Showing Spotlights 97 - 104 of 315 in category All (newest first):
 Many strategies to develop stretchable electronics rely on engineering new constructs from existing materials, e.g. ultrathin, stretchable silicon structures. Another approach is to fabricate ultrathin CMOS circuits on stretchable materials such as polymers. Nanotechnology allows a novel route to materials and structures that can be used to develop human-friendly devices with realistic functions and abilities that would not be feasible by mere extension of conventional technology. New research suggests devices that can act as part of human skin or clothing, and can therefore be used ubiquitously. Such devices could eventually find a wide range of applications in recreation, virtual reality, robotics and health care.
Many strategies to develop stretchable electronics rely on engineering new constructs from existing materials, e.g. ultrathin, stretchable silicon structures. Another approach is to fabricate ultrathin CMOS circuits on stretchable materials such as polymers. Nanotechnology allows a novel route to materials and structures that can be used to develop human-friendly devices with realistic functions and abilities that would not be feasible by mere extension of conventional technology. New research suggests devices that can act as part of human skin or clothing, and can therefore be used ubiquitously. Such devices could eventually find a wide range of applications in recreation, virtual reality, robotics and health care.
Apr 6th, 2011
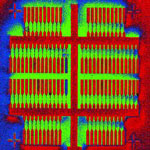 The commercial realization of carbon nanotube-based electronic devices and their integration into the existing silicon-based processor technologies is still hampered by the inability to scale up the - currently lab-based - fabrication processes to manufacture a large number of devices on a single chip. The fundamental issue of carbon nanotube device fabrication still remains the biggest challenge for effective commercialization of nanotube electronics. In a further step towards large-scale integration of single-walled carbon nanotubes (SWCNTs) into complex functional electronic circuits, researchers have now combined a previously developed dielectrophoretic deposition approach with SWCNT sorting using density gradient ultracentrifugation, ensuring high purity of SWCNTs in suspension and consequently in the assembled devices.
The commercial realization of carbon nanotube-based electronic devices and their integration into the existing silicon-based processor technologies is still hampered by the inability to scale up the - currently lab-based - fabrication processes to manufacture a large number of devices on a single chip. The fundamental issue of carbon nanotube device fabrication still remains the biggest challenge for effective commercialization of nanotube electronics. In a further step towards large-scale integration of single-walled carbon nanotubes (SWCNTs) into complex functional electronic circuits, researchers have now combined a previously developed dielectrophoretic deposition approach with SWCNT sorting using density gradient ultracentrifugation, ensuring high purity of SWCNTs in suspension and consequently in the assembled devices.
Mar 22nd, 2011
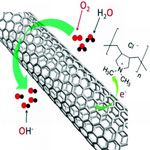 The efficiency of catalyzing the oxygen reduction reaction (ORR) - the process that breaks the bonds of oxygen molecules - to a large degree determines the electrochemical performance of fuel cells. Platinum and platinum-based composites have long been considered as the most efficient ORR catalysts. In their search for practically viable non-precious metal ORR catalysts, researchers have also been investigating vertically-aligned nitrogen-containing carbon nanotubes. Having a strong electron-withdrawing ability, poly(diallyldimethylammonium chloride) (PDDA) was used to create net positive charge for carbon atoms in the nanotube carbon plane via intermolecular charge transfer. The resultant PDDA functionalized/adsorbed carbon nanotubes were demonstrated to act as metal-free catalysts for oxygen reduction reaction in fuel cells with similar performance as platinum catalysts.
The efficiency of catalyzing the oxygen reduction reaction (ORR) - the process that breaks the bonds of oxygen molecules - to a large degree determines the electrochemical performance of fuel cells. Platinum and platinum-based composites have long been considered as the most efficient ORR catalysts. In their search for practically viable non-precious metal ORR catalysts, researchers have also been investigating vertically-aligned nitrogen-containing carbon nanotubes. Having a strong electron-withdrawing ability, poly(diallyldimethylammonium chloride) (PDDA) was used to create net positive charge for carbon atoms in the nanotube carbon plane via intermolecular charge transfer. The resultant PDDA functionalized/adsorbed carbon nanotubes were demonstrated to act as metal-free catalysts for oxygen reduction reaction in fuel cells with similar performance as platinum catalysts.
Mar 21st, 2011
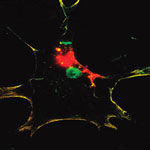 Owing to their large surface area, strong infrared photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms. However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies. Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries. In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages.
Owing to their large surface area, strong infrared photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms. However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies. Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries. In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages.
Mar 10th, 2011
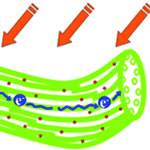 There is an increasing interest in flexible solar cells and researchers have been investigating weavable fiber solar cells based on metal wires, glass fibers, or polymer fibers. Unfortunately, the low efficiencies of these fiber-based solar cells greatly limit their promising applications. In order to improve these efficiencies, scientists are exploring various nanomaterials to improve charge separation and transport in these fiber-based photovoltaic devices. One recent promising result has been demonstrated by a research team in China who have developed a novel solar cell from flexible, light-weight, ultrastrong, and semiconductive carbon nanotube fiber. The high alignment of building nanotubes in the fiber allows charges to separate and transport along the fibers efficiently, which provides a fiber solar cell with high performance.
There is an increasing interest in flexible solar cells and researchers have been investigating weavable fiber solar cells based on metal wires, glass fibers, or polymer fibers. Unfortunately, the low efficiencies of these fiber-based solar cells greatly limit their promising applications. In order to improve these efficiencies, scientists are exploring various nanomaterials to improve charge separation and transport in these fiber-based photovoltaic devices. One recent promising result has been demonstrated by a research team in China who have developed a novel solar cell from flexible, light-weight, ultrastrong, and semiconductive carbon nanotube fiber. The high alignment of building nanotubes in the fiber allows charges to separate and transport along the fibers efficiently, which provides a fiber solar cell with high performance.
Feb 1st, 2011
 Imagine intelligent medical implants that can continuously monitor their condition inside the body and autonomously respond to changes such as infection by releasing anti-inflammatory agents. Thanks to nanotechnology, medical research is moving quickly towards this goal. A new study shows that the use of polypyrrole films as electrically controlled drug release devices on implant surfaces can potentially improve bone implants. By electrodepositing antibiotics or anti-inflammatory drugs in a polymer coating on medical devices, researchers demonstrate that such drugs can be released from polypyrrole on demand - by applying a voltage - and control cellular behavior important for orthopedic applications, i.e. inhibit inflammation and kill bacteria.
Imagine intelligent medical implants that can continuously monitor their condition inside the body and autonomously respond to changes such as infection by releasing anti-inflammatory agents. Thanks to nanotechnology, medical research is moving quickly towards this goal. A new study shows that the use of polypyrrole films as electrically controlled drug release devices on implant surfaces can potentially improve bone implants. By electrodepositing antibiotics or anti-inflammatory drugs in a polymer coating on medical devices, researchers demonstrate that such drugs can be released from polypyrrole on demand - by applying a voltage - and control cellular behavior important for orthopedic applications, i.e. inhibit inflammation and kill bacteria.
Jan 25th, 2011
 Ranging from electronic gadgets to medical applications, many nanomaterial-based devices have appeared in the market. One of the most important issues for these devices is their reliability and life-time of operation. A vital factor behind these issues is the structural stability of the nano-device - debonding of the nanomaterial from the substrate material being the single largest contribution for device degradation. In order to improve bonding between nanomaterials and their substrate, it is essential to understand and quantify the bonding mechanisms. A new nano-scratch technique developed by researchers in the U.S. could serve as the basis for a reliable quantification technique for interpreting nanomaterial-substrate bond strength.
Ranging from electronic gadgets to medical applications, many nanomaterial-based devices have appeared in the market. One of the most important issues for these devices is their reliability and life-time of operation. A vital factor behind these issues is the structural stability of the nano-device - debonding of the nanomaterial from the substrate material being the single largest contribution for device degradation. In order to improve bonding between nanomaterials and their substrate, it is essential to understand and quantify the bonding mechanisms. A new nano-scratch technique developed by researchers in the U.S. could serve as the basis for a reliable quantification technique for interpreting nanomaterial-substrate bond strength.
Jan 14th, 2011
 The toxicity issues surrounding carbon nanotubes (CNTs) are highly relevant for two reasons: Firstly, as more and more products containing CNTs come to market, there is a chance that free CNTs get released during their life cycles, most likely during production or disposal, and find their way through the environment into the body. Secondly, and much more pertinent with regard to potential health risks, is the use of CNTs in biological and medical settings. Some groups are using CNTs in research for vaccination as well as gene and cancer therapy. Here, the CNT applications are designed to interact directly with the immune system. Understanding the interplay between CNTs and immune proteins is therefore critical for both improving CNT applications in biology and medicine and avoiding potentially noxious immune responses.
The toxicity issues surrounding carbon nanotubes (CNTs) are highly relevant for two reasons: Firstly, as more and more products containing CNTs come to market, there is a chance that free CNTs get released during their life cycles, most likely during production or disposal, and find their way through the environment into the body. Secondly, and much more pertinent with regard to potential health risks, is the use of CNTs in biological and medical settings. Some groups are using CNTs in research for vaccination as well as gene and cancer therapy. Here, the CNT applications are designed to interact directly with the immune system. Understanding the interplay between CNTs and immune proteins is therefore critical for both improving CNT applications in biology and medicine and avoiding potentially noxious immune responses.
Jan 13th, 2011
 Many strategies to develop stretchable electronics rely on engineering new constructs from existing materials, e.g. ultrathin, stretchable silicon structures. Another approach is to fabricate ultrathin CMOS circuits on stretchable materials such as polymers. Nanotechnology allows a novel route to materials and structures that can be used to develop human-friendly devices with realistic functions and abilities that would not be feasible by mere extension of conventional technology. New research suggests devices that can act as part of human skin or clothing, and can therefore be used ubiquitously. Such devices could eventually find a wide range of applications in recreation, virtual reality, robotics and health care.
Many strategies to develop stretchable electronics rely on engineering new constructs from existing materials, e.g. ultrathin, stretchable silicon structures. Another approach is to fabricate ultrathin CMOS circuits on stretchable materials such as polymers. Nanotechnology allows a novel route to materials and structures that can be used to develop human-friendly devices with realistic functions and abilities that would not be feasible by mere extension of conventional technology. New research suggests devices that can act as part of human skin or clothing, and can therefore be used ubiquitously. Such devices could eventually find a wide range of applications in recreation, virtual reality, robotics and health care.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





