How slippery surfaces allow sticky pastes and gels to slide
 Engineered surface treatment can reduce waste and improve efficiency in many processes.
Engineered surface treatment can reduce waste and improve efficiency in many processes.
Apr 23rd, 2019
Read more
 Engineered surface treatment can reduce waste and improve efficiency in many processes.
Engineered surface treatment can reduce waste and improve efficiency in many processes.
Apr 23rd, 2019
Read more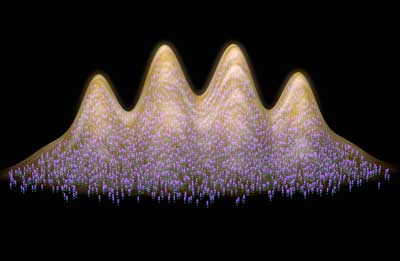 Scientists report on the observation of supersolid behavior in dipolar quantum gases of erbium and dysprosium. In the dysprosium gas these properties are unprecedentedly long-lived. This sets the stage for future investigations into the nature of this exotic phase of matter.
Scientists report on the observation of supersolid behavior in dipolar quantum gases of erbium and dysprosium. In the dysprosium gas these properties are unprecedentedly long-lived. This sets the stage for future investigations into the nature of this exotic phase of matter.
Apr 23rd, 2019
Read more A physical effect known as superinjection underlies modern light-emitting diodes (LEDs) and lasers. For decades this effect was believed to occur only in semiconductor heterostructures. Researchers now have found superinjection to be possible in homostructures, which are made of a single material.
A physical effect known as superinjection underlies modern light-emitting diodes (LEDs) and lasers. For decades this effect was believed to occur only in semiconductor heterostructures. Researchers now have found superinjection to be possible in homostructures, which are made of a single material.
Apr 22nd, 2019
Read more New research demonstrates the concept of a printable and wearable self-powered sensor system for ethanol/acetone detection.
New research demonstrates the concept of a printable and wearable self-powered sensor system for ethanol/acetone detection.
Apr 22nd, 2019
Read more Researchers are developing an automated system to synthesize entirely new materials made from stacked atomically thin two-dimensional sheets and to characterize their exotic quantum properties.
Researchers are developing an automated system to synthesize entirely new materials made from stacked atomically thin two-dimensional sheets and to characterize their exotic quantum properties.
Apr 22nd, 2019
Read more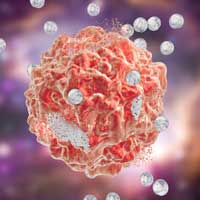 Tests showed that precision-guided anticancer nanoparticles have improved efficacy and reduced side effects.
Tests showed that precision-guided anticancer nanoparticles have improved efficacy and reduced side effects.
Apr 22nd, 2019
Read more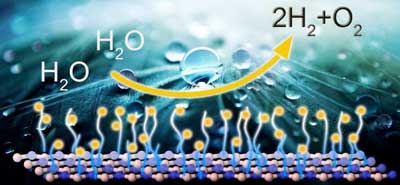 Scientists found a way how to use 2D material for hydrogen energy.
Scientists found a way how to use 2D material for hydrogen energy.
Apr 22nd, 2019
Read more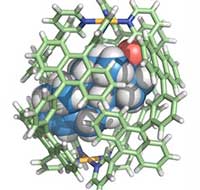 Study opens the door to developing ultrasensitive analytical devices for medical and sports applications.
Study opens the door to developing ultrasensitive analytical devices for medical and sports applications.
Apr 22nd, 2019
Read more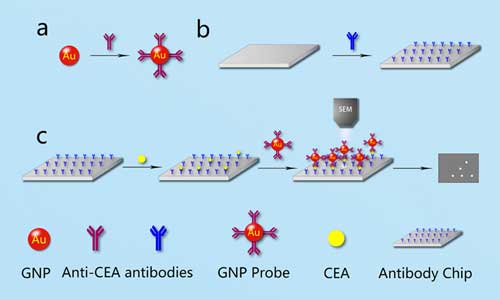 Researchers propose a gold nanoparticle (GNP) probe-assisted sandwich-counting strategy in vitro that relies on a GNP probe, an antibody-functionalized chip to 'count' antigen molecules using a scanning electron microscope.
Researchers propose a gold nanoparticle (GNP) probe-assisted sandwich-counting strategy in vitro that relies on a GNP probe, an antibody-functionalized chip to 'count' antigen molecules using a scanning electron microscope.
Apr 19th, 2019
Read more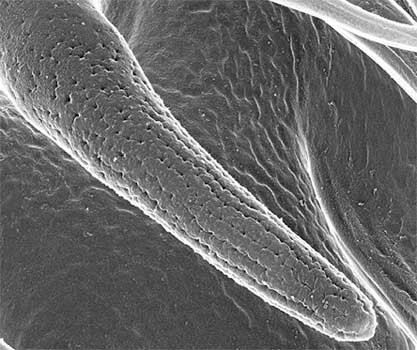 New insights into the formation of surface nanostructures.
New insights into the formation of surface nanostructures.
Apr 19th, 2019
Read more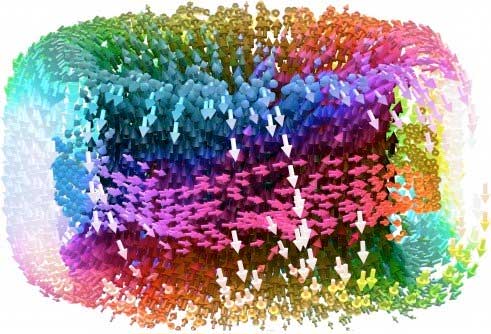 Research team makes a chiral skyrmion crystal with electric properties; puts new spin on future information storage applications.
Research team makes a chiral skyrmion crystal with electric properties; puts new spin on future information storage applications.
Apr 18th, 2019
Read more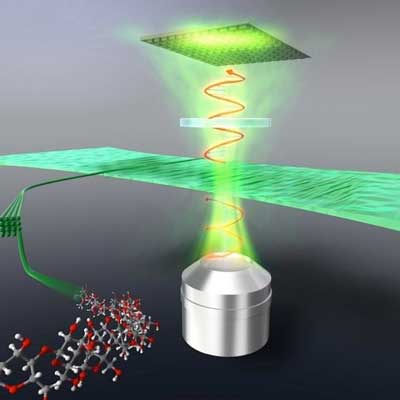 Researchers meticulously measured the optical birefringence of highly aligned cellulose nanofibers, paving the way for sharper television, computer, and smartphone screens.
Researchers meticulously measured the optical birefringence of highly aligned cellulose nanofibers, paving the way for sharper television, computer, and smartphone screens.
Apr 18th, 2019
Read more Scientists have designed an ultrathin display that can project dynamic, multi-colored, 3D holographic images.
Scientists have designed an ultrathin display that can project dynamic, multi-colored, 3D holographic images.
Apr 18th, 2019
Read more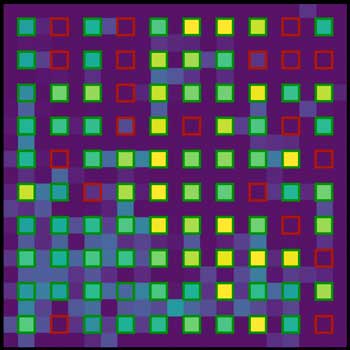 In a new study, scientists showed that it could load groups of individual atoms into large grids with an efficiency unmatched by existing methods.
In a new study, scientists showed that it could load groups of individual atoms into large grids with an efficiency unmatched by existing methods.
Apr 18th, 2019
Read more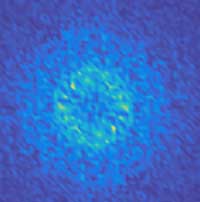 Researchers have discovered how magnets recover after being blasted by a laser. It turns out, they act a bit like oil and water in a jar.
Researchers have discovered how magnets recover after being blasted by a laser. It turns out, they act a bit like oil and water in a jar.
Apr 18th, 2019
Read more Researchers have improved their recycling process that regenerates degraded cathodes from spent lithium-ion batteries. The new process is safer and uses less energy than their previous method in restoring cathodes to their original capacity and cycle performance.
Researchers have improved their recycling process that regenerates degraded cathodes from spent lithium-ion batteries. The new process is safer and uses less energy than their previous method in restoring cathodes to their original capacity and cycle performance.
Apr 18th, 2019
Read more