Showing Spotlights 105 - 112 of 624 in category All (newest first):
 Being able to replicate the extraordinary mechanical properties of nacre and other biological structures could lead to synthetic materials that possess the exceptional mechanical properties required to perform under extreme conditions. In this context, ultrahigh molecular weight polyethylene is a high-performance specialty polymer with a unique set of properties and applications. It shows remarkable strength-to-weight ratio and an excellent energy absorption ability, making it a promising material for protective applications such as body armor, biomedical implants or heat-resistant coatings.
Being able to replicate the extraordinary mechanical properties of nacre and other biological structures could lead to synthetic materials that possess the exceptional mechanical properties required to perform under extreme conditions. In this context, ultrahigh molecular weight polyethylene is a high-performance specialty polymer with a unique set of properties and applications. It shows remarkable strength-to-weight ratio and an excellent energy absorption ability, making it a promising material for protective applications such as body armor, biomedical implants or heat-resistant coatings.
Oct 22nd, 2020
 Researchers have recently demonstrated that high-intensity focused ultrasound (HIFU) is a promising, non-invasive stimulus with multiple superior and unique capabilities to induce localized heating and achieve temporal and spatial thermal effects in polymers. The team proposes a new manner of stimulating stimuli-responsive polymers. These polymers demonstrate promise for controlled drug delivery, sensing and biosensing, smart coatings, soft robotics, and flexible electronics among others.
Researchers have recently demonstrated that high-intensity focused ultrasound (HIFU) is a promising, non-invasive stimulus with multiple superior and unique capabilities to induce localized heating and achieve temporal and spatial thermal effects in polymers. The team proposes a new manner of stimulating stimuli-responsive polymers. These polymers demonstrate promise for controlled drug delivery, sensing and biosensing, smart coatings, soft robotics, and flexible electronics among others.
Oct 20th, 2020
 Researchers have successfully produced durable antiviral surfaces that inactivate SARS-CoV-2 within 6 hours. In contrast, on various non-nanostructured surfaces or smooth surfaces, the SARS-COV-2 virus remained viable for up to 48 hours. These results provide evidence that surfaces that are structured with specific nanoscale surface features are effective in preventing SARS-CoV-2 and the subsequent environmental spread. Such nanostructured surfaces can be used in hospital environments and can be extended to other industrial sectors and public infrastructure such as transportation, where fomites or contaminated surfaces are carriers for viral infections.
Researchers have successfully produced durable antiviral surfaces that inactivate SARS-CoV-2 within 6 hours. In contrast, on various non-nanostructured surfaces or smooth surfaces, the SARS-COV-2 virus remained viable for up to 48 hours. These results provide evidence that surfaces that are structured with specific nanoscale surface features are effective in preventing SARS-CoV-2 and the subsequent environmental spread. Such nanostructured surfaces can be used in hospital environments and can be extended to other industrial sectors and public infrastructure such as transportation, where fomites or contaminated surfaces are carriers for viral infections.
Sep 18th, 2020
 Sintering is a critical processing technology in the production of ceramic materials that uses high heat to compact ceramic powders into a solid form. Sintering of pure oxide ceramics requires relatively long processing times (about 20 hours) and high temperatures of 800 degrees Celsius or more. Researchers have now developed an ultrafast high-temperature sintering technique to fabricate solid state electrolytes for solid state batteries with dense structure and excellent electrochemical performance.
Sintering is a critical processing technology in the production of ceramic materials that uses high heat to compact ceramic powders into a solid form. Sintering of pure oxide ceramics requires relatively long processing times (about 20 hours) and high temperatures of 800 degrees Celsius or more. Researchers have now developed an ultrafast high-temperature sintering technique to fabricate solid state electrolytes for solid state batteries with dense structure and excellent electrochemical performance.
May 4th, 2020
 Inspired by living organisms, researchers have developed a somatosensory light-driven robot (SLiR) that can simultaneously sense strain and temperature. The SLiR subsumes pyro/piezoelectric responses and piezoresistive strain sensation under a photoactuator transducer, enabling simultaneous yet non-interfering perception of its body temperature and actuation deformation states. This design confers soft robots with complex perceptions of their body status, as well as the surrounding environments.
Inspired by living organisms, researchers have developed a somatosensory light-driven robot (SLiR) that can simultaneously sense strain and temperature. The SLiR subsumes pyro/piezoelectric responses and piezoresistive strain sensation under a photoactuator transducer, enabling simultaneous yet non-interfering perception of its body temperature and actuation deformation states. This design confers soft robots with complex perceptions of their body status, as well as the surrounding environments.
Apr 28th, 2020
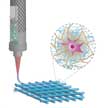 Recent developments in hydrogel chemistries, reinforcement approaches, and crosslinking methods have expanded the applications of 3D bioprinting to pharmaceutics, regenerative medicine, and biomedical devices. A multitude of 3D bioprinting techniques have been developed, but among these different approaches, extrusion-based 3D bioprinting has become a popular technique as it is easy to optimize and all its constituents are economical in price. A recent review elucidates the nuances of 3D bioprinting in a step-by-step guideline format, from its basics to more advanced levels.
Recent developments in hydrogel chemistries, reinforcement approaches, and crosslinking methods have expanded the applications of 3D bioprinting to pharmaceutics, regenerative medicine, and biomedical devices. A multitude of 3D bioprinting techniques have been developed, but among these different approaches, extrusion-based 3D bioprinting has become a popular technique as it is easy to optimize and all its constituents are economical in price. A recent review elucidates the nuances of 3D bioprinting in a step-by-step guideline format, from its basics to more advanced levels.
Apr 10th, 2020
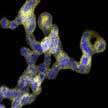 One of the key issues that is holding back widespread applications of noble metal aerogels is a lack of understanding of the underlying structure-performance correlations. Presumably, this is caused by an insufficient understanding of the sol-gel process that limits manipulating versatile parameters, such as ligament sizes, compositions, and spatial element distributions. Recently published findings not only take a big step towards the detailed understanding of reductant chemistry and gelation mechanisms, manipulating the microstructures, and enriching the compositions of NMAs, but also open a new dimension for devising high-performance electrocatalysts by taking advantage of the ligand effects.
One of the key issues that is holding back widespread applications of noble metal aerogels is a lack of understanding of the underlying structure-performance correlations. Presumably, this is caused by an insufficient understanding of the sol-gel process that limits manipulating versatile parameters, such as ligament sizes, compositions, and spatial element distributions. Recently published findings not only take a big step towards the detailed understanding of reductant chemistry and gelation mechanisms, manipulating the microstructures, and enriching the compositions of NMAs, but also open a new dimension for devising high-performance electrocatalysts by taking advantage of the ligand effects.
Mar 30th, 2020
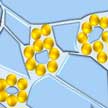 Noble metal aerogels are widely investigated for electrocatalysis applications due to their large specific surface areas and the high catalytic activity of noble metals. However, their fabrication methods are cumbersome. Now though, researchers have developed a freeze-thaw method capable of preparing various hierarchically structured noble metal gels within only one day directly from dilute solutions without extra additives. The method fits various noble metals, and multi-scale structures can be obtained across nanometer and micrometer scales.
Noble metal aerogels are widely investigated for electrocatalysis applications due to their large specific surface areas and the high catalytic activity of noble metals. However, their fabrication methods are cumbersome. Now though, researchers have developed a freeze-thaw method capable of preparing various hierarchically structured noble metal gels within only one day directly from dilute solutions without extra additives. The method fits various noble metals, and multi-scale structures can be obtained across nanometer and micrometer scales.
Mar 26th, 2020
 Being able to replicate the extraordinary mechanical properties of nacre and other biological structures could lead to synthetic materials that possess the exceptional mechanical properties required to perform under extreme conditions. In this context, ultrahigh molecular weight polyethylene is a high-performance specialty polymer with a unique set of properties and applications. It shows remarkable strength-to-weight ratio and an excellent energy absorption ability, making it a promising material for protective applications such as body armor, biomedical implants or heat-resistant coatings.
Being able to replicate the extraordinary mechanical properties of nacre and other biological structures could lead to synthetic materials that possess the exceptional mechanical properties required to perform under extreme conditions. In this context, ultrahigh molecular weight polyethylene is a high-performance specialty polymer with a unique set of properties and applications. It shows remarkable strength-to-weight ratio and an excellent energy absorption ability, making it a promising material for protective applications such as body armor, biomedical implants or heat-resistant coatings.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





