 New materials are being synthesized by twisting and stacking atomically thin layers. To bring it all under one roof, physicists propose this field of research be called 'electron quantum metamaterials'.
New materials are being synthesized by twisting and stacking atomically thin layers. To bring it all under one roof, physicists propose this field of research be called 'electron quantum metamaterials'.
Nov 5th, 2018
Read more
 The Life NanoEXPLORE project addresses current data gaps and barriers limiting the implementation of REACH regulation and the use of human bio-monitoring data in the protection of human health and the environment when dealing with particles in the nanometer range.
The Life NanoEXPLORE project addresses current data gaps and barriers limiting the implementation of REACH regulation and the use of human bio-monitoring data in the protection of human health and the environment when dealing with particles in the nanometer range.
Nov 5th, 2018
Read more
 Researchers have developed a photocatalytic system based on a material in the class of metal-organic frameworks. The system can be used to degrade pollutants present in water while simultaneously producing hydrogen that can be captured and used further.
Researchers have developed a photocatalytic system based on a material in the class of metal-organic frameworks. The system can be used to degrade pollutants present in water while simultaneously producing hydrogen that can be captured and used further.
Nov 5th, 2018
Read more
 Researchers have designed semiconductor nanoplatelets with a broadened range of colors to improve LCD and LED screens.
Researchers have designed semiconductor nanoplatelets with a broadened range of colors to improve LCD and LED screens.
Nov 5th, 2018
Read more
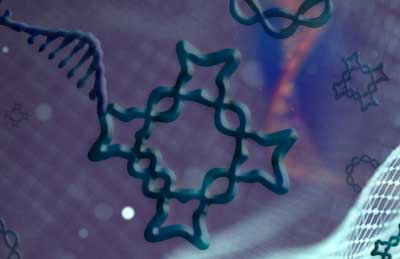 Researchers describe a method for coaxing segments of single-stranded DNA into complex 2- and 3D knotted structures.
Researchers describe a method for coaxing segments of single-stranded DNA into complex 2- and 3D knotted structures.
Nov 3rd, 2018
Read more
 Scientists developed specially coated nanometer-sized vehicles that can be actively moved through dense tissue like the vitreous of the eye.
Scientists developed specially coated nanometer-sized vehicles that can be actively moved through dense tissue like the vitreous of the eye.
Nov 2nd, 2018
Read more
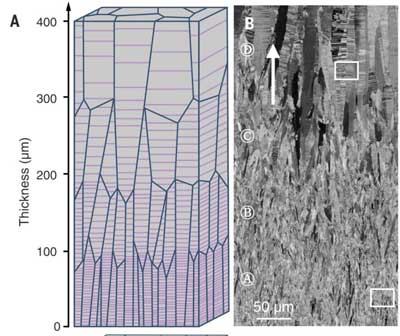 Researchers have found a new way to use nanotwins - tiny linear boundaries in a metal's atomic lattice that have identical crystalline structures on either side - to make stronger metals.
Researchers have found a new way to use nanotwins - tiny linear boundaries in a metal's atomic lattice that have identical crystalline structures on either side - to make stronger metals.
Nov 2nd, 2018
Read more
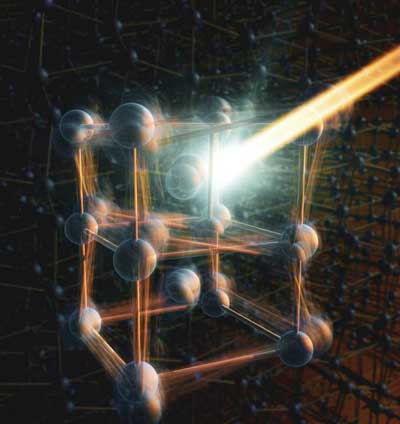 Scientists discover that disorder is part of the structural transition of Vanadium Dioxide from an insulator state to a metallic state at extremely small time resolutions This provides a new perspective on how to control matter, especially in the field of superconductivity, which could have major implications for nanotechnology and optoelectronics.
Scientists discover that disorder is part of the structural transition of Vanadium Dioxide from an insulator state to a metallic state at extremely small time resolutions This provides a new perspective on how to control matter, especially in the field of superconductivity, which could have major implications for nanotechnology and optoelectronics.
Nov 2nd, 2018
Read more
 A new report describes the structural and physical properties of a group of two-dimensional materials based on polycyclic molecules called circulenes. The possibility of flexible design and variable properties of these materials make them suitable for nanoelectronics.
A new report describes the structural and physical properties of a group of two-dimensional materials based on polycyclic molecules called circulenes. The possibility of flexible design and variable properties of these materials make them suitable for nanoelectronics.
Nov 2nd, 2018
Read more
 Spherical nanoparticles with a high content of dislocations (defects in the crystal and metal lattice) and with accumulated internal energy will significantly increase the chemical potential and provide conditions for high physicochemical activity of particles.
Spherical nanoparticles with a high content of dislocations (defects in the crystal and metal lattice) and with accumulated internal energy will significantly increase the chemical potential and provide conditions for high physicochemical activity of particles.
Nov 2nd, 2018
Read more
 For the first time, the targeted functionalization of carbon-based nanostructures allows the direct mapping of current paths, thereby paving the way for novel quantum devices.
For the first time, the targeted functionalization of carbon-based nanostructures allows the direct mapping of current paths, thereby paving the way for novel quantum devices.
Nov 2nd, 2018
Read more
 World-first demonstration of topologically protected biphoton states.
World-first demonstration of topologically protected biphoton states.
Nov 1st, 2018
Read more
 Revealed for the first time by a new X-ray laser technique, their surprisingly unruly response has profound implications for designing and controlling materials.
Revealed for the first time by a new X-ray laser technique, their surprisingly unruly response has profound implications for designing and controlling materials.
Nov 1st, 2018
Read more
 Physicists have visualized for the first time how atoms behave in between graphene and a substrate. This insight could be instrumental for future implementations of industrial graphene production.
Physicists have visualized for the first time how atoms behave in between graphene and a substrate. This insight could be instrumental for future implementations of industrial graphene production.
Nov 1st, 2018
Read more
 Engineers use a cost-effective process to make strong, flexible films that allow light in but keep electromagnetic interference out.
Engineers use a cost-effective process to make strong, flexible films that allow light in but keep electromagnetic interference out.
Nov 1st, 2018
Read more
 Environmentally friendly materials for producing smart textiles from conductive graphene biopolymer nanofibrous fabrics.
Environmentally friendly materials for producing smart textiles from conductive graphene biopolymer nanofibrous fabrics.
Nov 1st, 2018
Read more
 New materials are being synthesized by twisting and stacking atomically thin layers. To bring it all under one roof, physicists propose this field of research be called 'electron quantum metamaterials'.
New materials are being synthesized by twisting and stacking atomically thin layers. To bring it all under one roof, physicists propose this field of research be called 'electron quantum metamaterials'.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed