Showing Spotlights 73 - 80 of 199 in category All (newest first):
 Steam is important for desalination, hygiene systems, and sterilization; and in remote areas where the sun is the only source of energy, being able to generate steam with solar energy could be very useful. Researchers now have found that the mushroom structure can surprisingly benefit solar steam generation. The stipe of the mushroom can serve as efficient water supply path, meanwhile, due to the extreme small ratio of the areas of fibrous stipe and black pileus, only little heat (useless heat loss) conducted into water.
Steam is important for desalination, hygiene systems, and sterilization; and in remote areas where the sun is the only source of energy, being able to generate steam with solar energy could be very useful. Researchers now have found that the mushroom structure can surprisingly benefit solar steam generation. The stipe of the mushroom can serve as efficient water supply path, meanwhile, due to the extreme small ratio of the areas of fibrous stipe and black pileus, only little heat (useless heat loss) conducted into water.
Jun 6th, 2017
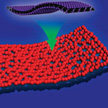 In new work, a research team has developed a general synthesis strategy by employing graphene oxide as a sacrificial template to prepare various 2D holey transition metal oxide (TMO) nanosheets, including mixed metal oxides and simple metal oxides. This approach is universal for the synthesis of various 2D holey TMO nanosheets including mixed transition-metal oxides and simple oxides. This unique holey structure can minimize the restacking of 2D nanosheets and provide more active sites for alkali-ion storage.
In new work, a research team has developed a general synthesis strategy by employing graphene oxide as a sacrificial template to prepare various 2D holey transition metal oxide (TMO) nanosheets, including mixed metal oxides and simple metal oxides. This approach is universal for the synthesis of various 2D holey TMO nanosheets including mixed transition-metal oxides and simple oxides. This unique holey structure can minimize the restacking of 2D nanosheets and provide more active sites for alkali-ion storage.
May 31st, 2017
 The development of porous one-dimensional (1D) nanomaterials with designed properties and architectures has led to significant advances in electrochemical energy storage. A recent review article highlights the state-of-the-art on porous 1D nanostructures, from methodologies for rational and controllable synthesis (electrospinning, liquid phase method, template-assisted method, chemical deposition method, and chemical etching) to their successful application in different types of energy storage devices.
The development of porous one-dimensional (1D) nanomaterials with designed properties and architectures has led to significant advances in electrochemical energy storage. A recent review article highlights the state-of-the-art on porous 1D nanostructures, from methodologies for rational and controllable synthesis (electrospinning, liquid phase method, template-assisted method, chemical deposition method, and chemical etching) to their successful application in different types of energy storage devices.
May 24th, 2017
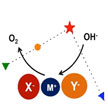 Researchers have proposed and proved an innovative strategy of anionic regulation. The construction of active sites with intrinsic oxygen evolution reaction (OER) is of great significance to overcome the limited efficiency of abundant sustainable energy devices such as fuel cells, rechargeable metal-air batteries, and in water splitting. Anionic regulation of electrocatalysts by modulating the electronic structure of active sites significantly promotes OER performance.
Researchers have proposed and proved an innovative strategy of anionic regulation. The construction of active sites with intrinsic oxygen evolution reaction (OER) is of great significance to overcome the limited efficiency of abundant sustainable energy devices such as fuel cells, rechargeable metal-air batteries, and in water splitting. Anionic regulation of electrocatalysts by modulating the electronic structure of active sites significantly promotes OER performance.
May 18th, 2017
 Supercapacitors, a rapidly emerging type of energy storage device, hold great potential due to their interesting characteristics: high power density, fast charge and discharge rates, and long-term cycling life. However, the use of supercapacitors is severely limited by their low energy density, which is one to two magnitudes lower in comparison with Li-ion batteries. In new work, researchers have developed a low-crystalline FeOOH nanoparticle anode with excellent comprehensive electrochemical performances at both low and high mass loadings as potential replacements for carbon negative electrodes in full supercapacitor devices.
Supercapacitors, a rapidly emerging type of energy storage device, hold great potential due to their interesting characteristics: high power density, fast charge and discharge rates, and long-term cycling life. However, the use of supercapacitors is severely limited by their low energy density, which is one to two magnitudes lower in comparison with Li-ion batteries. In new work, researchers have developed a low-crystalline FeOOH nanoparticle anode with excellent comprehensive electrochemical performances at both low and high mass loadings as potential replacements for carbon negative electrodes in full supercapacitor devices.
May 15th, 2017
 Lithium metal anodes with ultrahigh theoretical specific capacity and the lowest negative electrochemical potential, have been considered the most promising electrode for next-generation rechargeable batteries, including rechargeable Li-S, Li-air batteries, and even Li metal batteries which utilize intercalation compounds as cathodes. Designing a Li plating matrix with a high surface area and lithiophilic surface maybe can help gain a dendrite-free metal anode.
Lithium metal anodes with ultrahigh theoretical specific capacity and the lowest negative electrochemical potential, have been considered the most promising electrode for next-generation rechargeable batteries, including rechargeable Li-S, Li-air batteries, and even Li metal batteries which utilize intercalation compounds as cathodes. Designing a Li plating matrix with a high surface area and lithiophilic surface maybe can help gain a dendrite-free metal anode.
May 5th, 2017
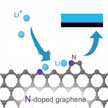
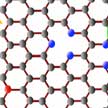 Despite their potential, the practical use of Li-O2 batteries is seriously limited by the corrosion of Li metal by ambient water vapor from air. One way to circumvent this issue is to use an oxygen selective membrane that allows only oxygen into the battery while stopping or slowing water vapor intake. The membrane must be mechanically robust and yet sufficiently thin and light so as to not increase deadweight of the battery. Researchers now have discovered a way to make the thinnest possible oxygen selective membrane using graphene.
Despite their potential, the practical use of Li-O2 batteries is seriously limited by the corrosion of Li metal by ambient water vapor from air. One way to circumvent this issue is to use an oxygen selective membrane that allows only oxygen into the battery while stopping or slowing water vapor intake. The membrane must be mechanically robust and yet sufficiently thin and light so as to not increase deadweight of the battery. Researchers now have discovered a way to make the thinnest possible oxygen selective membrane using graphene.
Apr 25th, 2017
 Researchers have demonstrated an all-stretchable-component sodium-ion full battery, designed and manufactured by integrating stretchable graphene-modified PDMS sponge current collector, sodium-ion conducting gel polymer separator, and elastic PDMS substrate. This first-of-its-kind battery design maintains better mechanical properties compared with most reported designs using one or more rigid components that fail to meet the stretchability requirement for the entire device.
Researchers have demonstrated an all-stretchable-component sodium-ion full battery, designed and manufactured by integrating stretchable graphene-modified PDMS sponge current collector, sodium-ion conducting gel polymer separator, and elastic PDMS substrate. This first-of-its-kind battery design maintains better mechanical properties compared with most reported designs using one or more rigid components that fail to meet the stretchability requirement for the entire device.
Apr 20th, 2017
 Steam is important for desalination, hygiene systems, and sterilization; and in remote areas where the sun is the only source of energy, being able to generate steam with solar energy could be very useful. Researchers now have found that the mushroom structure can surprisingly benefit solar steam generation. The stipe of the mushroom can serve as efficient water supply path, meanwhile, due to the extreme small ratio of the areas of fibrous stipe and black pileus, only little heat (useless heat loss) conducted into water.
Steam is important for desalination, hygiene systems, and sterilization; and in remote areas where the sun is the only source of energy, being able to generate steam with solar energy could be very useful. Researchers now have found that the mushroom structure can surprisingly benefit solar steam generation. The stipe of the mushroom can serve as efficient water supply path, meanwhile, due to the extreme small ratio of the areas of fibrous stipe and black pileus, only little heat (useless heat loss) conducted into water.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





