Showing Spotlights 1153 - 1160 of 2779 in category All (newest first):
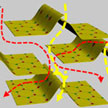 Researchers have developed a new separation membrane with 2D layered transition metal dichalcogenides (tungsten disulfide) for size-selective separation of small molecules of about 3 nm. The as-prepared WS2 membranes exhibit 5 times higher water permeance than graphene oxide membranes with similar rejection. To further improve the water permeance, they team employed ultrathin metal hydroxide nanostrands to create more fluidic channels while keeping the rejection rate of specific molecules unchanged.
Researchers have developed a new separation membrane with 2D layered transition metal dichalcogenides (tungsten disulfide) for size-selective separation of small molecules of about 3 nm. The as-prepared WS2 membranes exhibit 5 times higher water permeance than graphene oxide membranes with similar rejection. To further improve the water permeance, they team employed ultrathin metal hydroxide nanostrands to create more fluidic channels while keeping the rejection rate of specific molecules unchanged.
Jun 17th, 2014
 Researchers report for the first time the fabrication and measurement of all-inkjet-printed, all-air-processed organic solar cells. Organic photovoltaic technologies have the potential to become a thin-film alternative to inorganic silicon photovoltaics due to their intrinsic potential for low-cost print processing from solution - high-speed and at low temperature. Organic solar cells can be integrated into building facades and windows because they are optically translucent and can be manufactured on large areas at high throughput.
Researchers report for the first time the fabrication and measurement of all-inkjet-printed, all-air-processed organic solar cells. Organic photovoltaic technologies have the potential to become a thin-film alternative to inorganic silicon photovoltaics due to their intrinsic potential for low-cost print processing from solution - high-speed and at low temperature. Organic solar cells can be integrated into building facades and windows because they are optically translucent and can be manufactured on large areas at high throughput.
Jun 16th, 2014
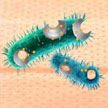 A growing body of medical nanotechnology research deals with the development of antibacterial applications, ranging from nanotechnology-based approaches for diagnosing superbugs to antimicrobial surface coatings and wound treatment with antibacterial nanomaterials. Especially silver nanomaterials have been used effectively against different bacteria, fungi and viruses but also carbon nanomaterials like nanotubes and graphene. In new work, researchers have now designed an antibacterial system combining graphene quantum dots with a low dose of a common medical reagent, hydrogen peroxide H2O2.
A growing body of medical nanotechnology research deals with the development of antibacterial applications, ranging from nanotechnology-based approaches for diagnosing superbugs to antimicrobial surface coatings and wound treatment with antibacterial nanomaterials. Especially silver nanomaterials have been used effectively against different bacteria, fungi and viruses but also carbon nanomaterials like nanotubes and graphene. In new work, researchers have now designed an antibacterial system combining graphene quantum dots with a low dose of a common medical reagent, hydrogen peroxide H2O2.
Jun 12th, 2014
 Proton-conducting materials have become important for a wide range of technologies, such as fuel cells, batteries, and biosensors. A great deal of research has been devoted to developing improved and application-specific proton conducting materials. Researchers even developed a proton-based transistor that could let machines communicate with living things. Scientists now have discovered and characterized novel electrical properties for the cephalopod structural protein reflectin.
Proton-conducting materials have become important for a wide range of technologies, such as fuel cells, batteries, and biosensors. A great deal of research has been devoted to developing improved and application-specific proton conducting materials. Researchers even developed a proton-based transistor that could let machines communicate with living things. Scientists now have discovered and characterized novel electrical properties for the cephalopod structural protein reflectin.
Jun 11th, 2014
 Renewable and high-capacity energy systems like fuel cells and metal-air batteries are key components in any scenario on future energy systems free of fossil fuels. The performance of fuel cells largely depends on the oxygen reduction reaction - the process that breaks the bonds of the oxygen molecules - which is substantially affected by the activity of the cathode catalyst. Researchers have now demonstrated the synthesis of a novel N-doped graphene/single-walled carbon nanotube hybrid material by a facile and cost-favorable one-step CVD method.
Renewable and high-capacity energy systems like fuel cells and metal-air batteries are key components in any scenario on future energy systems free of fossil fuels. The performance of fuel cells largely depends on the oxygen reduction reaction - the process that breaks the bonds of the oxygen molecules - which is substantially affected by the activity of the cathode catalyst. Researchers have now demonstrated the synthesis of a novel N-doped graphene/single-walled carbon nanotube hybrid material by a facile and cost-favorable one-step CVD method.
Jun 9th, 2014
 Almost all strategies for solar energy harvest and solar energy storage that exist today are developed as independent technologies. For instance, a solar cell generates electricity from the absorption and conversion of sunlight, while the storage of the produced electricity has to be implemented with another set of energy utilization solutions such as batteries/supercapacitors and fuel cells. With quite an ingenious solution, researchers have now demonstrated a hybrid, multifunctional material system that allows for simultaneous solar power generation (respectively hydrogen production), electrical energy storage, and chemical sensing.
Almost all strategies for solar energy harvest and solar energy storage that exist today are developed as independent technologies. For instance, a solar cell generates electricity from the absorption and conversion of sunlight, while the storage of the produced electricity has to be implemented with another set of energy utilization solutions such as batteries/supercapacitors and fuel cells. With quite an ingenious solution, researchers have now demonstrated a hybrid, multifunctional material system that allows for simultaneous solar power generation (respectively hydrogen production), electrical energy storage, and chemical sensing.
Jun 6th, 2014
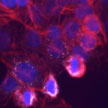 Numerous nanotoxicological studies reporting effects of nanomaterials typically address a single exposure at high dosages that are irrelevant to realistic human exposure. Recognizing that acute in vitro work had extremely low correlation to in vivo nanomaterial studies, coupled with the recognition that the unique characteristics that distinguish nanomaterials vary as a function of time, researchers sought to identify a model that would allow for the evaluation of nanomaterial behavior over a 3-month period, but be carried out in an in vitro model.
Numerous nanotoxicological studies reporting effects of nanomaterials typically address a single exposure at high dosages that are irrelevant to realistic human exposure. Recognizing that acute in vitro work had extremely low correlation to in vivo nanomaterial studies, coupled with the recognition that the unique characteristics that distinguish nanomaterials vary as a function of time, researchers sought to identify a model that would allow for the evaluation of nanomaterial behavior over a 3-month period, but be carried out in an in vitro model.
Jun 4th, 2014
 The formation of protein corona is a continuous state of flux in which many proteins compete to bind to the nanoparticle surface, each with their own characteristics such as concentration, structure and solubility determining their final affinity to the nanoparticle surface. This is the reason why biological responses to nanoparticles are strongly dependent to the type and amount of associated proteins in the composition of the protein corona. The protein corona determines the biological fate of nanoparticles and physiological responses. New research findings now show that the plasma protein alterations associated with different diseases, medical conditions, or even lifestyle, can affect the protein composition and content of the hard corona composition.
The formation of protein corona is a continuous state of flux in which many proteins compete to bind to the nanoparticle surface, each with their own characteristics such as concentration, structure and solubility determining their final affinity to the nanoparticle surface. This is the reason why biological responses to nanoparticles are strongly dependent to the type and amount of associated proteins in the composition of the protein corona. The protein corona determines the biological fate of nanoparticles and physiological responses. New research findings now show that the plasma protein alterations associated with different diseases, medical conditions, or even lifestyle, can affect the protein composition and content of the hard corona composition.
Jun 3rd, 2014
 Researchers have developed a new separation membrane with 2D layered transition metal dichalcogenides (tungsten disulfide) for size-selective separation of small molecules of about 3 nm. The as-prepared WS2 membranes exhibit 5 times higher water permeance than graphene oxide membranes with similar rejection. To further improve the water permeance, they team employed ultrathin metal hydroxide nanostrands to create more fluidic channels while keeping the rejection rate of specific molecules unchanged.
Researchers have developed a new separation membrane with 2D layered transition metal dichalcogenides (tungsten disulfide) for size-selective separation of small molecules of about 3 nm. The as-prepared WS2 membranes exhibit 5 times higher water permeance than graphene oxide membranes with similar rejection. To further improve the water permeance, they team employed ultrathin metal hydroxide nanostrands to create more fluidic channels while keeping the rejection rate of specific molecules unchanged.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





