.
Mar 1st, 2019
Read more Imagine a day when a bioprinter filled with a patient's own cells can be wheeled right to the bedside to treat large wounds or burns by printing skin, layer by layer, to begin the healing process. That day is not far off.
Imagine a day when a bioprinter filled with a patient's own cells can be wheeled right to the bedside to treat large wounds or burns by printing skin, layer by layer, to begin the healing process. That day is not far off.
Feb 28th, 2019
Read more New shape memory polymer doesn't require additional triggers like temperature changes and could have major implications for health care treatment.
New shape memory polymer doesn't require additional triggers like temperature changes and could have major implications for health care treatment.
Feb 27th, 2019
Read more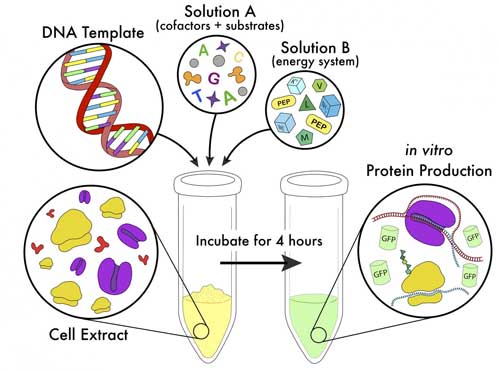 Researchers have radically simplified the method for cell-free protein synthesis (CFPS), a technique that could become fundamental to medical research.
Researchers have radically simplified the method for cell-free protein synthesis (CFPS), a technique that could become fundamental to medical research.
Feb 26th, 2019
Read more Technique sheds light on cells' health and development; may be useful for precision medicine.
Technique sheds light on cells' health and development; may be useful for precision medicine.
Feb 12th, 2019
Read more Scientists have developed a new method to produce hydrated materials, hydrogels, that have properties similar to the natural environment of cells in the body.
Scientists have developed a new method to produce hydrated materials, hydrogels, that have properties similar to the natural environment of cells in the body.
Feb 12th, 2019
Read more