Showing Spotlights 81 - 88 of 110 in category All (newest first):
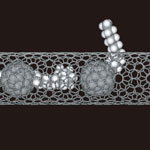 The processes by which molecules pass through pores in thin films and biological membranes are essential for understanding various physical, chemical and biological phenomena. For instance, the fundamental behavior of molecules in porous solids and their transfer through cell membranes necessarily involves a process of molecules passing through pores - knowledge that, for instance, is crucial for the development of nanotechnology-based hydrogen storage materials for fuel tanks. So far, research methods have been based on studying the statistical average of the behavior of groups of molecules; there were no experimental approaches that examined the interaction of a single molecule with a pore. There has been a lack of experimental methods that can obtain information on the structure and orientation of the molecules as they pass through a pore, and their interactions with the pore during passage. Researchers in Japan have now succeeded for the first time in observing a long chain of a fullerene-labeled hydrocarbon passing through a nanometer-size pore in the wall of a carbon nanotube as if it were alive.
The processes by which molecules pass through pores in thin films and biological membranes are essential for understanding various physical, chemical and biological phenomena. For instance, the fundamental behavior of molecules in porous solids and their transfer through cell membranes necessarily involves a process of molecules passing through pores - knowledge that, for instance, is crucial for the development of nanotechnology-based hydrogen storage materials for fuel tanks. So far, research methods have been based on studying the statistical average of the behavior of groups of molecules; there were no experimental approaches that examined the interaction of a single molecule with a pore. There has been a lack of experimental methods that can obtain information on the structure and orientation of the molecules as they pass through a pore, and their interactions with the pore during passage. Researchers in Japan have now succeeded for the first time in observing a long chain of a fullerene-labeled hydrocarbon passing through a nanometer-size pore in the wall of a carbon nanotube as if it were alive.
Sep 22nd, 2008
 Laser-based analytical techniques such as Raman spectroscopy, fluorescence spectroscopy or the state-of-the-art laser-induced breakdown spectroscopy (LIBS) are highly sophisticated techniques to analyze minute amounts of matter with regard to its structure, elemental composition, and other chemical properties. LIBS has been shown to be capable of analyzing extremely small samples with high sensitivity - nanoliter volumes with levels of detection in water of part per million. LIBS works by focusing short laser pulses onto the surface of a sample to create a hot plasma with temperatures of 10,000 - 20,000 C. The plasma emits radiation that allows the observation of the characteristic atomic emission lines of the elements. On the downside, LIBS is complicated by the need for multiple laser pulses to generate a sufficiently hot plasma and the need for focusing and switching a powerful laser, requiring relatively large and expensive instruments.
New research coming out of Drexel University has now shown that light emitted from a new form of cold plasma in liquid permits Optical Emission Spectroscopy (OES) analysis of the elemental composition of solutions within nanoseconds from femtoliter volumes.
Laser-based analytical techniques such as Raman spectroscopy, fluorescence spectroscopy or the state-of-the-art laser-induced breakdown spectroscopy (LIBS) are highly sophisticated techniques to analyze minute amounts of matter with regard to its structure, elemental composition, and other chemical properties. LIBS has been shown to be capable of analyzing extremely small samples with high sensitivity - nanoliter volumes with levels of detection in water of part per million. LIBS works by focusing short laser pulses onto the surface of a sample to create a hot plasma with temperatures of 10,000 - 20,000 C. The plasma emits radiation that allows the observation of the characteristic atomic emission lines of the elements. On the downside, LIBS is complicated by the need for multiple laser pulses to generate a sufficiently hot plasma and the need for focusing and switching a powerful laser, requiring relatively large and expensive instruments.
New research coming out of Drexel University has now shown that light emitted from a new form of cold plasma in liquid permits Optical Emission Spectroscopy (OES) analysis of the elemental composition of solutions within nanoseconds from femtoliter volumes.
Sep 3rd, 2008
 Detecting the presence of a given substance at the molecular level, down to a single molecule, remains a considerable challenge for many nanotechnology sensor applications that range from nanobiotechnology research to environmental monitoring and antiterror or military applications. Currently, chemical functionalization techniques are used to specify what a nanoscale detector will sense. For biological molecules, this might mean developing an antibody/antigen pair, or an alternative synthetically generated ligand. For chemical gases, it is much more challenging to develop the right 'glue' that sticks a given gas to a substrate. The advantage of spectroscopic techniques such as Raman, infrared, and nuclear magnetic resonance spectroscopy is that they are label-free, i.e. they require no preconditioning in order to identify a given analyte. They are also highly selective, capable of distinguishing species that are chemically or functionally very similar. On the downside, spectroscopic methods face enormous challenges in measuring dilute concentrations of an analyte and generally involve the use of large, expensive equipment. This article describes a novel chemical detection technique called nanomechanical resonance spectroscopy.
Detecting the presence of a given substance at the molecular level, down to a single molecule, remains a considerable challenge for many nanotechnology sensor applications that range from nanobiotechnology research to environmental monitoring and antiterror or military applications. Currently, chemical functionalization techniques are used to specify what a nanoscale detector will sense. For biological molecules, this might mean developing an antibody/antigen pair, or an alternative synthetically generated ligand. For chemical gases, it is much more challenging to develop the right 'glue' that sticks a given gas to a substrate. The advantage of spectroscopic techniques such as Raman, infrared, and nuclear magnetic resonance spectroscopy is that they are label-free, i.e. they require no preconditioning in order to identify a given analyte. They are also highly selective, capable of distinguishing species that are chemically or functionally very similar. On the downside, spectroscopic methods face enormous challenges in measuring dilute concentrations of an analyte and generally involve the use of large, expensive equipment. This article describes a novel chemical detection technique called nanomechanical resonance spectroscopy.
Aug 27th, 2008
 X-rays are at the short wavelength, high energy end of the electromagnetic spectrum (only gamma rays have shorter wavelength and carry more energy) and this form of radiation is primarily used for crystallography and diagnostic radiography. Due to the high energy they carry, and because their wavelengths are on the order of the size of atoms, X-rays can penetrate deeply into a material. X-rays play an important role in microscopy and X-ray microscopes have become very powerful scientific instruments for domains such as nanotechnology, materials and life sciences, microelectronics, and chemistry. Depending on the level of energy they carry, X-rays are termed 'hard' - highest energy rays, typically between 8-100 thousand electron volts (keV) - or 'soft' - lower energy rays from roughly 1-8 keV - however this distinction is not well defined. By combining the magnifying power of optical microscopy with the penetrating power of X-rays, X-ray microscopes can generate highly detailed two-dimensional images of features down into the nanoscale, including their internal structure and quantitative chemical information.
X-rays are at the short wavelength, high energy end of the electromagnetic spectrum (only gamma rays have shorter wavelength and carry more energy) and this form of radiation is primarily used for crystallography and diagnostic radiography. Due to the high energy they carry, and because their wavelengths are on the order of the size of atoms, X-rays can penetrate deeply into a material. X-rays play an important role in microscopy and X-ray microscopes have become very powerful scientific instruments for domains such as nanotechnology, materials and life sciences, microelectronics, and chemistry. Depending on the level of energy they carry, X-rays are termed 'hard' - highest energy rays, typically between 8-100 thousand electron volts (keV) - or 'soft' - lower energy rays from roughly 1-8 keV - however this distinction is not well defined. By combining the magnifying power of optical microscopy with the penetrating power of X-rays, X-ray microscopes can generate highly detailed two-dimensional images of features down into the nanoscale, including their internal structure and quantitative chemical information.
Aug 21st, 2008
 More than half a century ago, Erwin Schroedinger, nobel laureate in physics, claimed that it is 'impossible to carry out experiments on single molecules or atoms'. Today, the detection, tracking and study of single molecules and atoms has become an omnipresent tool in biology, chemistry and physics alike. For example, sequencing DNA one base pair (or letter) at a time currently provides the most likely solution to fulfill the quest for a $1,000 human genome. Nevertheless, observation of a single molecule, especially with standard light microscopes requires a good deal of laboratory skills. This is mostly due to the fact that a single molecule only gives a miniscule amount of detectable signal. In fact, people using light as a probe have relied exclusively on the use of fluorescence, the emission of lower energy light following absorption of radiation at a certain energy. In this scheme, the signal from the molecule of interest can be easily separated from residual excitation light or background fluorescence simply by filtering the detected light spectrally and only detecting the color that is emitted by the molecule. In this way, it is possible to suppress unwanted signals from the billions of other molecules that are in the vicinity of the molecule of interest. As powerful as this approach has been, it also has one major limitation: it is only possible to study molecules that are highly fluorescent, i.e. emit lower energy light with high efficiency. Scientists from the ETH Zurich have recently demonstrated a major step towards the detection and study of single molecules in absorption.
More than half a century ago, Erwin Schroedinger, nobel laureate in physics, claimed that it is 'impossible to carry out experiments on single molecules or atoms'. Today, the detection, tracking and study of single molecules and atoms has become an omnipresent tool in biology, chemistry and physics alike. For example, sequencing DNA one base pair (or letter) at a time currently provides the most likely solution to fulfill the quest for a $1,000 human genome. Nevertheless, observation of a single molecule, especially with standard light microscopes requires a good deal of laboratory skills. This is mostly due to the fact that a single molecule only gives a miniscule amount of detectable signal. In fact, people using light as a probe have relied exclusively on the use of fluorescence, the emission of lower energy light following absorption of radiation at a certain energy. In this scheme, the signal from the molecule of interest can be easily separated from residual excitation light or background fluorescence simply by filtering the detected light spectrally and only detecting the color that is emitted by the molecule. In this way, it is possible to suppress unwanted signals from the billions of other molecules that are in the vicinity of the molecule of interest. As powerful as this approach has been, it also has one major limitation: it is only possible to study molecules that are highly fluorescent, i.e. emit lower energy light with high efficiency. Scientists from the ETH Zurich have recently demonstrated a major step towards the detection and study of single molecules in absorption.
Aug 6th, 2008
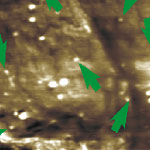 Nanomaterial-based drug-delivery and nanotoxicology are two of the areas that require sophisticated methods and techniques for characterizing, testing and imaging nanoparticulate matter inside the body. Especially the potential risk factors of certain nanomaterials have become a heated topic of discussion recently. Most, if not all, toxicological studies on nanoparticles rely on current methods, practices and terminology as gained and applied in the analysis of micro- and ultrafine particles and mineral fibers. The development of novel imaging techniques that can visualize local populations of nanoparticles at nanometer resolution within the structures of cells - without destroying or damaging the cell - are therefore important. Researchers in the U.S. have now demonstrated that at ultrasonic frequencies, intracellular nanomaterial cause sufficient wave scattering that a probe outside the cell can respond to. This ultrasonic holography technique provides a non-invasive way of looking inside a cell.
Nanomaterial-based drug-delivery and nanotoxicology are two of the areas that require sophisticated methods and techniques for characterizing, testing and imaging nanoparticulate matter inside the body. Especially the potential risk factors of certain nanomaterials have become a heated topic of discussion recently. Most, if not all, toxicological studies on nanoparticles rely on current methods, practices and terminology as gained and applied in the analysis of micro- and ultrafine particles and mineral fibers. The development of novel imaging techniques that can visualize local populations of nanoparticles at nanometer resolution within the structures of cells - without destroying or damaging the cell - are therefore important. Researchers in the U.S. have now demonstrated that at ultrasonic frequencies, intracellular nanomaterial cause sufficient wave scattering that a probe outside the cell can respond to. This ultrasonic holography technique provides a non-invasive way of looking inside a cell.
Jun 23rd, 2008
 'Reverse engineering' is the process of discovering the technological principles of a device or system through analysis of its structure, function and operation, often by taking it apart and analyzing its workings in detail. This approach is a common practice among industrial companies who use it to analyze the competition's products, be it cars or MP3 players, to understand where the latest product improvements come from and how individual components are made. An increasing number of scientists apply a similar approach to nature's own 'micro- and nanotechnology' systems. They believe that learning from nature's designs and engineering successes is more likely to provide the cues for designing practical nanodevices than by simply applying a 'trial and error' approach. The basic idea is that natural materials and systems can be adopted for human use beyond their original purpose in nature. Some examples of 'reverse' biophysics work and have already proven quite useful, for instance the use of individual red blood cells as reliable, ultrasensitive mechanotransducers.
'Reverse engineering' is the process of discovering the technological principles of a device or system through analysis of its structure, function and operation, often by taking it apart and analyzing its workings in detail. This approach is a common practice among industrial companies who use it to analyze the competition's products, be it cars or MP3 players, to understand where the latest product improvements come from and how individual components are made. An increasing number of scientists apply a similar approach to nature's own 'micro- and nanotechnology' systems. They believe that learning from nature's designs and engineering successes is more likely to provide the cues for designing practical nanodevices than by simply applying a 'trial and error' approach. The basic idea is that natural materials and systems can be adopted for human use beyond their original purpose in nature. Some examples of 'reverse' biophysics work and have already proven quite useful, for instance the use of individual red blood cells as reliable, ultrasensitive mechanotransducers.
May 22nd, 2008
 With the advance of nanotechnologies the demand for ever more precise instruments that measure, map and manipulate details at the nanoscale increases as well. For instance, the study of potential distributions with nanoscale resolution becomes increasingly important. In the early days of atomic force microscopy (AFM) the scanning force microscope was used to measure charges, dielectric constants, film thickness of insulating layers, photovoltage, and electrical potential of a given surface. Then, in 1991, the concept of a scanning contact potential microscope was introduced, allowing the simultaneous measurement of topography and contact potential difference. Named the scanning surface potential microscope (SSPM) - also often referred to as Kelvin probe force microscope - this is a variation of the AFM that measures the electrostatic forces (potential) between the probe tip and the surface of a material. Compared with other AFM techniques, the lateral resolution of traditional SSPM, from submicron down to 10 nm, is much lower.
With the advance of nanotechnologies the demand for ever more precise instruments that measure, map and manipulate details at the nanoscale increases as well. For instance, the study of potential distributions with nanoscale resolution becomes increasingly important. In the early days of atomic force microscopy (AFM) the scanning force microscope was used to measure charges, dielectric constants, film thickness of insulating layers, photovoltage, and electrical potential of a given surface. Then, in 1991, the concept of a scanning contact potential microscope was introduced, allowing the simultaneous measurement of topography and contact potential difference. Named the scanning surface potential microscope (SSPM) - also often referred to as Kelvin probe force microscope - this is a variation of the AFM that measures the electrostatic forces (potential) between the probe tip and the surface of a material. Compared with other AFM techniques, the lateral resolution of traditional SSPM, from submicron down to 10 nm, is much lower.
May 12th, 2008
 The processes by which molecules pass through pores in thin films and biological membranes are essential for understanding various physical, chemical and biological phenomena. For instance, the fundamental behavior of molecules in porous solids and their transfer through cell membranes necessarily involves a process of molecules passing through pores - knowledge that, for instance, is crucial for the development of nanotechnology-based hydrogen storage materials for fuel tanks. So far, research methods have been based on studying the statistical average of the behavior of groups of molecules; there were no experimental approaches that examined the interaction of a single molecule with a pore. There has been a lack of experimental methods that can obtain information on the structure and orientation of the molecules as they pass through a pore, and their interactions with the pore during passage. Researchers in Japan have now succeeded for the first time in observing a long chain of a fullerene-labeled hydrocarbon passing through a nanometer-size pore in the wall of a carbon nanotube as if it were alive.
The processes by which molecules pass through pores in thin films and biological membranes are essential for understanding various physical, chemical and biological phenomena. For instance, the fundamental behavior of molecules in porous solids and their transfer through cell membranes necessarily involves a process of molecules passing through pores - knowledge that, for instance, is crucial for the development of nanotechnology-based hydrogen storage materials for fuel tanks. So far, research methods have been based on studying the statistical average of the behavior of groups of molecules; there were no experimental approaches that examined the interaction of a single molecule with a pore. There has been a lack of experimental methods that can obtain information on the structure and orientation of the molecules as they pass through a pore, and their interactions with the pore during passage. Researchers in Japan have now succeeded for the first time in observing a long chain of a fullerene-labeled hydrocarbon passing through a nanometer-size pore in the wall of a carbon nanotube as if it were alive.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





