Showing Spotlights 89 - 96 of 150 in category All (newest first):
 One possible option for reducing CO2 emissions from power plants is to capture them before they hit the atmosphere and store the gas underground. This technique is called Carbon dioxide Capture and Storage. However, before CO2 can be stored, it must be separated from the other waste gases resulting from combustion or industrial processes. Most current methods used for this type of filtration are expensive and require the use of chemicals. Nanotechnology techniques to fabricate nanoscale thin membranes could lead to new membrane technology that could change that.
Current membranes are in many cases not competitive for large scale applications, because their permeance for carbon dioxide is not high enough. Researchers in Germany have now reported the development and manufacturing of nanometric thin film membranes with record performance.
One possible option for reducing CO2 emissions from power plants is to capture them before they hit the atmosphere and store the gas underground. This technique is called Carbon dioxide Capture and Storage. However, before CO2 can be stored, it must be separated from the other waste gases resulting from combustion or industrial processes. Most current methods used for this type of filtration are expensive and require the use of chemicals. Nanotechnology techniques to fabricate nanoscale thin membranes could lead to new membrane technology that could change that.
Current membranes are in many cases not competitive for large scale applications, because their permeance for carbon dioxide is not high enough. Researchers in Germany have now reported the development and manufacturing of nanometric thin film membranes with record performance.
Sep 21st, 2010
 Along with control of fire, iron smelting is one of the founding technological pillars of civilization. Industry has used the same basic process to make iron for over 3000 years. Yet, it is also one of the major global sources of greenhouse gas release. Iron, a basic commodity, is still produced by the greenhouse gas intensive reduction of iron oxide by carbon-coke and currently accounts for the release of one quarter of worldwide carbon dioxide emissions by industry. For instance, on average 1.9 tonnes of carbon dioxide are emitted for every tonne of steel produced. Due to a large share of coal in the energy mix of current production technology, the CO2 emissions are high. Through a new understanding of the chemistry of iron at high temperature, researchers have uncovered an effective new carbon-dioxide-free process to form iron.
Along with control of fire, iron smelting is one of the founding technological pillars of civilization. Industry has used the same basic process to make iron for over 3000 years. Yet, it is also one of the major global sources of greenhouse gas release. Iron, a basic commodity, is still produced by the greenhouse gas intensive reduction of iron oxide by carbon-coke and currently accounts for the release of one quarter of worldwide carbon dioxide emissions by industry. For instance, on average 1.9 tonnes of carbon dioxide are emitted for every tonne of steel produced. Due to a large share of coal in the energy mix of current production technology, the CO2 emissions are high. Through a new understanding of the chemistry of iron at high temperature, researchers have uncovered an effective new carbon-dioxide-free process to form iron.
Aug 25th, 2010
 Quite a lot of nanotechnology research and manufacturing efforts go into synthesizing metal-based nanoparticles. Unfortunately, some of the nanoparticle manufacturing processes themselves as well as the final nanoparticle materials may be of potential concern for environmental regulators and for researchers attempting to address nanomaterial toxicity. As an alternative to using these potentially hazardous metal-based nanoparticles, some researchers are suggesting the use of naturally occurring nanoparticles. However, this area has not yet been well explored with regard to natural nanoparticles' diverse properties and potential applications. Researchers have now made the discovery that naturally occurring nanoparticles have unique optical properties. In addition, they are less toxic and biodegradable than their synthesized, metal-based counterparts. This discovery makes it likely that scientists will be able to find more biocompatible nanoparticles to replace metal-based nanoparticles, predominantly for biomedical applications.
Quite a lot of nanotechnology research and manufacturing efforts go into synthesizing metal-based nanoparticles. Unfortunately, some of the nanoparticle manufacturing processes themselves as well as the final nanoparticle materials may be of potential concern for environmental regulators and for researchers attempting to address nanomaterial toxicity. As an alternative to using these potentially hazardous metal-based nanoparticles, some researchers are suggesting the use of naturally occurring nanoparticles. However, this area has not yet been well explored with regard to natural nanoparticles' diverse properties and potential applications. Researchers have now made the discovery that naturally occurring nanoparticles have unique optical properties. In addition, they are less toxic and biodegradable than their synthesized, metal-based counterparts. This discovery makes it likely that scientists will be able to find more biocompatible nanoparticles to replace metal-based nanoparticles, predominantly for biomedical applications.
Jul 21st, 2010
 The alarming rise of carbon dioxide in the atmosphere has led a numerous proposals on how to capture and store carbon dioxide in order to mitigate the damaging emissions from fossil fuels. Popular proposals, some already being tested on a large scale, involve carbon sequestration and subsequent storage in geological formations (geo-sequestration). Other ideas revolve around recycling captured carbon dioxide, for instance by converting it into hydrocarbons that can be re-used to make fuel or plastics. While these solutions would result in removing some carbon dioxide from the atmosphere, their disadvantages are that most of them are expensive, technologically challenging, or energy-intensive. Researchers have now presented the first experimental evidence of a new solar conversion process, combining electronic and chemical pathways, for carbon dioxide capture in what could become a revolutionary approach to remove and recycle CO2 from the atmosphere on a large scale. Rather than trying to sequester or hide away excess carbon dioxide, this new method allows it to be stored as solid carbon or converted in useful products.
The alarming rise of carbon dioxide in the atmosphere has led a numerous proposals on how to capture and store carbon dioxide in order to mitigate the damaging emissions from fossil fuels. Popular proposals, some already being tested on a large scale, involve carbon sequestration and subsequent storage in geological formations (geo-sequestration). Other ideas revolve around recycling captured carbon dioxide, for instance by converting it into hydrocarbons that can be re-used to make fuel or plastics. While these solutions would result in removing some carbon dioxide from the atmosphere, their disadvantages are that most of them are expensive, technologically challenging, or energy-intensive. Researchers have now presented the first experimental evidence of a new solar conversion process, combining electronic and chemical pathways, for carbon dioxide capture in what could become a revolutionary approach to remove and recycle CO2 from the atmosphere on a large scale. Rather than trying to sequester or hide away excess carbon dioxide, this new method allows it to be stored as solid carbon or converted in useful products.
Jul 16th, 2010
 Developing chemicals, molecular precursors, and industrial products from petroleum resources is a conventional practice. Plastics, detergents, even pharmaceuticals are derived from petrochemicals. With an increasing focus on the economic and environmental issues associated with the processing of petroleum-based chemicals, scientists are seeking for alternative routes to develop molecules from naturally available plant or crop-based raw materials. Particularly interesting for the fields of nanotechnology is the design and development of soft nanomaterials from renewable sources. Generating these materials from renewable resources could have a significant impact on production technologies and economies.
Developing chemicals, molecular precursors, and industrial products from petroleum resources is a conventional practice. Plastics, detergents, even pharmaceuticals are derived from petrochemicals. With an increasing focus on the economic and environmental issues associated with the processing of petroleum-based chemicals, scientists are seeking for alternative routes to develop molecules from naturally available plant or crop-based raw materials. Particularly interesting for the fields of nanotechnology is the design and development of soft nanomaterials from renewable sources. Generating these materials from renewable resources could have a significant impact on production technologies and economies.
Jun 1st, 2010
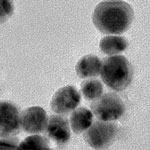 Metal nanomaterials are often synthesized using the toxic reagent formaldehyde at concentrations thousands of times higher than necessary. Many of these same nanomaterials are being investigated for use in cancer treatment - however, there is a risk that they could do more harm than good. The large excess of formaldehyde that is used originates from methods developed 100 years ago. Because these methods work well, they have stood the test of time. By better understanding the role that formaldehyde plays in nanomaterial synthesis it will become possible to reduce or eliminate this toxic reagent. By eliminating formaldehyde it will become safer to prepare these nanomaterials and safer to use them in cancer treatment.
Metal nanomaterials are often synthesized using the toxic reagent formaldehyde at concentrations thousands of times higher than necessary. Many of these same nanomaterials are being investigated for use in cancer treatment - however, there is a risk that they could do more harm than good. The large excess of formaldehyde that is used originates from methods developed 100 years ago. Because these methods work well, they have stood the test of time. By better understanding the role that formaldehyde plays in nanomaterial synthesis it will become possible to reduce or eliminate this toxic reagent. By eliminating formaldehyde it will become safer to prepare these nanomaterials and safer to use them in cancer treatment.
May 24th, 2010
 'Smart' windows, or smart glass, refers to glass technology that includes electrochromic devices, suspended particle devices, micro-blinds and liquid crystal devices. Their major feature is that they can control the amount of light passing through the glass and increase energy efficiency of the room by reducing costs for heating or air-conditioning. In the case of self-powered smart windows the glass even generates the energy needed to electrically switch its transparency. A new type of smart window proposed by researchers in The Netherlands makes use of a luminescent dye-doped liquid-crystal solution sandwiched in between electrically conductive plates as an energy-generating window.
'Smart' windows, or smart glass, refers to glass technology that includes electrochromic devices, suspended particle devices, micro-blinds and liquid crystal devices. Their major feature is that they can control the amount of light passing through the glass and increase energy efficiency of the room by reducing costs for heating or air-conditioning. In the case of self-powered smart windows the glass even generates the energy needed to electrically switch its transparency. A new type of smart window proposed by researchers in The Netherlands makes use of a luminescent dye-doped liquid-crystal solution sandwiched in between electrically conductive plates as an energy-generating window.
May 12th, 2010
 Bioethanol, unlike petroleum, is a form of renewable energy that can be produced from common agricultural feedstocks such as sugar cane or corn. Ethanol is already widely used in siome countries, mainly as biofuel additive for gasoline. The tremendous hype about bioethanol in the past few years has now been followed by a debate about how useful bioethanol actually can replace gasoline. Concerns about its production and use relate to the large amount of arable land required for crops, as well as the energy and pollution balance of the whole cycle of ethanol production. Recent developments with cellulosic ethanol production and commercialization may allay some of these concerns. It offers the promise that any plant material, from grass to wood, and not just edible crops, could be used in the production of ethanol fuels. Consequently, cellulosic ethanol could allow ethanol fuels to play a much bigger role in the future than previously thought. With regard to cellulosic production and commercialization, bioethanol production from woody biomass by enzymatic hydrolysis of cellulosic components and fermentation has attracted much attention.
Bioethanol, unlike petroleum, is a form of renewable energy that can be produced from common agricultural feedstocks such as sugar cane or corn. Ethanol is already widely used in siome countries, mainly as biofuel additive for gasoline. The tremendous hype about bioethanol in the past few years has now been followed by a debate about how useful bioethanol actually can replace gasoline. Concerns about its production and use relate to the large amount of arable land required for crops, as well as the energy and pollution balance of the whole cycle of ethanol production. Recent developments with cellulosic ethanol production and commercialization may allay some of these concerns. It offers the promise that any plant material, from grass to wood, and not just edible crops, could be used in the production of ethanol fuels. Consequently, cellulosic ethanol could allow ethanol fuels to play a much bigger role in the future than previously thought. With regard to cellulosic production and commercialization, bioethanol production from woody biomass by enzymatic hydrolysis of cellulosic components and fermentation has attracted much attention.
Mar 25th, 2010
 One possible option for reducing CO2 emissions from power plants is to capture them before they hit the atmosphere and store the gas underground. This technique is called Carbon dioxide Capture and Storage. However, before CO2 can be stored, it must be separated from the other waste gases resulting from combustion or industrial processes. Most current methods used for this type of filtration are expensive and require the use of chemicals. Nanotechnology techniques to fabricate nanoscale thin membranes could lead to new membrane technology that could change that.
Current membranes are in many cases not competitive for large scale applications, because their permeance for carbon dioxide is not high enough. Researchers in Germany have now reported the development and manufacturing of nanometric thin film membranes with record performance.
One possible option for reducing CO2 emissions from power plants is to capture them before they hit the atmosphere and store the gas underground. This technique is called Carbon dioxide Capture and Storage. However, before CO2 can be stored, it must be separated from the other waste gases resulting from combustion or industrial processes. Most current methods used for this type of filtration are expensive and require the use of chemicals. Nanotechnology techniques to fabricate nanoscale thin membranes could lead to new membrane technology that could change that.
Current membranes are in many cases not competitive for large scale applications, because their permeance for carbon dioxide is not high enough. Researchers in Germany have now reported the development and manufacturing of nanometric thin film membranes with record performance.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





