Scientists developed a technique that 'exfoliates' aggregated clumps of CNTs and disperses them in solvents. It involves wrapping the tubes in a polymer using a bond that does not involve the sharing of electrons. The technique is called non-covalent polymer wrapping.
Mar 30th, 2015
Read more
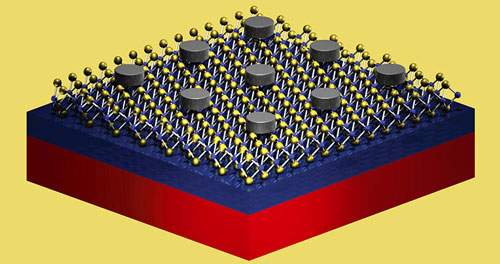 Research team increases material's light emission by twelve times.
Research team increases material's light emission by twelve times.
Mar 27th, 2015
Read more
Scientists have devised and demonstrated a new, shape-shifting probe which is capable of sensitive, high-resolution remote biological sensing that is not possible with current technology.
Mar 27th, 2015
Read more
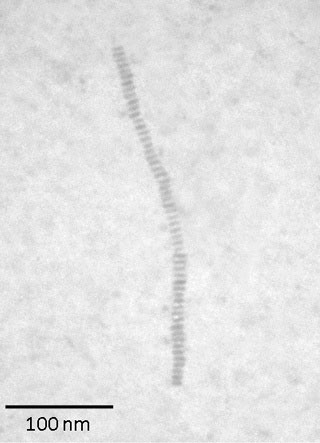 Researchers have developed a novel technique for crafting nanometer-scale necklaces based on tiny star-like structures threaded onto a polymeric backbone. The technique could provide a new way to produce hybrid organic-inorganic shish kebab structures from semiconducting, magnetic, ferroelectric and other materials that may afford useful nanoscale properties.
Researchers have developed a novel technique for crafting nanometer-scale necklaces based on tiny star-like structures threaded onto a polymeric backbone. The technique could provide a new way to produce hybrid organic-inorganic shish kebab structures from semiconducting, magnetic, ferroelectric and other materials that may afford useful nanoscale properties.
Mar 27th, 2015
Read more
 The results of a three-year research project mean that advanced manufacturing companies will have on-line access to a new method of testing the accuracy of their crucial measurement software.
The results of a three-year research project mean that advanced manufacturing companies will have on-line access to a new method of testing the accuracy of their crucial measurement software.
Mar 27th, 2015
Read more
Incorporating colored pigments in nanocomposites make coatings possible which are not only protective but also deliver additional visual information via their coloration.
Mar 27th, 2015
Read more
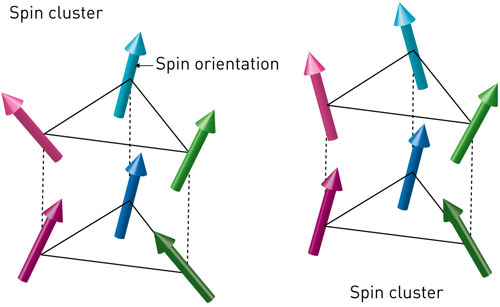 A deeper understanding of quantum fluctuations in 'frustrated' layered magnetic crystals could speed the development of devices that probe real-world systems using quantum effects.
A deeper understanding of quantum fluctuations in 'frustrated' layered magnetic crystals could speed the development of devices that probe real-world systems using quantum effects.
Mar 27th, 2015
Read more
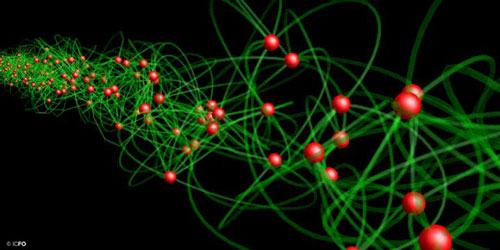 Scientists have detected, for the first time, entanglement among individual photon pairs in a beam of squeezed light.
Scientists have detected, for the first time, entanglement among individual photon pairs in a beam of squeezed light.
Mar 27th, 2015
Read more
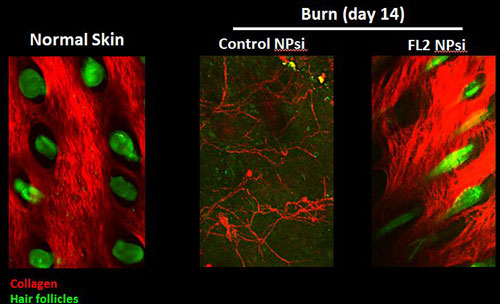 An experimental therapy cuts in half the time it takes to heal wounds compared to no treatment at all.
An experimental therapy cuts in half the time it takes to heal wounds compared to no treatment at all.
Mar 26th, 2015
Read more
The bacteria can load electrons onto and discharge electrons from microscopic particles of magnetite. This discovery holds out the potential of using this mechanism to help clean up environmental pollution, and other bioengineering applications.
Mar 26th, 2015
Read more
 The latest DNA nanodevices - including a robot with movable arms, a book that opens and closes, a switchable gear, and an actuator - may be intriguing in their own right, but that's not the point. They demonstrate a breakthrough in the science of using DNA as a programmable building material for nanometer-scale structures and machines.
The latest DNA nanodevices - including a robot with movable arms, a book that opens and closes, a switchable gear, and an actuator - may be intriguing in their own right, but that's not the point. They demonstrate a breakthrough in the science of using DNA as a programmable building material for nanometer-scale structures and machines.
Mar 26th, 2015
Read more
 Researchers describe methods for making nanoribbons and nanoplates from a compound called silicon telluride. The materials are pure, p-type semiconductors (positive charge carriers) that could be used in a variety of electronic and optical devices.
Researchers describe methods for making nanoribbons and nanoplates from a compound called silicon telluride. The materials are pure, p-type semiconductors (positive charge carriers) that could be used in a variety of electronic and optical devices.
Mar 26th, 2015
Read more
 In biology, materials science and the energy sciences, structural information provides important insights into the understanding of matter. The link between a structure and its properties can suggest new avenues for designed improvements of synthetic materials or provide new fundamental insights in biology and medicine at the molecular level.
In biology, materials science and the energy sciences, structural information provides important insights into the understanding of matter. The link between a structure and its properties can suggest new avenues for designed improvements of synthetic materials or provide new fundamental insights in biology and medicine at the molecular level.
Mar 26th, 2015
Read more
 A new theory gives new insights into the transport of liquid flowing along the surface under applied electric field.
A new theory gives new insights into the transport of liquid flowing along the surface under applied electric field.
Mar 26th, 2015
Read more
Nanoparticles are specifically adapted to the particular application by Small Molecule Surface Modification (SMSM). Thereby surfaces of workpieces or mouldings are expected to exhibit several different functions at one and the same time.
Mar 26th, 2015
Read more
 A plastic used in filters and tubing has an unusual trait: It can produce electricity when pulled or pressed. This ability has been used in small ways, but now researchers are coaxing fibers of the material to make even more electricity for a wider range of applications from green energy to 'artificial muscles'.
A plastic used in filters and tubing has an unusual trait: It can produce electricity when pulled or pressed. This ability has been used in small ways, but now researchers are coaxing fibers of the material to make even more electricity for a wider range of applications from green energy to 'artificial muscles'.
Mar 26th, 2015
Read more












 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed