Showing Spotlights 129 - 136 of 252 in category All (newest first):
 Silicon offers a unique combination between mechanical and electrical properties making it one of the most developed materials in semiconductor industry. However, silicon is brittle and cannot be flexed, hindering its potential for high performance electronics that is flexible, stretchable or applied to irregular shapes. Researchers have now developed a pragmatic approach to achieve high performance integrated electronic systems, including thermoelectric energy harvesters, onto flexible silicon substrates.
Silicon offers a unique combination between mechanical and electrical properties making it one of the most developed materials in semiconductor industry. However, silicon is brittle and cannot be flexed, hindering its potential for high performance electronics that is flexible, stretchable or applied to irregular shapes. Researchers have now developed a pragmatic approach to achieve high performance integrated electronic systems, including thermoelectric energy harvesters, onto flexible silicon substrates.
Dec 11th, 2013
 Previous work in stretchable, flexible electronics has shown that conventional, silicon wafer based fabrication techniques can be modified to apply electronics conformally to the heterogeneous topography of the skin. Now, researchers have demonstrated the development of a device platform that enables high precision temperature mapping of the skin in ways that have, until now, been extremely difficult in research and impossible to implement for widespread use.
Previous work in stretchable, flexible electronics has shown that conventional, silicon wafer based fabrication techniques can be modified to apply electronics conformally to the heterogeneous topography of the skin. Now, researchers have demonstrated the development of a device platform that enables high precision temperature mapping of the skin in ways that have, until now, been extremely difficult in research and impossible to implement for widespread use.
Dec 9th, 2013
 Advances in materials, fabrication strategies and device designs for flexible and stretchable electronics and sensors make it possible to envision a not-too-distant future where ultra-thin, flexible circuits based on inorganic semiconductors can be wrapped and attached to any imaginable surface, including body parts and even internal organs. Robotic technologies will also benefit as it becomes possible to fabricate electronic skin ('e-skin') that, for instance, could allow surgical robots to interact, in a soft contacting mode, with their surroundings through touch.
Advances in materials, fabrication strategies and device designs for flexible and stretchable electronics and sensors make it possible to envision a not-too-distant future where ultra-thin, flexible circuits based on inorganic semiconductors can be wrapped and attached to any imaginable surface, including body parts and even internal organs. Robotic technologies will also benefit as it becomes possible to fabricate electronic skin ('e-skin') that, for instance, could allow surgical robots to interact, in a soft contacting mode, with their surroundings through touch.
Nov 15th, 2013
 Heat has become one of the most critical issues in computer and semiconductor design: The ever increasing number of transistors in computer chips requires more efficient cooling approaches for the hot spots which are generated as a result of the operation of the transistors. Researchers have now demonstrated a microfluidic technique of using thermally conductive and magnetic nanoparticles that can form low-dimensional fins in the vicinity of hot spots. This work is the first ever report on using nanoparticles for making nanofins on demand in microfluidics.
Heat has become one of the most critical issues in computer and semiconductor design: The ever increasing number of transistors in computer chips requires more efficient cooling approaches for the hot spots which are generated as a result of the operation of the transistors. Researchers have now demonstrated a microfluidic technique of using thermally conductive and magnetic nanoparticles that can form low-dimensional fins in the vicinity of hot spots. This work is the first ever report on using nanoparticles for making nanofins on demand in microfluidics.
Oct 3rd, 2013
 Over the past few years, we have seen an explosion of interest in electronic devices based on paper or textile components. These substrates are attractive because they can impart flexibility and low- cost manufacturing to devices such as transistors, circuits, light-emitting diodes, and batteries. They also can be folded. Researchers now have have shown that paper-folding concepts can be applied to Li-ion batteries in order to realize a device with higher areal energy densities.
Over the past few years, we have seen an explosion of interest in electronic devices based on paper or textile components. These substrates are attractive because they can impart flexibility and low- cost manufacturing to devices such as transistors, circuits, light-emitting diodes, and batteries. They also can be folded. Researchers now have have shown that paper-folding concepts can be applied to Li-ion batteries in order to realize a device with higher areal energy densities.
Oct 1st, 2013
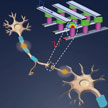 The idea of building bio-inspired cognitive adaptive solid-state devices has been around for decades. It forms the basis for synaptic electronics, a field of research that aims to build artificial synaptic devices to emulate the computation performed by biological synapses. Synapses dominate the architecture of the brain and are responsible for massive parallelism, structural plasticity, and robustness of the brain. They are also crucial to biological computations that underlie perception and learning. Therefore, a compact nanoelectronic device emulating the functions and plasticity of biological synapses will be the most important building block of brain-inspired computational systems. Now, a new review looks at the recent progress of synaptic electronics.
The idea of building bio-inspired cognitive adaptive solid-state devices has been around for decades. It forms the basis for synaptic electronics, a field of research that aims to build artificial synaptic devices to emulate the computation performed by biological synapses. Synapses dominate the architecture of the brain and are responsible for massive parallelism, structural plasticity, and robustness of the brain. They are also crucial to biological computations that underlie perception and learning. Therefore, a compact nanoelectronic device emulating the functions and plasticity of biological synapses will be the most important building block of brain-inspired computational systems. Now, a new review looks at the recent progress of synaptic electronics.
Sep 24th, 2013
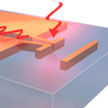 A team of researchers in Germany and the U.S. demonstrates that it is possible to operate extremely compact optical circuits on the nanoscale, a size scale that makes it compatible and potentially competitive with state-of-the-art electronic microchips, while substantially reducing the limiting factor of heating loss and while strongly increasing the efficiency to funnel infrared laser light into these circuits with a novel design of optical nanoantennas.
A team of researchers in Germany and the U.S. demonstrates that it is possible to operate extremely compact optical circuits on the nanoscale, a size scale that makes it compatible and potentially competitive with state-of-the-art electronic microchips, while substantially reducing the limiting factor of heating loss and while strongly increasing the efficiency to funnel infrared laser light into these circuits with a novel design of optical nanoantennas.
Sep 13th, 2013
 Notwithstanding the red-hot research area of flexible electronics, today's state-of-the-art electronic devices rely on rigid and brittle mono-crystalline silicon based transistors which are unmatched with regard to low-cost production, high-performance computing, and ultra-low power consumption. Researchers have now developed a low-cost generic batch process using a state-of-the-art CMOS process to transform conventional silicon electronics into flexible and transparent electronics while retaining its high-performance, ultra-large-scale-integration density and cost.
Notwithstanding the red-hot research area of flexible electronics, today's state-of-the-art electronic devices rely on rigid and brittle mono-crystalline silicon based transistors which are unmatched with regard to low-cost production, high-performance computing, and ultra-low power consumption. Researchers have now developed a low-cost generic batch process using a state-of-the-art CMOS process to transform conventional silicon electronics into flexible and transparent electronics while retaining its high-performance, ultra-large-scale-integration density and cost.
Sep 11th, 2013
 Silicon offers a unique combination between mechanical and electrical properties making it one of the most developed materials in semiconductor industry. However, silicon is brittle and cannot be flexed, hindering its potential for high performance electronics that is flexible, stretchable or applied to irregular shapes. Researchers have now developed a pragmatic approach to achieve high performance integrated electronic systems, including thermoelectric energy harvesters, onto flexible silicon substrates.
Silicon offers a unique combination between mechanical and electrical properties making it one of the most developed materials in semiconductor industry. However, silicon is brittle and cannot be flexed, hindering its potential for high performance electronics that is flexible, stretchable or applied to irregular shapes. Researchers have now developed a pragmatic approach to achieve high performance integrated electronic systems, including thermoelectric energy harvesters, onto flexible silicon substrates.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





