Showing Spotlights 81 - 88 of 315 in category All (newest first):
 Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy. These devices combine the advantages of conventional capacitors - they can rapidly deliver high current densities on demand - and batteries - they can store a large amount of electrical energy. Supercapacitors offer a low-cost alternative source of energy to replace rechargeable batteries. Although the energy density of capacitors is quite low compared to batteries, their power density is much higher, allowing them to provide bursts of electric energy. Researchers have now fabricated novel high-performance sponge supercapacitors using a simple and scalable method. Their results shows that three-dimensional electrodes potentially have a huge advantage over conventional mixed electrode materials.
Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy. These devices combine the advantages of conventional capacitors - they can rapidly deliver high current densities on demand - and batteries - they can store a large amount of electrical energy. Supercapacitors offer a low-cost alternative source of energy to replace rechargeable batteries. Although the energy density of capacitors is quite low compared to batteries, their power density is much higher, allowing them to provide bursts of electric energy. Researchers have now fabricated novel high-performance sponge supercapacitors using a simple and scalable method. Their results shows that three-dimensional electrodes potentially have a huge advantage over conventional mixed electrode materials.
Dec 6th, 2011
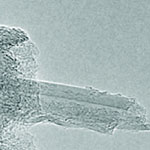 Using the CVD process, manufacturers can combine a metal catalyst such as iron with reaction gases such as hydrocarbon to form carbon nanotubes inside a high-temperature furnace. This process creates CNTs that are subsequently deposited in a collection environment and harvested into the desired end-product structural form. Understanding the processes in CVD growth of carbon nanotubes is important for their high yield and extended production. But even today, some 20 years since their discovery, the finer details of CNT growth mechanisms remain poorly understood. One of the issues that manufacturers have to grapple with is the levelling off and ultimate stoppage of CNT growth several minutes into the CVD process. This is referred to as catalyst poisoning - an inactivation or poisoning process of catalyst particles that has been attributed to an overcoat of amorphous carbon.
Using the CVD process, manufacturers can combine a metal catalyst such as iron with reaction gases such as hydrocarbon to form carbon nanotubes inside a high-temperature furnace. This process creates CNTs that are subsequently deposited in a collection environment and harvested into the desired end-product structural form. Understanding the processes in CVD growth of carbon nanotubes is important for their high yield and extended production. But even today, some 20 years since their discovery, the finer details of CNT growth mechanisms remain poorly understood. One of the issues that manufacturers have to grapple with is the levelling off and ultimate stoppage of CNT growth several minutes into the CVD process. This is referred to as catalyst poisoning - an inactivation or poisoning process of catalyst particles that has been attributed to an overcoat of amorphous carbon.
Oct 31st, 2011
 More than 100 companies around the world today are manufacturing carbon nanotubes and this number is expected to increase to more than 200 within the next five years, while there are more than 1,000 companies and institutions that are actively engaged in CNT research and development. This article looks at the market size, applications, processing technology and end-user products of carbon nanotubes. In addition, the study looks at industry leaders in the value chain, potential applications, products which are under development and are likely to enter the market in the next five to ten years. For this study, we have surveyed industry professionals/stakeholders in the CNTs value chain, extracted information from our proprietary in-house databases/ inter-linked databases as well as researched other primary and secondary sources and triangulated data and the findings are presented in this article.
More than 100 companies around the world today are manufacturing carbon nanotubes and this number is expected to increase to more than 200 within the next five years, while there are more than 1,000 companies and institutions that are actively engaged in CNT research and development. This article looks at the market size, applications, processing technology and end-user products of carbon nanotubes. In addition, the study looks at industry leaders in the value chain, potential applications, products which are under development and are likely to enter the market in the next five to ten years. For this study, we have surveyed industry professionals/stakeholders in the CNTs value chain, extracted information from our proprietary in-house databases/ inter-linked databases as well as researched other primary and secondary sources and triangulated data and the findings are presented in this article.
Oct 20th, 2011
 Vertical arrays of carbon nanotubes, called 'forests', are a common type of three-dimentional (3D) nanotube assembly that researchers work with in their labs. These forests can be produced by chemical vapor deposition technique and used for diverse applications such as in photo- or thermoacoustics, highly elastic conductive composites, for mechanical nanomanipulation, in catalysis, or as sensors in nanomedicine, just to name a few examples. These and other applications relay on connectivity of carbon nanotubes in the forest structure. New measurements show that room temperature electrical properties of this nanotube network reveal quite substantial nonlinerarities that became more pronounced at sample cooling.
Vertical arrays of carbon nanotubes, called 'forests', are a common type of three-dimentional (3D) nanotube assembly that researchers work with in their labs. These forests can be produced by chemical vapor deposition technique and used for diverse applications such as in photo- or thermoacoustics, highly elastic conductive composites, for mechanical nanomanipulation, in catalysis, or as sensors in nanomedicine, just to name a few examples. These and other applications relay on connectivity of carbon nanotubes in the forest structure. New measurements show that room temperature electrical properties of this nanotube network reveal quite substantial nonlinerarities that became more pronounced at sample cooling.
Oct 7th, 2011
 Carbon nanotubes (CNTs) have not yet met commercial expectations from a decade ago, and now hot on its heels is graphene. Graphene is considered a hot candidate for applications such as computers, displays, photovoltaics, and flexible electronics. The biggest opportunity for both materials is in printed and potentially printed electronics. In a comparably short time a large amount of graphene materials have become commercially available contributing to further advancements and application development. At a fraction of the weight and cost of CNTs, graphene may displace carbon nanotubes and even indium tin oxide in some applications. Flexible, see-through displays may be the one application that finally puts graphene into the commercial spotlight.
Carbon nanotubes (CNTs) have not yet met commercial expectations from a decade ago, and now hot on its heels is graphene. Graphene is considered a hot candidate for applications such as computers, displays, photovoltaics, and flexible electronics. The biggest opportunity for both materials is in printed and potentially printed electronics. In a comparably short time a large amount of graphene materials have become commercially available contributing to further advancements and application development. At a fraction of the weight and cost of CNTs, graphene may displace carbon nanotubes and even indium tin oxide in some applications. Flexible, see-through displays may be the one application that finally puts graphene into the commercial spotlight.
Sep 28th, 2011
 Electronic devices with muscles-like stretchability have long been pursued, but not achieved due to the requirement that all materials in the devices - electrodes, semiconductor, and dielectric - are stretchable. In their pursuit of fully flexible and stretchable electronic devices, researchers have already reported stretchable solar cells and transistors as well as stretchable active-matrix displays. The nanomaterials used for these purposes range from coiled nanowires to graphene. Recently, researchers at UCLA have successfully demonstrated a stretchable polymer composite that is highly transparent and highly conductive, and applied this nanocomposite material to fabricating stretchable devices. This work represents a proof-of-concept, highly stretchable semiconductor device wherein every part of the device is intrinsically stretchable.
Electronic devices with muscles-like stretchability have long been pursued, but not achieved due to the requirement that all materials in the devices - electrodes, semiconductor, and dielectric - are stretchable. In their pursuit of fully flexible and stretchable electronic devices, researchers have already reported stretchable solar cells and transistors as well as stretchable active-matrix displays. The nanomaterials used for these purposes range from coiled nanowires to graphene. Recently, researchers at UCLA have successfully demonstrated a stretchable polymer composite that is highly transparent and highly conductive, and applied this nanocomposite material to fabricating stretchable devices. This work represents a proof-of-concept, highly stretchable semiconductor device wherein every part of the device is intrinsically stretchable.
Aug 22nd, 2011
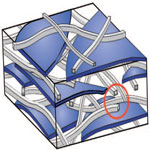 Super-tough materials with exceptional mechanical properties are in critical need for applications under extreme conditions such as jet engines, power turbines, catalytic heat exchangers, military armors, airplanes, and spacecraft. Researchers involved in improving man-made composite materials are trying to understand how some of the amazing high-performance materials found in nature can be copied or even improved upon. Nature has evolved complex bottom-up methods for fabricating ordered nanostructured materials that often have extraordinary mechanical strength and toughness. The main problem in making nanocomposite materials is how the separate components can be interfaced without losing the good properties of each component. Researchers were now able to show that biomolecules that may seem soft and fragile can actually strengthen a composite material by creating cohesion between two materials that differ much from each other.
Super-tough materials with exceptional mechanical properties are in critical need for applications under extreme conditions such as jet engines, power turbines, catalytic heat exchangers, military armors, airplanes, and spacecraft. Researchers involved in improving man-made composite materials are trying to understand how some of the amazing high-performance materials found in nature can be copied or even improved upon. Nature has evolved complex bottom-up methods for fabricating ordered nanostructured materials that often have extraordinary mechanical strength and toughness. The main problem in making nanocomposite materials is how the separate components can be interfaced without losing the good properties of each component. Researchers were now able to show that biomolecules that may seem soft and fragile can actually strengthen a composite material by creating cohesion between two materials that differ much from each other.
Aug 8th, 2011
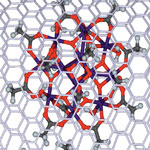 Single-molecule magnets (SMM) are fascinating nanoscale structures with unique functional properties showing promise for high-density electronic data storage devices, solid state quantum computers, spintronic devices such as spin valves, and other advanced technological applications. Despite a flurry of research in this area - since an individual magnetic molecule represents the ultimate size limit to storing and processing information - the main challenge related to harnessing properties of SMM remained unsolved. A new study by a group of European researchers reports the successful encapsulation of single-molecule magnets in carbon nanotubes, yielding a new type of hybrid nanostructure that combines all the key single-molecule magnet properties of the guest molecules with the functional properties of the host CNT.
Single-molecule magnets (SMM) are fascinating nanoscale structures with unique functional properties showing promise for high-density electronic data storage devices, solid state quantum computers, spintronic devices such as spin valves, and other advanced technological applications. Despite a flurry of research in this area - since an individual magnetic molecule represents the ultimate size limit to storing and processing information - the main challenge related to harnessing properties of SMM remained unsolved. A new study by a group of European researchers reports the successful encapsulation of single-molecule magnets in carbon nanotubes, yielding a new type of hybrid nanostructure that combines all the key single-molecule magnet properties of the guest molecules with the functional properties of the host CNT.
Jul 27th, 2011
 Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy. These devices combine the advantages of conventional capacitors - they can rapidly deliver high current densities on demand - and batteries - they can store a large amount of electrical energy. Supercapacitors offer a low-cost alternative source of energy to replace rechargeable batteries. Although the energy density of capacitors is quite low compared to batteries, their power density is much higher, allowing them to provide bursts of electric energy. Researchers have now fabricated novel high-performance sponge supercapacitors using a simple and scalable method. Their results shows that three-dimensional electrodes potentially have a huge advantage over conventional mixed electrode materials.
Ultra- or supercapacitors are emerging as a key enabling storage technology for use in fuel-efficient transport as well as in renewable energy. These devices combine the advantages of conventional capacitors - they can rapidly deliver high current densities on demand - and batteries - they can store a large amount of electrical energy. Supercapacitors offer a low-cost alternative source of energy to replace rechargeable batteries. Although the energy density of capacitors is quite low compared to batteries, their power density is much higher, allowing them to provide bursts of electric energy. Researchers have now fabricated novel high-performance sponge supercapacitors using a simple and scalable method. Their results shows that three-dimensional electrodes potentially have a huge advantage over conventional mixed electrode materials.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





