Showing Spotlights 1 - 8 of 22 in category All (newest first):
 Recent advances allow electronic materials to reshape themselves, leading researchers to envision artificial organisms made of self-assembling, self-repairing electronic modules.
Recent advances allow electronic materials to reshape themselves, leading researchers to envision artificial organisms made of self-assembling, self-repairing electronic modules.
Nov 11th, 2023
 Using a combination of 3D printed template and self-assembly allows the fabrication of materials with complex Lego-like models with tailorable feature size crossing a record seven orders of magnitude - from nanometers to centimeters. This allows the fabrication of graphene structures with tunable mechanical properties, from super rigid (modules one order of magnitude higher than other ultralight materials) to superelastic (able to recover from extreme 95% compression).
Using a combination of 3D printed template and self-assembly allows the fabrication of materials with complex Lego-like models with tailorable feature size crossing a record seven orders of magnitude - from nanometers to centimeters. This allows the fabrication of graphene structures with tunable mechanical properties, from super rigid (modules one order of magnitude higher than other ultralight materials) to superelastic (able to recover from extreme 95% compression).
May 10th, 2022
 The ultimate challenge of nanotechnology is to control the structure of matter with atomic precision. The better we are at shaping and structuring material on a small scale, the more powerful technology we can dream of. Unfortunately, the atomic scale is entirely out of range for conventional patterning. Researchers now report that they have achieved nanoscale self-assembly within a two-dimensional layer. Dosing of ethylene and borazine near a hot iridium surface, leads for self-organising of a two-dimensional superlattice of graphene dots.
The ultimate challenge of nanotechnology is to control the structure of matter with atomic precision. The better we are at shaping and structuring material on a small scale, the more powerful technology we can dream of. Unfortunately, the atomic scale is entirely out of range for conventional patterning. Researchers now report that they have achieved nanoscale self-assembly within a two-dimensional layer. Dosing of ethylene and borazine near a hot iridium surface, leads for self-organising of a two-dimensional superlattice of graphene dots.
Jun 29th, 2017
 Researchers have now shown that, by varying the shape of magnetite nanoparticles, they can control the nature of the self-assembled structures as the nanoparticles assemble. This new work provides guidelines for the design of new self-assembled materials. Self-assembly of nanoparticles driven by competing forces can result in truly unique structures, the diversity and complexity of which could be particularly striking if the building blocks were simultaneously coupled by short- and long-range forces of different symmetries.
Researchers have now shown that, by varying the shape of magnetite nanoparticles, they can control the nature of the self-assembled structures as the nanoparticles assemble. This new work provides guidelines for the design of new self-assembled materials. Self-assembly of nanoparticles driven by competing forces can result in truly unique structures, the diversity and complexity of which could be particularly striking if the building blocks were simultaneously coupled by short- and long-range forces of different symmetries.
Aug 4th, 2014
 Industrial production processes, by and large, rely on robotic assembly lines that place, package, and connect a variety of disparate components. While the manufacturing world is dominated by robots, there are applications where the established processes of serial 'pick and place' and manipulation of single objects reach scaling limits in terms of throughput, alignment precision, and the minimal component dimension they can handle effectively. By contrast, the emerging methods of engineered self-assembly are massively parallel and have the potential to overcome these scaling limitations.
Industrial production processes, by and large, rely on robotic assembly lines that place, package, and connect a variety of disparate components. While the manufacturing world is dominated by robots, there are applications where the established processes of serial 'pick and place' and manipulation of single objects reach scaling limits in terms of throughput, alignment precision, and the minimal component dimension they can handle effectively. By contrast, the emerging methods of engineered self-assembly are massively parallel and have the potential to overcome these scaling limitations.
Jul 10th, 2014
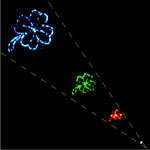 The long-term vision of revolutionary bottom-up nanotechnology is based on two different concepts of molecular assembly technologies. One follows Nature's blueprint, which uses molecular recognition for self-assembly of nanoscale materials and structures; the other is man-made and uses instruments to assemble nanoscale building blocs into larger structures and devices. In contrast, the most common nanoscale fabrication techniques used today, for instance in the sub 100-nanometer semiconductor industry, are top-down approaches where fabrication technologies such as lithography or stamping are used. Here, you create ever smaller structures by starting with a block of material and remove the bits and pieces you don't want until you get the shape and size you do want. While top-down techniques can be highly parallel (semiconductor industry) it is not feasible to control single molecules with them. Using a hybrid approach that combines the precision of an atomic force microscope with the selectivity of DNA interactions, researchers in Germany have successfully demonstrated a technique that fills the gap between top-down and bottom-up since it allows for the control of single molecules with the precision of atomic force microscopy and combines it with the selectivity of self-assembly.
The long-term vision of revolutionary bottom-up nanotechnology is based on two different concepts of molecular assembly technologies. One follows Nature's blueprint, which uses molecular recognition for self-assembly of nanoscale materials and structures; the other is man-made and uses instruments to assemble nanoscale building blocs into larger structures and devices. In contrast, the most common nanoscale fabrication techniques used today, for instance in the sub 100-nanometer semiconductor industry, are top-down approaches where fabrication technologies such as lithography or stamping are used. Here, you create ever smaller structures by starting with a block of material and remove the bits and pieces you don't want until you get the shape and size you do want. While top-down techniques can be highly parallel (semiconductor industry) it is not feasible to control single molecules with them. Using a hybrid approach that combines the precision of an atomic force microscope with the selectivity of DNA interactions, researchers in Germany have successfully demonstrated a technique that fills the gap between top-down and bottom-up since it allows for the control of single molecules with the precision of atomic force microscopy and combines it with the selectivity of self-assembly.
Oct 10th, 2008
 The key to using self-assembly as a controlled and directed nanofabrication process lies in designing the components that are required to self-assemble into desired patterns and functions. Self-assembly reflects information coded in individual components - characteristics such as shape, surface properties, charge, polarizability, magnetic dipole, mass, etc. These characteristics determine the interactions among the components and the whole essence of self-assembly arises from these dynamic properties. In this respect, many self-assembled nanostructures show to be responsive to external stimuli such as temperature, pH, or solvent polarity. An exciting field for nanotechnology researchers is the challenge of extending the scope of nanostructures with stimulus-responsive properties towards the fabrication of 'smart' nanoscale materials. New work by Korean scientists demonstrates that simple addition of small guest molecules triggers reversible structural transformation. The novelty of this research is that, so far, switching of material properties triggered by external stimuli via nanoscale objects had not been realized yet.
The key to using self-assembly as a controlled and directed nanofabrication process lies in designing the components that are required to self-assemble into desired patterns and functions. Self-assembly reflects information coded in individual components - characteristics such as shape, surface properties, charge, polarizability, magnetic dipole, mass, etc. These characteristics determine the interactions among the components and the whole essence of self-assembly arises from these dynamic properties. In this respect, many self-assembled nanostructures show to be responsive to external stimuli such as temperature, pH, or solvent polarity. An exciting field for nanotechnology researchers is the challenge of extending the scope of nanostructures with stimulus-responsive properties towards the fabrication of 'smart' nanoscale materials. New work by Korean scientists demonstrates that simple addition of small guest molecules triggers reversible structural transformation. The novelty of this research is that, so far, switching of material properties triggered by external stimuli via nanoscale objects had not been realized yet.
Aug 11th, 2008
 The use of spontaneous self-assembly as a lithography- and external field-free means to construct well-ordered, often intriguing structures has received much attention for its ease of organizing materials on the nanoscale into ordered structures and producing complex, large-scale structures with small feature sizes. These self-organized structures promise new opportunities for developing miniaturized optical, electronic, optoelectronic, and magnetic devices. An extremely simple route to intriguing structures is the evaporation-induced self-assembly of polymers and nanoparticles from a droplet on a solid substrate. However, flow instabilities within the evaporating droplet often result in non-equilibrium and irregular dissipative structures, e.g., randomly organized convection patterns, stochastically distributed multi-rings, etc. Therefore, fully utilizing evaporation as a simple tool for creating well-ordered structures with numerous technological applications requires precise control over several factors, including evaporative flux, solution concentration, and the interfacial interaction between solute and substrate.
The use of spontaneous self-assembly as a lithography- and external field-free means to construct well-ordered, often intriguing structures has received much attention for its ease of organizing materials on the nanoscale into ordered structures and producing complex, large-scale structures with small feature sizes. These self-organized structures promise new opportunities for developing miniaturized optical, electronic, optoelectronic, and magnetic devices. An extremely simple route to intriguing structures is the evaporation-induced self-assembly of polymers and nanoparticles from a droplet on a solid substrate. However, flow instabilities within the evaporating droplet often result in non-equilibrium and irregular dissipative structures, e.g., randomly organized convection patterns, stochastically distributed multi-rings, etc. Therefore, fully utilizing evaporation as a simple tool for creating well-ordered structures with numerous technological applications requires precise control over several factors, including evaporative flux, solution concentration, and the interfacial interaction between solute and substrate.
Jul 16th, 2008
 Recent advances allow electronic materials to reshape themselves, leading researchers to envision artificial organisms made of self-assembling, self-repairing electronic modules.
Recent advances allow electronic materials to reshape themselves, leading researchers to envision artificial organisms made of self-assembling, self-repairing electronic modules.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





