Heat used to transform antiskyrmions to skyrmions and back
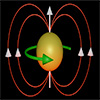 Experiment shows heat and magnetic fields transform spin textures in devices, achieving low-energy skyrmion shifts at room temperature.
Experiment shows heat and magnetic fields transform spin textures in devices, achieving low-energy skyrmion shifts at room temperature.
Mar 19th, 2024
Read more
 Experiment shows heat and magnetic fields transform spin textures in devices, achieving low-energy skyrmion shifts at room temperature.
Experiment shows heat and magnetic fields transform spin textures in devices, achieving low-energy skyrmion shifts at room temperature.
Mar 19th, 2024
Read more Breakthrough in photoluminescence: Achieving >99% PLQY in gold-copper nanoclusters at room temperature, enabling advances in biomedical imaging.
Breakthrough in photoluminescence: Achieving >99% PLQY in gold-copper nanoclusters at room temperature, enabling advances in biomedical imaging.
Mar 18th, 2024
Read more Leafhoppers| buckyball-shaped brochosomes with through-holes reduce light reflection, showcasing unique antireflection properties in nature.
Leafhoppers| buckyball-shaped brochosomes with through-holes reduce light reflection, showcasing unique antireflection properties in nature.
Mar 18th, 2024
Read more This finding is expected to increase capacity of rechargeable batteries for electric vehicles and telecommunications equipment with two to few-layer graphene electrodes.
This finding is expected to increase capacity of rechargeable batteries for electric vehicles and telecommunications equipment with two to few-layer graphene electrodes.
Mar 18th, 2024
Read more Researchers have published groundbreaking work demonstrating, for the first time, the ability to recycle high-performance carbon nanotube sheets while preserving their shape, structural alignment, mechanical and electrical properties, and intrinsic flexibility.
Researchers have published groundbreaking work demonstrating, for the first time, the ability to recycle high-performance carbon nanotube sheets while preserving their shape, structural alignment, mechanical and electrical properties, and intrinsic flexibility.
Mar 18th, 2024
Read more Scientists have synthesized bulk van der Waals materials at near-room temperature (ranging from room temperature to 60 C), significantly reducing the energy consumption required for their fabrication by at least one order of magnitude.
Scientists have synthesized bulk van der Waals materials at near-room temperature (ranging from room temperature to 60 C), significantly reducing the energy consumption required for their fabrication by at least one order of magnitude.
Mar 18th, 2024
Read more The flexible perovskite panels have electrodes made of tiny carbon nanotubes. These can generate more power with greater efficiency and at a cost 70% lower than existing solar panels.
The flexible perovskite panels have electrodes made of tiny carbon nanotubes. These can generate more power with greater efficiency and at a cost 70% lower than existing solar panels.
Mar 18th, 2024
Read more Materials scientists are shedding light on the intricate growth processes of 2D crystals, paving the way for controlled synthesis of these materials with unprecedented precision.
Materials scientists are shedding light on the intricate growth processes of 2D crystals, paving the way for controlled synthesis of these materials with unprecedented precision.
Mar 15th, 2024
Read more A study introduces a novel method for calibrating the spring constant of FluidFM micropipette cantilevers, crucial for the accurate measurement of forces in microfluidic environments. This method addresses the limitations of current calibration techniques, offering a significant advancement in the field of force microscopy.
A study introduces a novel method for calibrating the spring constant of FluidFM micropipette cantilevers, crucial for the accurate measurement of forces in microfluidic environments. This method addresses the limitations of current calibration techniques, offering a significant advancement in the field of force microscopy.
Mar 15th, 2024
Read more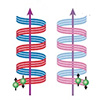 A printable organic polymer that assembles into chiral structures when printed has enabled researchers to reliably measure the amount of charge produced in spin-to-charge conversion within a spintronic material at room temperature.
A printable organic polymer that assembles into chiral structures when printed has enabled researchers to reliably measure the amount of charge produced in spin-to-charge conversion within a spintronic material at room temperature.
Mar 15th, 2024
Read more New etching process could enable the integration of electronics and photonics on a single chip, combining light sources and optical fibers.
New etching process could enable the integration of electronics and photonics on a single chip, combining light sources and optical fibers.
Mar 15th, 2024
Read more A newly developed method enables the synthesis of hundreds of new 2D materials.
A newly developed method enables the synthesis of hundreds of new 2D materials.
Mar 15th, 2024
Read more Tumor-specific drug release through controlled endosomal escape.
Tumor-specific drug release through controlled endosomal escape.
Mar 14th, 2024
Read more Scientists developed a new nanoparticle treatment that significantly improves outcomes for severe burn-induced intestinal barrier disruption.
Scientists developed a new nanoparticle treatment that significantly improves outcomes for severe burn-induced intestinal barrier disruption.
Mar 14th, 2024
Read more This breakthrough addresses crucial technological challenges related to the lattice mismatch problem in the growth of high-quality heterostructure semiconductors, leading to enhanced carrier transport and photoelectric properties.
This breakthrough addresses crucial technological challenges related to the lattice mismatch problem in the growth of high-quality heterostructure semiconductors, leading to enhanced carrier transport and photoelectric properties.
Mar 14th, 2024
Read more Physicists choreograph the shift of a quantized electronic energy level with atomic oscillations faster than a trillionth of a second.
Physicists choreograph the shift of a quantized electronic energy level with atomic oscillations faster than a trillionth of a second.
Mar 14th, 2024
Read more