Showing Spotlights 1137 - 1144 of 2784 in category All (newest first):
 A three-dimensional crumpled graphene-encapsulated nickel sulfide electrode is reported as a superior high-energy lithium storage material. Compared with an electrode without crumpled graphene encapsulation, the optimized electrode yields significant improvements, especially in the cycling stability and rate capability. This enhanced performance is attributed to the 3D framework providing high continuous electron pathway and more free space for charge and mass transfer, and the stabilizing effect of the crumpled graphene based stretchy shell.
A three-dimensional crumpled graphene-encapsulated nickel sulfide electrode is reported as a superior high-energy lithium storage material. Compared with an electrode without crumpled graphene encapsulation, the optimized electrode yields significant improvements, especially in the cycling stability and rate capability. This enhanced performance is attributed to the 3D framework providing high continuous electron pathway and more free space for charge and mass transfer, and the stabilizing effect of the crumpled graphene based stretchy shell.
Aug 6th, 2014
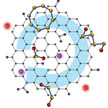 Sulfur is a very intriguing solution for the design of high energy density storage devices. The lithium-sulfur battery theoretically delivers an energy density which is 3-5 times higher than traditional lithium-ion batteries. Unfortunately, several obstacles so far have prevented the practical demonstration of sulfur-based cathodes for Li-S batteries. Among them, the most important one is the rapid capacity fading. Researchers have now developed a novel strategy towards highly stable Li-S batteries by building a strongly coupled interface between surface- mediated carbon hosts and various sulfur-containing guests.
Sulfur is a very intriguing solution for the design of high energy density storage devices. The lithium-sulfur battery theoretically delivers an energy density which is 3-5 times higher than traditional lithium-ion batteries. Unfortunately, several obstacles so far have prevented the practical demonstration of sulfur-based cathodes for Li-S batteries. Among them, the most important one is the rapid capacity fading. Researchers have now developed a novel strategy towards highly stable Li-S batteries by building a strongly coupled interface between surface- mediated carbon hosts and various sulfur-containing guests.
Aug 5th, 2014
 Researchers have now shown that, by varying the shape of magnetite nanoparticles, they can control the nature of the self-assembled structures as the nanoparticles assemble. This new work provides guidelines for the design of new self-assembled materials. Self-assembly of nanoparticles driven by competing forces can result in truly unique structures, the diversity and complexity of which could be particularly striking if the building blocks were simultaneously coupled by short- and long-range forces of different symmetries.
Researchers have now shown that, by varying the shape of magnetite nanoparticles, they can control the nature of the self-assembled structures as the nanoparticles assemble. This new work provides guidelines for the design of new self-assembled materials. Self-assembly of nanoparticles driven by competing forces can result in truly unique structures, the diversity and complexity of which could be particularly striking if the building blocks were simultaneously coupled by short- and long-range forces of different symmetries.
Aug 4th, 2014
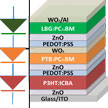 Researchers present an efficient design for a triple-junction organic tandem solar cell featuring a configuration of bandgap energies designed to maximize the tandem photocurrent output. The key innovation in this study is the demonstration of organic materials being able to mimic the record-setting efficiency of triple-junction structures in III-V solar cells. The team set out to determine a practical combination of bandgap energies for triple junctions to develop an efficient organic tandem solar cell structure.
Researchers present an efficient design for a triple-junction organic tandem solar cell featuring a configuration of bandgap energies designed to maximize the tandem photocurrent output. The key innovation in this study is the demonstration of organic materials being able to mimic the record-setting efficiency of triple-junction structures in III-V solar cells. The team set out to determine a practical combination of bandgap energies for triple junctions to develop an efficient organic tandem solar cell structure.
Jul 31st, 2014
 It seems that computer memory technology is coming full circle. Pioneers in the early 19th century, such as Charles Babbage, first proposed the use of paper memory (albeit non-electronic), where a bit was stored as the presence or absence of a hole in a paper card. State-of-the-art research today again is proposing the use of paper as memory devices. This time, although he paper may be very similar, the bits are not crudely punched holes but nanofabricated device structures. In new work, researchers demonstrated a paper-based, nonvolatile memory device. Theyused a combination of inkjet and screen printing to fabricate resistive RAM memory cells on commercial printing paper.
It seems that computer memory technology is coming full circle. Pioneers in the early 19th century, such as Charles Babbage, first proposed the use of paper memory (albeit non-electronic), where a bit was stored as the presence or absence of a hole in a paper card. State-of-the-art research today again is proposing the use of paper as memory devices. This time, although he paper may be very similar, the bits are not crudely punched holes but nanofabricated device structures. In new work, researchers demonstrated a paper-based, nonvolatile memory device. Theyused a combination of inkjet and screen printing to fabricate resistive RAM memory cells on commercial printing paper.
Jul 29th, 2014
 Ever since its discovery in 2004, graphene has been considered a relatively stable, high surface area platform to anchor nanostructured catalyst materials for various electrochemical and photocatalytic applications. The emergence of solution-based graphene in the form of graphene oxide has enabled new wet-chemistry approaches to the creation of graphene-based nanocomposites. A new study raises questions about the long-term stability of reduced graphene oxide in an aqueous environment where hydroxyl radicals can be present as part of the photocatalytic reaction cycle.
Ever since its discovery in 2004, graphene has been considered a relatively stable, high surface area platform to anchor nanostructured catalyst materials for various electrochemical and photocatalytic applications. The emergence of solution-based graphene in the form of graphene oxide has enabled new wet-chemistry approaches to the creation of graphene-based nanocomposites. A new study raises questions about the long-term stability of reduced graphene oxide in an aqueous environment where hydroxyl radicals can be present as part of the photocatalytic reaction cycle.
Jul 28th, 2014
 Transparent conductive coatings pervade modern technology. They are a critical component of optoelectronic devices such as smartphone and tablet displays as well as solar cells. The search for novel transparent electrode materials with good stability, high transparency and excellent conductivity is driven by the required trade-off between transparency and conductivity. In new work, researchers now have simultaneously increased the conductivity and transparency of ultra thin graphite by lithium intercalation.
Transparent conductive coatings pervade modern technology. They are a critical component of optoelectronic devices such as smartphone and tablet displays as well as solar cells. The search for novel transparent electrode materials with good stability, high transparency and excellent conductivity is driven by the required trade-off between transparency and conductivity. In new work, researchers now have simultaneously increased the conductivity and transparency of ultra thin graphite by lithium intercalation.
Jul 24th, 2014
 Conjugated polymer based organic photovoltaic (OPV) devices have been the subject of increasing research interest over the past years due to their potential of being light weight, mechanically flexible, semitransparent. To increase the efficiency of OPV, it is necessary to achieve a precisely controlled donor-acceptor phase separation within the short exciton diffusion length without dead ends, as well as a high hole mobility within the polymer. Now, researchers have demonstrated the effects of nanostructure geometry on the nanoimprint induced P3HT chain alignment and the performance of nanoimprinted photovoltaic devices.
Conjugated polymer based organic photovoltaic (OPV) devices have been the subject of increasing research interest over the past years due to their potential of being light weight, mechanically flexible, semitransparent. To increase the efficiency of OPV, it is necessary to achieve a precisely controlled donor-acceptor phase separation within the short exciton diffusion length without dead ends, as well as a high hole mobility within the polymer. Now, researchers have demonstrated the effects of nanostructure geometry on the nanoimprint induced P3HT chain alignment and the performance of nanoimprinted photovoltaic devices.
Jul 22nd, 2014
 A three-dimensional crumpled graphene-encapsulated nickel sulfide electrode is reported as a superior high-energy lithium storage material. Compared with an electrode without crumpled graphene encapsulation, the optimized electrode yields significant improvements, especially in the cycling stability and rate capability. This enhanced performance is attributed to the 3D framework providing high continuous electron pathway and more free space for charge and mass transfer, and the stabilizing effect of the crumpled graphene based stretchy shell.
A three-dimensional crumpled graphene-encapsulated nickel sulfide electrode is reported as a superior high-energy lithium storage material. Compared with an electrode without crumpled graphene encapsulation, the optimized electrode yields significant improvements, especially in the cycling stability and rate capability. This enhanced performance is attributed to the 3D framework providing high continuous electron pathway and more free space for charge and mass transfer, and the stabilizing effect of the crumpled graphene based stretchy shell.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





