Metallic nano-particles light up another path towards eco-friendly catalysts
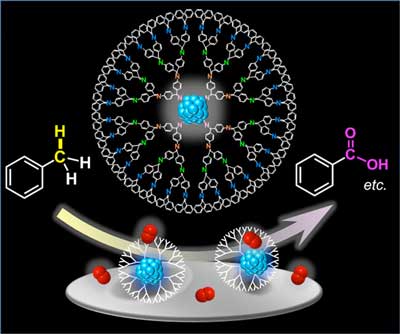 Platinum clusters consisting 19 atoms perform 50 times higher catalytic activity.
Platinum clusters consisting 19 atoms perform 50 times higher catalytic activity.
Nov 16th, 2018
Read more
 Platinum clusters consisting 19 atoms perform 50 times higher catalytic activity.
Platinum clusters consisting 19 atoms perform 50 times higher catalytic activity.
Nov 16th, 2018
Read more Next-generation composites may monitor their own structural health.
Next-generation composites may monitor their own structural health.
Nov 16th, 2018
Read more A new technique that mimics biological nanofiber arrays can grow chain-like molecules into 3D nanostructures.
A new technique that mimics biological nanofiber arrays can grow chain-like molecules into 3D nanostructures.
Nov 15th, 2018
Read more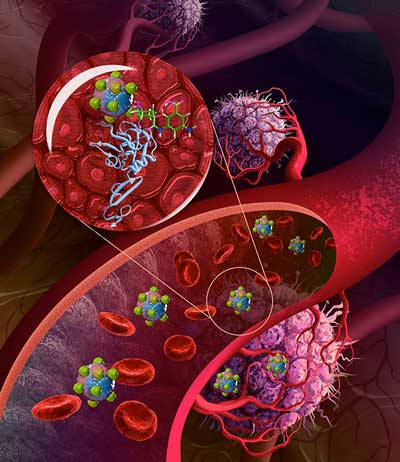 Improved drug delivery method is aimed at making chemotherapy easier to help treat people with various tumors.
Improved drug delivery method is aimed at making chemotherapy easier to help treat people with various tumors.
Nov 15th, 2018
Read more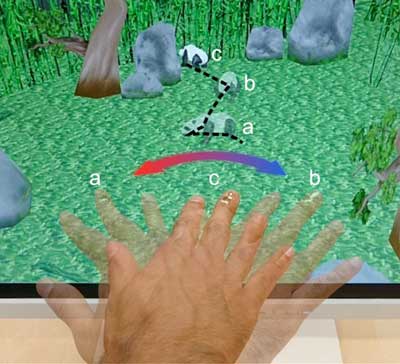 Researchers have developed an electronic skin (e-skin) with magnetosensitive capabilities, sensitive enough to detect and digitize body motion in the Earth's magnetic field. As this e-skin is extremely thin and malleable, it can easily be affixed to human skin to create a bionic analog of a compass. This might not only help people with orientation issues, but also facilitate interaction with objects in virtual and augmented reality.
Researchers have developed an electronic skin (e-skin) with magnetosensitive capabilities, sensitive enough to detect and digitize body motion in the Earth's magnetic field. As this e-skin is extremely thin and malleable, it can easily be affixed to human skin to create a bionic analog of a compass. This might not only help people with orientation issues, but also facilitate interaction with objects in virtual and augmented reality.
Nov 15th, 2018
Read more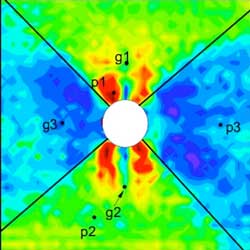 Thanks to one peculiar characteristic of carbon nanotubes, engineers will soon be able to measure the accumulated strain in an airplane, a bridge or a pipeline - or just about anything - over the entire surface or down to microscopic levels.
Thanks to one peculiar characteristic of carbon nanotubes, engineers will soon be able to measure the accumulated strain in an airplane, a bridge or a pipeline - or just about anything - over the entire surface or down to microscopic levels.
Nov 15th, 2018
Read more Radical techniques in electron microscopy could revolutionise studies in physics, biochemistry, materials and more.
Radical techniques in electron microscopy could revolutionise studies in physics, biochemistry, materials and more.
Nov 15th, 2018
Read more Scientists with different scientific backgrounds, working in the field of nanotechnology, have cooperated with the main objective to provide as broad an overview as possible about the young and rapidly evolving field of nanoinformatics.
Scientists with different scientific backgrounds, working in the field of nanotechnology, have cooperated with the main objective to provide as broad an overview as possible about the young and rapidly evolving field of nanoinformatics.
Nov 15th, 2018
Read more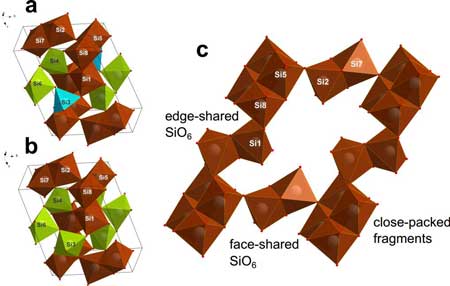 Scientists discovered 'impossible' modifications of silica coesite-IV and coesite-V - i.e., materials that are not supposed to exist.
Scientists discovered 'impossible' modifications of silica coesite-IV and coesite-V - i.e., materials that are not supposed to exist.
Nov 15th, 2018
Read more Researchers have developed a version of a hyperbolical metamaterial in colloidal form. They may find applications in plasmon-enhanced spectroscopy, nanolasers, design of nonlinear phenomena, photothermal conversions, and hot?electron generation.
Researchers have developed a version of a hyperbolical metamaterial in colloidal form. They may find applications in plasmon-enhanced spectroscopy, nanolasers, design of nonlinear phenomena, photothermal conversions, and hot?electron generation.
Nov 15th, 2018
Read more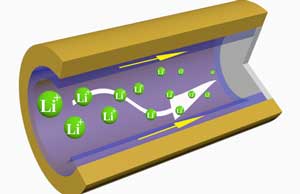 Dramatically longer lasting, faster charging, and safer lithium metal batteries may be possible thanks to a three-dimensional cross-linked polymer sponge that attaches to the metal plating of a battery anode.
Dramatically longer lasting, faster charging, and safer lithium metal batteries may be possible thanks to a three-dimensional cross-linked polymer sponge that attaches to the metal plating of a battery anode.
Nov 14th, 2018
Read more A research team has discovered that different metasurfaces exhibit the same behavior provided a symmetry breaking is introduced to their unit cells 'meta-atoms'. Asymmetry of meta-atoms results in high-quality (high Q) resonances in the transmittance spectra of metasurfaces.
A research team has discovered that different metasurfaces exhibit the same behavior provided a symmetry breaking is introduced to their unit cells 'meta-atoms'. Asymmetry of meta-atoms results in high-quality (high Q) resonances in the transmittance spectra of metasurfaces.
Nov 14th, 2018
Read more A new thermal transistor could help conduct heat away from delicate electronic components and also insulate them against chip and circuit failure.
A new thermal transistor could help conduct heat away from delicate electronic components and also insulate them against chip and circuit failure.
Nov 14th, 2018
Read more Scientists combine graphene foam, epoxy into tough, conductive composite.
Scientists combine graphene foam, epoxy into tough, conductive composite.
Nov 14th, 2018
Read more Scientists have discovered that by combining copper with organic molecules, they can create metal complexes that exhibit photoluminescence. What's more, by varying the sizes of those organic molecules, they can control the brightness of the emitted light.
Scientists have discovered that by combining copper with organic molecules, they can create metal complexes that exhibit photoluminescence. What's more, by varying the sizes of those organic molecules, they can control the brightness of the emitted light.
Nov 14th, 2018
Read more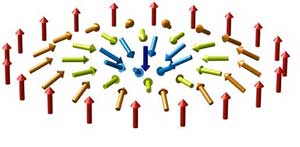 Researchers report the discovery of small and ferroelectrically tunable skyrmions. This work introduces new compelling advantages that bring skyrmion research a step closer to application.
Researchers report the discovery of small and ferroelectrically tunable skyrmions. This work introduces new compelling advantages that bring skyrmion research a step closer to application.
Nov 14th, 2018
Read more