Showing Spotlights 1425 - 1432 of 2781 in category All (newest first):
 The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
Sep 20th, 2012
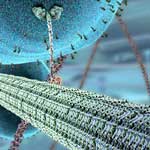 A look at what is happening in the field of artificial nanoscale motors and molecular machinery. These nanomachines could one day perform functions similar to the biological molecular motors found in living cells, things like transporting and assembling molecules, or facilitating chemical reactions by pumping protons through membranes. Researchers have reported a light-powered DNA locomotion device that is capable of autonomous and reversible motion along an oligonucleotide track. The direction of motion can be switched using different wavelengths of light. Compared with other reported DNA walkers, this new strategy not only preserves the autonomous and controllable movement but also provides a reusable track, making it feasible to reset the device after the complete trip, as observed in nature for kinesin and myosin.
A look at what is happening in the field of artificial nanoscale motors and molecular machinery. These nanomachines could one day perform functions similar to the biological molecular motors found in living cells, things like transporting and assembling molecules, or facilitating chemical reactions by pumping protons through membranes. Researchers have reported a light-powered DNA locomotion device that is capable of autonomous and reversible motion along an oligonucleotide track. The direction of motion can be switched using different wavelengths of light. Compared with other reported DNA walkers, this new strategy not only preserves the autonomous and controllable movement but also provides a reusable track, making it feasible to reset the device after the complete trip, as observed in nature for kinesin and myosin.
Sep 19th, 2012
 Labelling is a central regulatory tool for risk governance. It aims at meeting a number of goals: It should enable consumers to make informed purchase decisions, avoid consumers being misled and promote innovation. Hence, consumers take part in the risk management of different product groups. Labelling of nanotechnology products has been part of the early discussion on nanotechnology regulation, both at national and EU level. Member states have refrained from independent national initiatives. However, nano-specific labelling obligations have been adopted in European law for cosmetics, food and biocidal products. In contrast, international initiatives for voluntary labelling have not succeeded on the market.
Labelling is a central regulatory tool for risk governance. It aims at meeting a number of goals: It should enable consumers to make informed purchase decisions, avoid consumers being misled and promote innovation. Hence, consumers take part in the risk management of different product groups. Labelling of nanotechnology products has been part of the early discussion on nanotechnology regulation, both at national and EU level. Member states have refrained from independent national initiatives. However, nano-specific labelling obligations have been adopted in European law for cosmetics, food and biocidal products. In contrast, international initiatives for voluntary labelling have not succeeded on the market.
Sep 17th, 2012
 In the construction industry and in architecture, nanotechnology and nanomaterials provide new opportunities. 'Nano-products' for construction purposes are currently found in four main sectors: cement-bound construction materials, noise reduction and thermal insulation or temperature regulation, surface coatings to improve the functionalities of various materials, and fire protection. At the present time, nanomaterials - and therefore 'nano-products' - remain considerably more expensive than conventional alternatives due to the required production technology, and the technical performance of many products remains to be demonstrated. Also, Information on which nanomaterial is found in which form and concentration in a product is often unavailable, particularly to end users.
In the construction industry and in architecture, nanotechnology and nanomaterials provide new opportunities. 'Nano-products' for construction purposes are currently found in four main sectors: cement-bound construction materials, noise reduction and thermal insulation or temperature regulation, surface coatings to improve the functionalities of various materials, and fire protection. At the present time, nanomaterials - and therefore 'nano-products' - remain considerably more expensive than conventional alternatives due to the required production technology, and the technical performance of many products remains to be demonstrated. Also, Information on which nanomaterial is found in which form and concentration in a product is often unavailable, particularly to end users.
Sep 13th, 2012
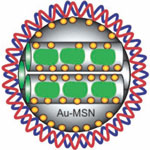
 Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae. Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. Researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles.
Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae. Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. Researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles.
Sep 12th, 2012
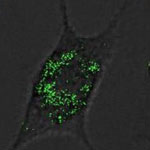
 Nanotechnology is gaining significant interest in plant sciences with research focusing on investigating plant genomics and gene function as well as improving crop species. The impact on agriculture could be dramatic. Most of the work done with nanoparticles in plant sciences relies on a passive uptake of nanoparticles by plant cells - a process that cannot be controlled. Plant cells differ from animal cells in several aspects, a major one being that they possess a wall surrounding them to provide, among other things, mechanical and structural support. Researchers are commonly using surface-functionalized silica nanoparticles as nonviral nanocarriers for experimental drug and DNA delivery into animal cells but their use with plants so far was limited due to the barriers posed by the cell wall. Following up on previous work, researchers have made this process more efficient and introduced a much more challenging biomolecule to plant cells - proteins.
Nanotechnology is gaining significant interest in plant sciences with research focusing on investigating plant genomics and gene function as well as improving crop species. The impact on agriculture could be dramatic. Most of the work done with nanoparticles in plant sciences relies on a passive uptake of nanoparticles by plant cells - a process that cannot be controlled. Plant cells differ from animal cells in several aspects, a major one being that they possess a wall surrounding them to provide, among other things, mechanical and structural support. Researchers are commonly using surface-functionalized silica nanoparticles as nonviral nanocarriers for experimental drug and DNA delivery into animal cells but their use with plants so far was limited due to the barriers posed by the cell wall. Following up on previous work, researchers have made this process more efficient and introduced a much more challenging biomolecule to plant cells - proteins.
Sep 10th, 2012
 Multiphoton lithography (MPL) is a microfabrication technique used to create three-dimensional microscale objects with complex geometrical arrangements. Of the various chemistries used to produce solid forms in MPL, protein photocrosslinking has been of particular value in biological applications. In new work, researchers have now described a strategy for creating a nearly unlimited range of microforms from crosslinked protein, including structures composed of multiple proteins. They also describe MPL microfabrication of complex unconstrained objects using high-viscosity protein-based reagents. To avoid drift during fabrication of microforms that are not in integral contact with a surface, the team developed a methodology for producing high-viscosity protein-based reagents, or "protogels". These materials allow the fabrication of protein-based objects that retain rotational and translational degrees of freedom.
Multiphoton lithography (MPL) is a microfabrication technique used to create three-dimensional microscale objects with complex geometrical arrangements. Of the various chemistries used to produce solid forms in MPL, protein photocrosslinking has been of particular value in biological applications. In new work, researchers have now described a strategy for creating a nearly unlimited range of microforms from crosslinked protein, including structures composed of multiple proteins. They also describe MPL microfabrication of complex unconstrained objects using high-viscosity protein-based reagents. To avoid drift during fabrication of microforms that are not in integral contact with a surface, the team developed a methodology for producing high-viscosity protein-based reagents, or "protogels". These materials allow the fabrication of protein-based objects that retain rotational and translational degrees of freedom.
Sep 6th, 2012
 Lithography based on block copolymer self-assembly has received significant attention due to the ability to achieve morphologies with dimensions in the range of 10 to 20 nm or even below. Block copolymer lithography is a cost-effective, parallel, and scalable nanolithography for densely packed periodic arrays of nanoscale features, whose typical dimension scale is beyond the resolution limit of conventional photolithography. Researchers have now introduced a conceptually new and versatile strategy to achieve asymmetric line patterns. This is the first work to demonstrate that highly asymmetric line nanopatterning is possible even though a block copolymer self-assembly technique is used.
Lithography based on block copolymer self-assembly has received significant attention due to the ability to achieve morphologies with dimensions in the range of 10 to 20 nm or even below. Block copolymer lithography is a cost-effective, parallel, and scalable nanolithography for densely packed periodic arrays of nanoscale features, whose typical dimension scale is beyond the resolution limit of conventional photolithography. Researchers have now introduced a conceptually new and versatile strategy to achieve asymmetric line patterns. This is the first work to demonstrate that highly asymmetric line nanopatterning is possible even though a block copolymer self-assembly technique is used.
Sep 5th, 2012
 The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





