Showing Spotlights 113 - 120 of 235 in category All (newest first):
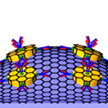 As a prime example of how the integration of multiple disparate nanotechnology fields allows the realization of novel or expanded functionalities, researchers have demonstrated a multimodal sensing device which integrates the functionalities of three traditional single mode sensors. Specifically, the team fabricated a graphene-based multimodal biosensing device, capable of transducing protein binding events into optical, electrical, and mechanical signals.
As a prime example of how the integration of multiple disparate nanotechnology fields allows the realization of novel or expanded functionalities, researchers have demonstrated a multimodal sensing device which integrates the functionalities of three traditional single mode sensors. Specifically, the team fabricated a graphene-based multimodal biosensing device, capable of transducing protein binding events into optical, electrical, and mechanical signals.
Sep 8th, 2014
 The nanotechnology-enabled detection of a change in individual cells, for instance cell surface charge, presents a new alternative and complementary method for disease detection and diagnosis. Since diseased cells, such as cancer cells, frequently carry information that distinguishes them from normal cells, accurate probing of these cells is critical for early detection of a disease. For this purpose, researchers have now designed a graphene-based optical refractive index sensor.
The nanotechnology-enabled detection of a change in individual cells, for instance cell surface charge, presents a new alternative and complementary method for disease detection and diagnosis. Since diseased cells, such as cancer cells, frequently carry information that distinguishes them from normal cells, accurate probing of these cells is critical for early detection of a disease. For this purpose, researchers have now designed a graphene-based optical refractive index sensor.
May 15th, 2014
 Graphene's piezoresistive effect, combined with its other properties such as ultra-translucency, superior mechanical flexibility and stability, high restorability, and carrier mobility, enables the use of graphene in high-sensitivity strain sensors. Potential application areas for these sensors could be found in flexible display technology, robotics, smart clothing, electronic skin, in vitro diagnostics, implantable devices, and human physiological motion detection - which has been considered as an effective approach to evaluate human health. To demonstrate this application, researchers have now reported on a method to monitor human motions.
Graphene's piezoresistive effect, combined with its other properties such as ultra-translucency, superior mechanical flexibility and stability, high restorability, and carrier mobility, enables the use of graphene in high-sensitivity strain sensors. Potential application areas for these sensors could be found in flexible display technology, robotics, smart clothing, electronic skin, in vitro diagnostics, implantable devices, and human physiological motion detection - which has been considered as an effective approach to evaluate human health. To demonstrate this application, researchers have now reported on a method to monitor human motions.
Apr 30th, 2014
 Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Nanowire field-effect sensors show significant advantages of real-time, label-free and highly sensitive detection of a wide range of analytes in liquid phase, including proteins, nucleic acids, small molecules, and viruses in single-element or multiplexed formats. Motivated by the unique features of these sensors and the ease to integrate them in the currently available VLSI technology, researchers used molecularly modified silicon nanowire FETs to detect volatile organic compounds (VOCs) that are associated with environmental pollution, quality control, explosive materials, or various diseases.
Jan 30th, 2014
 Recently, nanotechnology researchers have begun to work with graphene foams - three-dimensional structures of interconnected graphene sheets with extremely high conductivity. Since graphene foam possesses a high porosity of close to 100%, this offers the opportunity to use it as a scaffold for other nanomaterials to generate synergistic effects. Now, researchers have fabricated vertically aligned ZnO nanowire arrays on 3D graphene foam and used this electrode to detect uric acid in a reliable statistical level from the serum of Parkinson's disease patients.
Recently, nanotechnology researchers have begun to work with graphene foams - three-dimensional structures of interconnected graphene sheets with extremely high conductivity. Since graphene foam possesses a high porosity of close to 100%, this offers the opportunity to use it as a scaffold for other nanomaterials to generate synergistic effects. Now, researchers have fabricated vertically aligned ZnO nanowire arrays on 3D graphene foam and used this electrode to detect uric acid in a reliable statistical level from the serum of Parkinson's disease patients.
Jan 27th, 2014
 Previous work in stretchable, flexible electronics has shown that conventional, silicon wafer based fabrication techniques can be modified to apply electronics conformally to the heterogeneous topography of the skin. Now, researchers have demonstrated the development of a device platform that enables high precision temperature mapping of the skin in ways that have, until now, been extremely difficult in research and impossible to implement for widespread use.
Previous work in stretchable, flexible electronics has shown that conventional, silicon wafer based fabrication techniques can be modified to apply electronics conformally to the heterogeneous topography of the skin. Now, researchers have demonstrated the development of a device platform that enables high precision temperature mapping of the skin in ways that have, until now, been extremely difficult in research and impossible to implement for widespread use.
Dec 9th, 2013
 Functionalized graphene holds exceptional promise for biological and chemical sensors. In new work, researchers have shown that the distinctive 2D structure of graphene oxide, combined with its superpermeability to water molecules, leads to sensing devices with an unprecedented speed. The team reports the experimental observation of the unparalleled response speed of humidity sensors based on graphene oxide, which are - to the best of the scientists' knowledge - the fastest humidity sensors ever reported.
Functionalized graphene holds exceptional promise for biological and chemical sensors. In new work, researchers have shown that the distinctive 2D structure of graphene oxide, combined with its superpermeability to water molecules, leads to sensing devices with an unprecedented speed. The team reports the experimental observation of the unparalleled response speed of humidity sensors based on graphene oxide, which are - to the best of the scientists' knowledge - the fastest humidity sensors ever reported.
Nov 27th, 2013
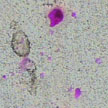 Different strategies for creation of self-healing coatings to prevent corrosion degradation have been suggested. The goal is to significantly reduce the maintenance costs in many industrial applications by applying active sensing coatings. Today we look at a novel sensing coating on the basis of nanocapsules containing pH-indicating agent. The main idea of this work is to create a novel active protective coating which is able to indicate when corrosion processes start under the coatings or in different defects.
Different strategies for creation of self-healing coatings to prevent corrosion degradation have been suggested. The goal is to significantly reduce the maintenance costs in many industrial applications by applying active sensing coatings. Today we look at a novel sensing coating on the basis of nanocapsules containing pH-indicating agent. The main idea of this work is to create a novel active protective coating which is able to indicate when corrosion processes start under the coatings or in different defects.
Sep 30th, 2013
 As a prime example of how the integration of multiple disparate nanotechnology fields allows the realization of novel or expanded functionalities, researchers have demonstrated a multimodal sensing device which integrates the functionalities of three traditional single mode sensors. Specifically, the team fabricated a graphene-based multimodal biosensing device, capable of transducing protein binding events into optical, electrical, and mechanical signals.
As a prime example of how the integration of multiple disparate nanotechnology fields allows the realization of novel or expanded functionalities, researchers have demonstrated a multimodal sensing device which integrates the functionalities of three traditional single mode sensors. Specifically, the team fabricated a graphene-based multimodal biosensing device, capable of transducing protein binding events into optical, electrical, and mechanical signals.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





