 Researchers used a specific kind of photoluminescence spectroscopy, a way to detect light, to characterize the semiconductors. The emitted light energy was used as an indicator of the crystal's quality.
Researchers used a specific kind of photoluminescence spectroscopy, a way to detect light, to characterize the semiconductors. The emitted light energy was used as an indicator of the crystal's quality.
Aug 23rd, 2019
Read more
 Scientists find a simple method for producing atomically thin layers of oxidized borophene, a promising 2D boron-based nanomaterial that could serve in a variety of fields.
Scientists find a simple method for producing atomically thin layers of oxidized borophene, a promising 2D boron-based nanomaterial that could serve in a variety of fields.
Aug 23rd, 2019
Read more
 Chemists use fluorescence of molecular motors to sense conditions.
Chemists use fluorescence of molecular motors to sense conditions.
Aug 22nd, 2019
Read more
 By using nanocapsules, scientists and industrial partners are developing innovative garments and skin products that provide thermal comfort, and anti-ageing and antimicrobial properties.
By using nanocapsules, scientists and industrial partners are developing innovative garments and skin products that provide thermal comfort, and anti-ageing and antimicrobial properties.
Aug 22nd, 2019
Read more
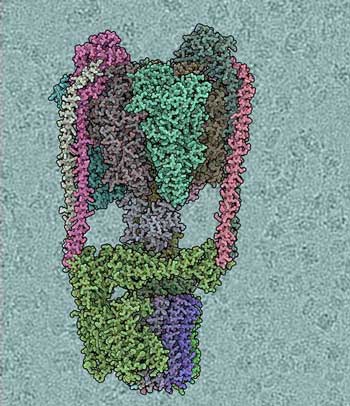 Scientists determine the first structure of a cell's rotary engine using state-of-art microscopy.
Scientists determine the first structure of a cell's rotary engine using state-of-art microscopy.
Aug 22nd, 2019
Read more
 Experiments yield conductivity measurements between two identical carbon nanotubes.
Experiments yield conductivity measurements between two identical carbon nanotubes.
Aug 22nd, 2019
Read more
 Scientists discovered a new imaging method - nanoscale nucleation kinetic mapping - which allows direct imaging of nucleation events at a solid-liquid interface.
Scientists discovered a new imaging method - nanoscale nucleation kinetic mapping - which allows direct imaging of nucleation events at a solid-liquid interface.
Aug 22nd, 2019
Read more
 Wearing a flower brooch that blooms before your eyes sounds like magic. Researchers have made it real with MXenes and robotic muscles.
Wearing a flower brooch that blooms before your eyes sounds like magic. Researchers have made it real with MXenes and robotic muscles.
Aug 22nd, 2019
Read more
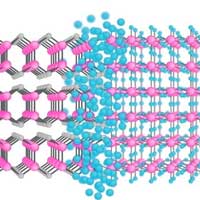 Researchers have developed a new method to synthesize 2D materials that are thought to be impossible or, at least, unobtainable by current technologies.
Researchers have developed a new method to synthesize 2D materials that are thought to be impossible or, at least, unobtainable by current technologies.
Aug 22nd, 2019
Read more
 Properties of complex materials are often determined by the interplay of several electron properties. Scientists have now succeeded in disentangling this mess.
Properties of complex materials are often determined by the interplay of several electron properties. Scientists have now succeeded in disentangling this mess.
Aug 22nd, 2019
Read more
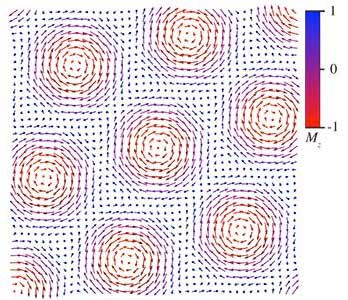 Researchers are investigating the dynamics of magnetic skyrmions with the eventual goal of developing spintronic devices.
Researchers are investigating the dynamics of magnetic skyrmions with the eventual goal of developing spintronic devices.
Aug 22nd, 2019
Read more
 A research has discovered the root cause of why lithium metal batteries fail - bits of lithium metal deposits break off from the surface of the anode during discharging and are trapped as 'dead' or inactive lithium that the battery can no longer access.
A research has discovered the root cause of why lithium metal batteries fail - bits of lithium metal deposits break off from the surface of the anode during discharging and are trapped as 'dead' or inactive lithium that the battery can no longer access.
Aug 22nd, 2019
Read more
 The research explains how random fluctuations affect the operation of microscopic machines like this tiny motor. In the future, such devices could be incorporated into other technologies to recycle waste heat and thus improve energy efficiency.
The research explains how random fluctuations affect the operation of microscopic machines like this tiny motor. In the future, such devices could be incorporated into other technologies to recycle waste heat and thus improve energy efficiency.
Aug 21st, 2019
Read more
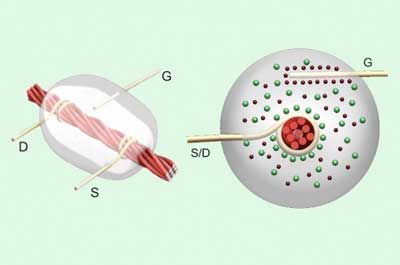 Transistors and integrated circuits made from threads are combined with thread-based sensors to create fully flexible devices.
Transistors and integrated circuits made from threads are combined with thread-based sensors to create fully flexible devices.
Aug 21st, 2019
Read more
 Physicists have demonstrated the real possibility of using superconductor/ferromagnet systems to create magnonic crystals, which will be at the core of spin-wave devices to come in the post-silicon era of electronics.
Physicists have demonstrated the real possibility of using superconductor/ferromagnet systems to create magnonic crystals, which will be at the core of spin-wave devices to come in the post-silicon era of electronics.
Aug 21st, 2019
Read more
 Researchers have developed artificial 'chameleon skin' that changes colour when exposed to light and could be used in applications such as active camouflage and large-scale dynamic displays.
Researchers have developed artificial 'chameleon skin' that changes colour when exposed to light and could be used in applications such as active camouflage and large-scale dynamic displays.
Aug 21st, 2019
Read more
 Researchers used a specific kind of photoluminescence spectroscopy, a way to detect light, to characterize the semiconductors. The emitted light energy was used as an indicator of the crystal's quality.
Researchers used a specific kind of photoluminescence spectroscopy, a way to detect light, to characterize the semiconductors. The emitted light energy was used as an indicator of the crystal's quality.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed