Showing Spotlights 1 - 8 of 137 in category All (newest first):
 Nanoparticles have shown a lot of promise in biomedical applications. However, accumulation of nanoparticles in the liver is a major concern, and may be one of the greatest barriers to the widespread adoption of nanoparticles in the clinic. This is especially true for metallic nanoparticles, since the long-term effects of their accumulation in the liver has not been widely studied.
A team of researchers has looked to nature for inspiration in solving this problem. They decided to use biliverdin, a bile pigment, as the building block for their nanoparticles.
Nanoparticles have shown a lot of promise in biomedical applications. However, accumulation of nanoparticles in the liver is a major concern, and may be one of the greatest barriers to the widespread adoption of nanoparticles in the clinic. This is especially true for metallic nanoparticles, since the long-term effects of their accumulation in the liver has not been widely studied.
A team of researchers has looked to nature for inspiration in solving this problem. They decided to use biliverdin, a bile pigment, as the building block for their nanoparticles.
Nov 18th, 2019
 The increase in production and applications of metal-oxide nanoparticles (MO-NPs) raises concerns for the environment as well as for human health. The terrestrial environment, especially soil, is expected to be the largest repository for environmentally released MO-NPs. Therefore, the accurate determination of environmentally released MO-NPs is imperative to assess the real-time scenario of toxicity to soil biota and the threat to human beings via the food chain.
The increase in production and applications of metal-oxide nanoparticles (MO-NPs) raises concerns for the environment as well as for human health. The terrestrial environment, especially soil, is expected to be the largest repository for environmentally released MO-NPs. Therefore, the accurate determination of environmentally released MO-NPs is imperative to assess the real-time scenario of toxicity to soil biota and the threat to human beings via the food chain.
Oct 21st, 2019
 Nanoparticle characterization is a broad and complex discipline. Even today, researchers are still challenged by the task of determining the physicochemical properties of nanoparticles and exploring their structure-function relationships. A recent review article provides a set of guidelines to investigate and characterize the key parameters defining a nanoparticle sample, namely size, shape, surface charge, and porosity. It also provides recommendations of how the physicochemical parameters of nanoparticles should be investigated, and how to characterize these key properties in different environments according to the intended nanoparticle use.
Nanoparticle characterization is a broad and complex discipline. Even today, researchers are still challenged by the task of determining the physicochemical properties of nanoparticles and exploring their structure-function relationships. A recent review article provides a set of guidelines to investigate and characterize the key parameters defining a nanoparticle sample, namely size, shape, surface charge, and porosity. It also provides recommendations of how the physicochemical parameters of nanoparticles should be investigated, and how to characterize these key properties in different environments according to the intended nanoparticle use.
Jun 5th, 2019
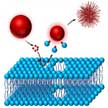 Significant bioaccumulation of nanomaterials in the liver via inadvertent or systemic exposure, as well as the lack of a mechanistic knowledge that describes the hazard potential of metal oxide nanoparticles in liver cells, prompted researchers to comprehensively explore metal oxide nanoparticle interactions with major liver cells, including phagocytic cells and hepatocytes. They now report on the assessment of the toxicity of 29 metal oxide nanoparticles in liver macrophages (Kupffer cells) and hepatocytes.
Significant bioaccumulation of nanomaterials in the liver via inadvertent or systemic exposure, as well as the lack of a mechanistic knowledge that describes the hazard potential of metal oxide nanoparticles in liver cells, prompted researchers to comprehensively explore metal oxide nanoparticle interactions with major liver cells, including phagocytic cells and hepatocytes. They now report on the assessment of the toxicity of 29 metal oxide nanoparticles in liver macrophages (Kupffer cells) and hepatocytes.
May 18th, 2018
 Currently, most graphene-based innovations are not yet at the level of large-scale commercial production. But public and private investments into graphene and its applications in products are large and whichever production methods eventually turn out to be successful, exposure to humans or the environment somewhere along the value chain or life-cycle of the material or product should be anticipated timely. A new review paper offers suggestions on how potential nanospecific safety issues can be addressed, by who and at what stage of the innovation process.
Currently, most graphene-based innovations are not yet at the level of large-scale commercial production. But public and private investments into graphene and its applications in products are large and whichever production methods eventually turn out to be successful, exposure to humans or the environment somewhere along the value chain or life-cycle of the material or product should be anticipated timely. A new review paper offers suggestions on how potential nanospecific safety issues can be addressed, by who and at what stage of the innovation process.
Oct 16th, 2017
 The optical manipulation of plasmonic nanoparticles has advantages for applications such as nanofabrication, drug delivery and biosensing. To that end, researchers have been developing techniques for the reversible assembly of plasmonic nanoparticles that can be used to modulate their structural, electrical and optical properties. The latest such technique is a low-power assembly that is enabled by thermophoretic migration of nanoparticles due to the plasmon-enhanced photothermal effect and the associated enhanced local electric field over a plasmonic substrate.
The optical manipulation of plasmonic nanoparticles has advantages for applications such as nanofabrication, drug delivery and biosensing. To that end, researchers have been developing techniques for the reversible assembly of plasmonic nanoparticles that can be used to modulate their structural, electrical and optical properties. The latest such technique is a low-power assembly that is enabled by thermophoretic migration of nanoparticles due to the plasmon-enhanced photothermal effect and the associated enhanced local electric field over a plasmonic substrate.
Sep 21st, 2016
 In the fields of toxicology and ecotoxicology, doses are commonly expressed in weight concentration for non-soluble compounds because this is very convenient experimentally. However, when it comes to nanoparticles, the weight of the nanomaterial is not a relevant parameter, especially when it is required to compare different kinds of nanoparticles because their density is very different. Researchers have now shown that the usual approach based on mass concentrations fails to compare the toxicities of different engineered carbon nanoparticles.
In the fields of toxicology and ecotoxicology, doses are commonly expressed in weight concentration for non-soluble compounds because this is very convenient experimentally. However, when it comes to nanoparticles, the weight of the nanomaterial is not a relevant parameter, especially when it is required to compare different kinds of nanoparticles because their density is very different. Researchers have now shown that the usual approach based on mass concentrations fails to compare the toxicities of different engineered carbon nanoparticles.
May 24th, 2016
 Novel materials designed and fabricated with the help of nanotechnologies offer the promise of radical technological development. Many of these will improve our quality of life, and develop our economies, but all will be measured against the overarching principle that we do not make some error, and harm ourselves and our environment by exposure to new forms of hazard. A publication explores recent developments in nanomaterials research, and possibilities for safe, practical and resource-efficient applications.
Novel materials designed and fabricated with the help of nanotechnologies offer the promise of radical technological development. Many of these will improve our quality of life, and develop our economies, but all will be measured against the overarching principle that we do not make some error, and harm ourselves and our environment by exposure to new forms of hazard. A publication explores recent developments in nanomaterials research, and possibilities for safe, practical and resource-efficient applications.
Mar 27th, 2015
 Nanoparticles have shown a lot of promise in biomedical applications. However, accumulation of nanoparticles in the liver is a major concern, and may be one of the greatest barriers to the widespread adoption of nanoparticles in the clinic. This is especially true for metallic nanoparticles, since the long-term effects of their accumulation in the liver has not been widely studied.
A team of researchers has looked to nature for inspiration in solving this problem. They decided to use biliverdin, a bile pigment, as the building block for their nanoparticles.
Nanoparticles have shown a lot of promise in biomedical applications. However, accumulation of nanoparticles in the liver is a major concern, and may be one of the greatest barriers to the widespread adoption of nanoparticles in the clinic. This is especially true for metallic nanoparticles, since the long-term effects of their accumulation in the liver has not been widely studied.
A team of researchers has looked to nature for inspiration in solving this problem. They decided to use biliverdin, a bile pigment, as the building block for their nanoparticles.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





