Showing Spotlights 113 - 120 of 544 in category All (newest first):
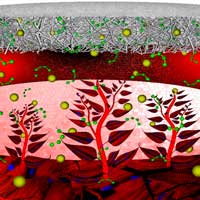 Among the various therapeutic advances, regenerative medicine and nanotechnologies have demonstrated a considerable capacity to salvage or regenerate damaged heart tissue in animal models. The superior characteristics of nanobiomaterials have shown great promise in developing engineered cardiovascular constructs for a variety of tissue engineering applications. But in order to develop efficient nanotechnology-based regenerative medicine platforms in humans, clinicians and engineers must achieve a greater common understanding of the problems, challenges, and opportunities in both fields.
Among the various therapeutic advances, regenerative medicine and nanotechnologies have demonstrated a considerable capacity to salvage or regenerate damaged heart tissue in animal models. The superior characteristics of nanobiomaterials have shown great promise in developing engineered cardiovascular constructs for a variety of tissue engineering applications. But in order to develop efficient nanotechnology-based regenerative medicine platforms in humans, clinicians and engineers must achieve a greater common understanding of the problems, challenges, and opportunities in both fields.
Sep 10th, 2019
 Despite considerable, decade-long efforts in the field of nanomedicine, fewer nanoparticle technologies than expected have made it to clinical trials. Understanding the role of the nano-bio interface - the protein corona - appears to be a critical factor for successful clinical translation of medical nanotechnologies. New research demonstrates that the protein corona controls the interaction of liposomes with immune cells and reveals that the pre-coating of liposomes with an artificial corona made of human plasma proteins significantly decreases capture by circulating leukocytes in whole blood.
Despite considerable, decade-long efforts in the field of nanomedicine, fewer nanoparticle technologies than expected have made it to clinical trials. Understanding the role of the nano-bio interface - the protein corona - appears to be a critical factor for successful clinical translation of medical nanotechnologies. New research demonstrates that the protein corona controls the interaction of liposomes with immune cells and reveals that the pre-coating of liposomes with an artificial corona made of human plasma proteins significantly decreases capture by circulating leukocytes in whole blood.
Aug 21st, 2019
 Researchers demonstrate the ease of use of jellyfish biomass as a biodegradable and biocompatible raw material for producing antibacterial mats. They report the successful electrospun-based preparation of novel composite biocompatible nanofibers based on jellyfish biomass. The antibacterial properties of the resulting nanofiber mats were enhanced by incorporating silver nanoparticles within applying an in situ bio-template synthesis that the team developed. This work suggests exploiting the jellyfish as an accessible, cheap and valuable renewable material that can be used in various applications, such as cosmetics, wound dressings, artificial skin, and bioplastics.
Researchers demonstrate the ease of use of jellyfish biomass as a biodegradable and biocompatible raw material for producing antibacterial mats. They report the successful electrospun-based preparation of novel composite biocompatible nanofibers based on jellyfish biomass. The antibacterial properties of the resulting nanofiber mats were enhanced by incorporating silver nanoparticles within applying an in situ bio-template synthesis that the team developed. This work suggests exploiting the jellyfish as an accessible, cheap and valuable renewable material that can be used in various applications, such as cosmetics, wound dressings, artificial skin, and bioplastics.
Jul 30th, 2019
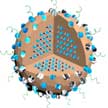 Researchers have for the first time proposed a novel tumor treatment strategy for microwave dynamic therapy to effectively treat cancers. This technique can promote the efficacy of dynamic treatment of deep tumors in clinical research and could have a broad range of applications. The team found that liquid metal supernanoparticles can generate reactive oxygen species (ROS) under microwave irradiation. That makes them suitable as microwave sensitizers for revolutionizing the ROS generation techniques.
Researchers have for the first time proposed a novel tumor treatment strategy for microwave dynamic therapy to effectively treat cancers. This technique can promote the efficacy of dynamic treatment of deep tumors in clinical research and could have a broad range of applications. The team found that liquid metal supernanoparticles can generate reactive oxygen species (ROS) under microwave irradiation. That makes them suitable as microwave sensitizers for revolutionizing the ROS generation techniques.
Jul 25th, 2019
 Turmeric has a long history of usage in traditional medicine in India and China. Ancient Indians have known the medicinal properties of turmeric - i.e. curcumin - for several millennia. Researchers have now investigated the antibacterial effects of curcumin loaded into halloysite nanotubes and found that this nanoformulation drastically increased the life expectancy of nematodes infected by pathogenic bacteria. The most interesting finding here is the selective antimicrobial action of curcumin, which suppressed one bacterial species, while demonstrating no adverse effect on another.
Turmeric has a long history of usage in traditional medicine in India and China. Ancient Indians have known the medicinal properties of turmeric - i.e. curcumin - for several millennia. Researchers have now investigated the antibacterial effects of curcumin loaded into halloysite nanotubes and found that this nanoformulation drastically increased the life expectancy of nematodes infected by pathogenic bacteria. The most interesting finding here is the selective antimicrobial action of curcumin, which suppressed one bacterial species, while demonstrating no adverse effect on another.
Jul 15th, 2019
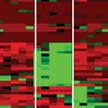 When nanoparticles enter the blood stream or other biological fluids, they are quickly surrounded by a layer of proteins. This so-called protein corona on the nanoparticles hinders interactions between the targeting ligands on the nanoparticles and their binding partners on the cells' surface. Different individuals may have a personalized protein corona owing to their distinct type, severity and period of disease, heterogeneity, individual genetic variations and environmental factors. By combining the concepts of disease-specific protein corona and sensor array technology, researchers now have created a label-free platform for the early detection and identification of diseases.
When nanoparticles enter the blood stream or other biological fluids, they are quickly surrounded by a layer of proteins. This so-called protein corona on the nanoparticles hinders interactions between the targeting ligands on the nanoparticles and their binding partners on the cells' surface. Different individuals may have a personalized protein corona owing to their distinct type, severity and period of disease, heterogeneity, individual genetic variations and environmental factors. By combining the concepts of disease-specific protein corona and sensor array technology, researchers now have created a label-free platform for the early detection and identification of diseases.
Jun 17th, 2019
 A critical challenge in gene therapy is the safe and effective delivery of genetic materials across cell membranes into target cells. Although viral systems have been the most effective method for delivering genetic matter into cells, they pose significant safety problems. Demonstrating a novel alternative delivery vector, researchers have developed a facile one-pot strategy to encapsulate plasmid DNA into nanoscale metal-organic frameworks (MOFs) and a MOF-polymer system Experiments showed that pDNA molecules could be well distributed throughout the MOF nanostructures and benefited from effective protection against the enzymatic degradation.
A critical challenge in gene therapy is the safe and effective delivery of genetic materials across cell membranes into target cells. Although viral systems have been the most effective method for delivering genetic matter into cells, they pose significant safety problems. Demonstrating a novel alternative delivery vector, researchers have developed a facile one-pot strategy to encapsulate plasmid DNA into nanoscale metal-organic frameworks (MOFs) and a MOF-polymer system Experiments showed that pDNA molecules could be well distributed throughout the MOF nanostructures and benefited from effective protection against the enzymatic degradation.
Jun 13th, 2019
 The use of TiO2 nanoparticles activated by light and ultrasound has been studied extensively for cancer treatments. For the first time, researchers have shown the nanoparticles can be effectively activated by microwaves for cancer cell destruction - potentially opening new doors for patients fighting cancer. They propose the use of microwaves and TiO2 nanoparticles to induce the production of reactive oxygen species (ROS), which has the potential to be a safer, more effective modality for cancer treatment.
The use of TiO2 nanoparticles activated by light and ultrasound has been studied extensively for cancer treatments. For the first time, researchers have shown the nanoparticles can be effectively activated by microwaves for cancer cell destruction - potentially opening new doors for patients fighting cancer. They propose the use of microwaves and TiO2 nanoparticles to induce the production of reactive oxygen species (ROS), which has the potential to be a safer, more effective modality for cancer treatment.
May 30th, 2019
 Among the various therapeutic advances, regenerative medicine and nanotechnologies have demonstrated a considerable capacity to salvage or regenerate damaged heart tissue in animal models. The superior characteristics of nanobiomaterials have shown great promise in developing engineered cardiovascular constructs for a variety of tissue engineering applications. But in order to develop efficient nanotechnology-based regenerative medicine platforms in humans, clinicians and engineers must achieve a greater common understanding of the problems, challenges, and opportunities in both fields.
Among the various therapeutic advances, regenerative medicine and nanotechnologies have demonstrated a considerable capacity to salvage or regenerate damaged heart tissue in animal models. The superior characteristics of nanobiomaterials have shown great promise in developing engineered cardiovascular constructs for a variety of tissue engineering applications. But in order to develop efficient nanotechnology-based regenerative medicine platforms in humans, clinicians and engineers must achieve a greater common understanding of the problems, challenges, and opportunities in both fields.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





