Showing Spotlights 97 - 104 of 137 in category All (newest first):
 The World Economic Forum, whose 2008 Annual Meeting ended on Sunday, has founded the Global Risk Network in 2004 in response to concern that the international community and the global business community were not yet able to respond adequately to a changing global risk landscape. The Program has moved forward in partnership with Citigroup, Marsh & McLennan Companies, Merrill Lynch, Swiss Re and the Center for Risk Management and Decision Processes, and Wharton School. In an increasingly complex and interconnected global environment, risks can no longer be contained within geographical or system boundaries. No one company, industry or state can successfully understand and mitigate global risks. The World Economic Forum, with numerous links to business networks, policy-makers and government, NGOs and think-tanks, is in a unique position to advance new thinking on global risks, to generate risk mitigation measures and to integrate current knowledge on global risks. Over the past few years, the Global Risk Network team has released an annual report. This years' report 'Global Risks 2008' was published two weeks ago. In it, as in previous years, nanotechnology was characterized as a global core risk.
The World Economic Forum, whose 2008 Annual Meeting ended on Sunday, has founded the Global Risk Network in 2004 in response to concern that the international community and the global business community were not yet able to respond adequately to a changing global risk landscape. The Program has moved forward in partnership with Citigroup, Marsh & McLennan Companies, Merrill Lynch, Swiss Re and the Center for Risk Management and Decision Processes, and Wharton School. In an increasingly complex and interconnected global environment, risks can no longer be contained within geographical or system boundaries. No one company, industry or state can successfully understand and mitigate global risks. The World Economic Forum, with numerous links to business networks, policy-makers and government, NGOs and think-tanks, is in a unique position to advance new thinking on global risks, to generate risk mitigation measures and to integrate current knowledge on global risks. Over the past few years, the Global Risk Network team has released an annual report. This years' report 'Global Risks 2008' was published two weeks ago. In it, as in previous years, nanotechnology was characterized as a global core risk.
Jan 28th, 2008
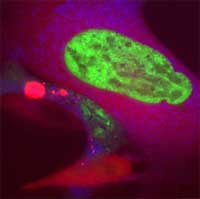 Toxicology is an interdisciplinary research field concerned with the study of the adverse effects of chemicals on living organisms. It applies knowledge, methods and techniques from such fields as chemistry, physics, material sciences, pharmacy, medicine and molecular biology. Toxicology established itself in the last 25-30 years as a testing science in the course of efforts of industrial nations to regulate toxic chemicals. Particle toxicology, as a subdiscipline, developed in the context of lung disease arising from inhalation exposure to dust particles of workers in the mining industry. It later expanded to the area of air pollution. With the rapid development of nanotechnology applications and materials, nanotoxicology is emerging as an important subdiscipline of nanotechnology as well as toxicology. Most, if not all, toxicological studies on nanoparticles rely on current methods, practices and terminology as gained and applied in the analysis of micro- and ultrafine particles and mineral fibers. Together with recent studies on nanoparticles, this has provided an initial basis for evaluating the primary issues in a risk assessment framework for nanomaterials. However, current toxicological knowledge about engineered nanoparticles is extremely limited and traditional toxicology does not allow for a complete understanding of the size, shape, composition and aggregation-dependent interactions of nanostructures with biological systems. An understanding of the relationship between the physical and chemical properties of nanostructures and their in vivo behavior would provide a basis for assessing toxic response and more importantly could lead to predictive models for assessing toxicity.
Toxicology is an interdisciplinary research field concerned with the study of the adverse effects of chemicals on living organisms. It applies knowledge, methods and techniques from such fields as chemistry, physics, material sciences, pharmacy, medicine and molecular biology. Toxicology established itself in the last 25-30 years as a testing science in the course of efforts of industrial nations to regulate toxic chemicals. Particle toxicology, as a subdiscipline, developed in the context of lung disease arising from inhalation exposure to dust particles of workers in the mining industry. It later expanded to the area of air pollution. With the rapid development of nanotechnology applications and materials, nanotoxicology is emerging as an important subdiscipline of nanotechnology as well as toxicology. Most, if not all, toxicological studies on nanoparticles rely on current methods, practices and terminology as gained and applied in the analysis of micro- and ultrafine particles and mineral fibers. Together with recent studies on nanoparticles, this has provided an initial basis for evaluating the primary issues in a risk assessment framework for nanomaterials. However, current toxicological knowledge about engineered nanoparticles is extremely limited and traditional toxicology does not allow for a complete understanding of the size, shape, composition and aggregation-dependent interactions of nanostructures with biological systems. An understanding of the relationship between the physical and chemical properties of nanostructures and their in vivo behavior would provide a basis for assessing toxic response and more importantly could lead to predictive models for assessing toxicity.
Jan 18th, 2008
 The UK government has published its second research report on nanotechnology risks, outlining progress on its research agenda to address the potential risk posed by the products of nanotechnology. The report places the UK research program in an international context. The Nanotechnology Research Coordination Group (NRCG) is collaborating with international partners, particularly through the Organization for Economic Co-operation and Development (OECD) and the International Standards Organization (ISO), to share data and experiences. In this way they hope to be able to maximize the effectiveness and speed with which potential risks may be identified and managed. The report also responds to the recommendations made by the Council for Science and Technology (CST) review (March 2007) on the UK research program and the activities of the NRCG.
The UK government has published its second research report on nanotechnology risks, outlining progress on its research agenda to address the potential risk posed by the products of nanotechnology. The report places the UK research program in an international context. The Nanotechnology Research Coordination Group (NRCG) is collaborating with international partners, particularly through the Organization for Economic Co-operation and Development (OECD) and the International Standards Organization (ISO), to share data and experiences. In this way they hope to be able to maximize the effectiveness and speed with which potential risks may be identified and managed. The report also responds to the recommendations made by the Council for Science and Technology (CST) review (March 2007) on the UK research program and the activities of the NRCG.
Jan 9th, 2008
 One of the more interesting concerns of nanotechnology is 'grey goo.' The term was invented by Eric Drexler to describe one of the dangerous issues that must be faced as nanotechnology capabilities evolve. Here's how it works. 1. Pretend that nanotechnology truly exists to the point where we can fabricate machines of arbitrary complexity using individual atoms or molecules. 2. Pretend that these machines have sufficient complexity and computational means that they can make copies of themselves using whatever happens to be lying within their reach. 3. Pretend that their fabrication systems are such that they can make a copy of themselves about once an hour. 4. Pretend that one of these machines decides to do nothing except make copies of itself.
One of the more interesting concerns of nanotechnology is 'grey goo.' The term was invented by Eric Drexler to describe one of the dangerous issues that must be faced as nanotechnology capabilities evolve. Here's how it works. 1. Pretend that nanotechnology truly exists to the point where we can fabricate machines of arbitrary complexity using individual atoms or molecules. 2. Pretend that these machines have sufficient complexity and computational means that they can make copies of themselves using whatever happens to be lying within their reach. 3. Pretend that their fabrication systems are such that they can make a copy of themselves about once an hour. 4. Pretend that one of these machines decides to do nothing except make copies of itself.
Dec 21st, 2007
 As an emerging science, nanotoxicology is expanding the boundaries of traditional toxicology from a testing and auxiliary science to a new discipline where toxicological knowledge of nanomaterials can be put to constructive use in therapeutics as well as the development of new and better biocompatible materials. Until now, though, no one has been able to pin-point which properties determines or influences the inherent hazards of nanoparticles. Now, scientists have developed a framework that can be applied to a suggested hazard identification approach and is aimed at identifying causality between inherent physical and chemical properties and observed adverse effects reported in the literature.
As an emerging science, nanotoxicology is expanding the boundaries of traditional toxicology from a testing and auxiliary science to a new discipline where toxicological knowledge of nanomaterials can be put to constructive use in therapeutics as well as the development of new and better biocompatible materials. Until now, though, no one has been able to pin-point which properties determines or influences the inherent hazards of nanoparticles. Now, scientists have developed a framework that can be applied to a suggested hazard identification approach and is aimed at identifying causality between inherent physical and chemical properties and observed adverse effects reported in the literature.
Dec 14th, 2007
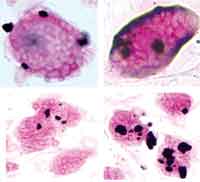 Carbon nanotubes (CNTs) have shown promise as an important new class of multifunctional building blocks and innovative tools in a large variety of nanotechnology applications, ranging from nanocomposite materials through nanoelectronics to biomedical devices (e.g. gene and drug carriers). The recent rapid development in nanotechnology has renewed the pressing demand for large-scale production of CNTs for potential applications in commercial products. The number of industrial-scale facilities for the increasingly low-cost production of multi-walled carbon nanotubes (MWCNTs) continues to grow, and with that the professional and public exposure to MWCNTs is expected to increase significantly in the coming years. Owing to their unusual one-dimensional hollow nanostructure and unique physicochemical properties, CNTs are particularly useful as novel drug delivery tools and imaging agents. However, such biomedical, and many other related, applications will not be realized if there is no proper assessment of the potential hazards of CNTs to humans and other biological systems. This situation prompted a group of researchers to carry out the first genotoxicity study of nanomaterials. Although the health effects of nanomaterials have attracted considerable attention, the scientific community has thus far focused primarily on the studies of nanomaterials toxicity at the cellular level. Very little is known about the toxicity at the molecular level, or genotoxicity, of nanomaterials in mammalian cells. Researchers at the University of Dayton have assessed the DNA damage response to MWCNTs in mouse embryonic stem cells (ES). This new work emphasizes the importance of careful scrutiny of the genotoxicity of nanomaterials.
Carbon nanotubes (CNTs) have shown promise as an important new class of multifunctional building blocks and innovative tools in a large variety of nanotechnology applications, ranging from nanocomposite materials through nanoelectronics to biomedical devices (e.g. gene and drug carriers). The recent rapid development in nanotechnology has renewed the pressing demand for large-scale production of CNTs for potential applications in commercial products. The number of industrial-scale facilities for the increasingly low-cost production of multi-walled carbon nanotubes (MWCNTs) continues to grow, and with that the professional and public exposure to MWCNTs is expected to increase significantly in the coming years. Owing to their unusual one-dimensional hollow nanostructure and unique physicochemical properties, CNTs are particularly useful as novel drug delivery tools and imaging agents. However, such biomedical, and many other related, applications will not be realized if there is no proper assessment of the potential hazards of CNTs to humans and other biological systems. This situation prompted a group of researchers to carry out the first genotoxicity study of nanomaterials. Although the health effects of nanomaterials have attracted considerable attention, the scientific community has thus far focused primarily on the studies of nanomaterials toxicity at the cellular level. Very little is known about the toxicity at the molecular level, or genotoxicity, of nanomaterials in mammalian cells. Researchers at the University of Dayton have assessed the DNA damage response to MWCNTs in mouse embryonic stem cells (ES). This new work emphasizes the importance of careful scrutiny of the genotoxicity of nanomaterials.
Dec 11th, 2007
 Governments always struggle when faced with regulating highly complex subject matters such as nanotechnologies. Primarily concerned with managing the potential risks to the environment, human health and the safety of workers (EHS), regulators often feel overwhelmed by the complexity and novelty of new technologies, stymied by a lack of data, and confused by conflicting research findings and advice from various interest groups. In the meantime, against a backdrop of a legal environment that ranges from gaping holes to regulatory vacuum, research organizations and early-adopting industry players push ahead with the new technology. Not being able to create any breathing room for lengthy political and legal considerations, the last 15-20 years have seen several governments adopting voluntary environmental programs (VEPs), arguing that this is the only viable proportional option for the time being. It is estimated that there are some 300 VEPs in the European Union and over 200 in the United States, dealing with matters such as climate change, energy, waste, water, toxic materials, agriculture, manufacturing, mining, forestry, hotels, hospitals, and financial institutions. If these voluntary programs work is subject to debate - some apparently do, some less so. In the case of manufactured nanomaterials, the risk properties remain largely unknown and it is unclear what exactly should be regulated. For the VEPs that are in place for nanomaterials, governments are urging companies to submit health and safety information on the nanomaterials they produce or commercialize. In order to investigate whether voluntary government programs will be sufficient to ensure the safety of manufactured nanomaterials, researchers have analyzed a sampling of voluntary programs in the fields of environmental health and safety in the United States over the past 20 years, with a view towards their applicability in the case of manufactured nanomaterials
Governments always struggle when faced with regulating highly complex subject matters such as nanotechnologies. Primarily concerned with managing the potential risks to the environment, human health and the safety of workers (EHS), regulators often feel overwhelmed by the complexity and novelty of new technologies, stymied by a lack of data, and confused by conflicting research findings and advice from various interest groups. In the meantime, against a backdrop of a legal environment that ranges from gaping holes to regulatory vacuum, research organizations and early-adopting industry players push ahead with the new technology. Not being able to create any breathing room for lengthy political and legal considerations, the last 15-20 years have seen several governments adopting voluntary environmental programs (VEPs), arguing that this is the only viable proportional option for the time being. It is estimated that there are some 300 VEPs in the European Union and over 200 in the United States, dealing with matters such as climate change, energy, waste, water, toxic materials, agriculture, manufacturing, mining, forestry, hotels, hospitals, and financial institutions. If these voluntary programs work is subject to debate - some apparently do, some less so. In the case of manufactured nanomaterials, the risk properties remain largely unknown and it is unclear what exactly should be regulated. For the VEPs that are in place for nanomaterials, governments are urging companies to submit health and safety information on the nanomaterials they produce or commercialize. In order to investigate whether voluntary government programs will be sufficient to ensure the safety of manufactured nanomaterials, researchers have analyzed a sampling of voluntary programs in the fields of environmental health and safety in the United States over the past 20 years, with a view towards their applicability in the case of manufactured nanomaterials
Nov 29th, 2007
 The benefits of new technologies, whether they are new medical treatments, an innovative approach to farming or new ways of generating energy, almost always come with some new risks as well. In the emerging stages of a new technology, experts and the public generally differ in their perceptions of risk. While this might be due to social and demographic factors, it is generally assumed by scientists who conduct risk research that experts' risk assessments are based more strongly on actual or perceived knowledge about a technology than lay people's risk assessments. Nevertheless, whether the risks are real or not, the public perception of an emerging technology will have a major influence on the acceptance of this technology and its commercial success. If the public perception turns negative, potentially beneficial technologies will be severely constrained as is the case for instance with gene technology. It is not surprising that a new study found that, in general, nanoscientists are more optimistic than the public about the potential benefits of nanotechnology. What is surprising though, is that, for some issues related to the environmental and long-term health impacts of nanotechnology, nanoscientists seem to be significantly more concerned than the public.
The benefits of new technologies, whether they are new medical treatments, an innovative approach to farming or new ways of generating energy, almost always come with some new risks as well. In the emerging stages of a new technology, experts and the public generally differ in their perceptions of risk. While this might be due to social and demographic factors, it is generally assumed by scientists who conduct risk research that experts' risk assessments are based more strongly on actual or perceived knowledge about a technology than lay people's risk assessments. Nevertheless, whether the risks are real or not, the public perception of an emerging technology will have a major influence on the acceptance of this technology and its commercial success. If the public perception turns negative, potentially beneficial technologies will be severely constrained as is the case for instance with gene technology. It is not surprising that a new study found that, in general, nanoscientists are more optimistic than the public about the potential benefits of nanotechnology. What is surprising though, is that, for some issues related to the environmental and long-term health impacts of nanotechnology, nanoscientists seem to be significantly more concerned than the public.
Nov 26th, 2007
 The World Economic Forum, whose 2008 Annual Meeting ended on Sunday, has founded the Global Risk Network in 2004 in response to concern that the international community and the global business community were not yet able to respond adequately to a changing global risk landscape. The Program has moved forward in partnership with Citigroup, Marsh & McLennan Companies, Merrill Lynch, Swiss Re and the Center for Risk Management and Decision Processes, and Wharton School. In an increasingly complex and interconnected global environment, risks can no longer be contained within geographical or system boundaries. No one company, industry or state can successfully understand and mitigate global risks. The World Economic Forum, with numerous links to business networks, policy-makers and government, NGOs and think-tanks, is in a unique position to advance new thinking on global risks, to generate risk mitigation measures and to integrate current knowledge on global risks. Over the past few years, the Global Risk Network team has released an annual report. This years' report 'Global Risks 2008' was published two weeks ago. In it, as in previous years, nanotechnology was characterized as a global core risk.
The World Economic Forum, whose 2008 Annual Meeting ended on Sunday, has founded the Global Risk Network in 2004 in response to concern that the international community and the global business community were not yet able to respond adequately to a changing global risk landscape. The Program has moved forward in partnership with Citigroup, Marsh & McLennan Companies, Merrill Lynch, Swiss Re and the Center for Risk Management and Decision Processes, and Wharton School. In an increasingly complex and interconnected global environment, risks can no longer be contained within geographical or system boundaries. No one company, industry or state can successfully understand and mitigate global risks. The World Economic Forum, with numerous links to business networks, policy-makers and government, NGOs and think-tanks, is in a unique position to advance new thinking on global risks, to generate risk mitigation measures and to integrate current knowledge on global risks. Over the past few years, the Global Risk Network team has released an annual report. This years' report 'Global Risks 2008' was published two weeks ago. In it, as in previous years, nanotechnology was characterized as a global core risk. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





