Showing Spotlights 1537 - 1544 of 2783 in category All (newest first):
 Thermal interface materials (TIMs) are essential ingredients of thermal management. TIMs are applied between the heat source, e.g. computer chips, and heat sinks and their function is to fill the voids and grooves created by imperfect surface finish of mating surfaces. Conventional TIMs filled with thermally conductive particles require high volume fractions of filler particles. But now, researchers have achieved a record enhancement of the thermal conductivity of TIMs by addition of an optimized mixture of graphene and multilayer graphene. The thermal conductivity of the epoxy matrix material was increased by an impressive factor of 23 at the 10 volume % of graphene loading. The epoxy-graphene composite preserved all the properties required for industrial TIM applications.
Thermal interface materials (TIMs) are essential ingredients of thermal management. TIMs are applied between the heat source, e.g. computer chips, and heat sinks and their function is to fill the voids and grooves created by imperfect surface finish of mating surfaces. Conventional TIMs filled with thermally conductive particles require high volume fractions of filler particles. But now, researchers have achieved a record enhancement of the thermal conductivity of TIMs by addition of an optimized mixture of graphene and multilayer graphene. The thermal conductivity of the epoxy matrix material was increased by an impressive factor of 23 at the 10 volume % of graphene loading. The epoxy-graphene composite preserved all the properties required for industrial TIM applications.
Jan 30th, 2012
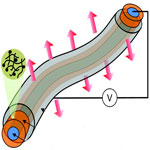 OLEDs - organic light-emitting diodes - are full of promise for a range of practical applications. OLED technology is based on the phenomenon that certain organic materials emit light when fed by an electric current and it is already used in small electronic device displays in mobile phones, MP3 players, digital cameras, and also some TV screens. OLEDs in fiber form could lead to revolutionary applications by integrating optical and optoelectronic devices into textile. Combined with nanoelectronic devices, we might one day see flexible optical sensors and display screens woven into shirts and other garments. You could literally wear your next-generation smart phone or iPad on your sleeves; including the solar panels to power them.
OLEDs - organic light-emitting diodes - are full of promise for a range of practical applications. OLED technology is based on the phenomenon that certain organic materials emit light when fed by an electric current and it is already used in small electronic device displays in mobile phones, MP3 players, digital cameras, and also some TV screens. OLEDs in fiber form could lead to revolutionary applications by integrating optical and optoelectronic devices into textile. Combined with nanoelectronic devices, we might one day see flexible optical sensors and display screens woven into shirts and other garments. You could literally wear your next-generation smart phone or iPad on your sleeves; including the solar panels to power them.
Jan 27th, 2012
 In a project funded by the Danish Environmental Protection Agency, researchers have initiated the development of a screening tool called NanoRiskCat for the evaluation of exposure and hazard of nanomaterials contained in products for professional and private use. The project's aim was to identify, categorize and rank the possible exposure and hazards associated with a nanomaterial in a product. NanoRiskCat is using a stepwise approach based on existing data on the conventional form of the chemical as well as the data that may exist on the nanoform. However, the tool still needs to be further validated and tested on a series of various nano products in order to adjust and optimize the concept and thereby to achieve a screening tool as informative and practical as possible.
In a project funded by the Danish Environmental Protection Agency, researchers have initiated the development of a screening tool called NanoRiskCat for the evaluation of exposure and hazard of nanomaterials contained in products for professional and private use. The project's aim was to identify, categorize and rank the possible exposure and hazards associated with a nanomaterial in a product. NanoRiskCat is using a stepwise approach based on existing data on the conventional form of the chemical as well as the data that may exist on the nanoform. However, the tool still needs to be further validated and tested on a series of various nano products in order to adjust and optimize the concept and thereby to achieve a screening tool as informative and practical as possible.
Jan 25th, 2012
 Fabrication conditions for nanoscale field-effect transistors (nano-FETs) have to meet very high requirements in order for these transistors to be used reliably as ultrasensitive and label-free molecular sensors in medical and environmental applications. Current fabrication routes for silicon-nanowire sensor construction involve high-cost, high-complexity - and often low-yield - top-down techniques such as e-beam lithography and focused ion beam. An alternative, and lower-cost, fabrication method is the use of pre-synthesized nanotubes or nanowires that are integrated into microstructures to form nano-FET sensors. Now, researchers have developed an automated vision-based nanomanipulation technique that is capable of precisely controlling the number of nanowires incorporated into each device.
Fabrication conditions for nanoscale field-effect transistors (nano-FETs) have to meet very high requirements in order for these transistors to be used reliably as ultrasensitive and label-free molecular sensors in medical and environmental applications. Current fabrication routes for silicon-nanowire sensor construction involve high-cost, high-complexity - and often low-yield - top-down techniques such as e-beam lithography and focused ion beam. An alternative, and lower-cost, fabrication method is the use of pre-synthesized nanotubes or nanowires that are integrated into microstructures to form nano-FET sensors. Now, researchers have developed an automated vision-based nanomanipulation technique that is capable of precisely controlling the number of nanowires incorporated into each device.
Jan 24th, 2012
 In January 2007, China successfully tested an Anti-satellite (ASAT) missile system by destroying their own defunct LEO satellite, which generated huge amounts of space debris. This ASAT test raised worldwide concerns about the vulnerability of satellites and other space assets and possibility of triggering an arms race in space. In order to meet emerging challenges posed by such ASAT missile systems, military strategists and researchers are developing novel technologies to protect their space assets. In view of the above, Raytheon Company has developed a counter measure system using quantum dots to protect space assets such as satellites from missile attacks. They have developed a decoy consisting of quantum dots of different sizes and shapes that are engineered to emit radiation having a radiation profile similar to that of the asset.
In January 2007, China successfully tested an Anti-satellite (ASAT) missile system by destroying their own defunct LEO satellite, which generated huge amounts of space debris. This ASAT test raised worldwide concerns about the vulnerability of satellites and other space assets and possibility of triggering an arms race in space. In order to meet emerging challenges posed by such ASAT missile systems, military strategists and researchers are developing novel technologies to protect their space assets. In view of the above, Raytheon Company has developed a counter measure system using quantum dots to protect space assets such as satellites from missile attacks. They have developed a decoy consisting of quantum dots of different sizes and shapes that are engineered to emit radiation having a radiation profile similar to that of the asset.
Jan 23rd, 2012
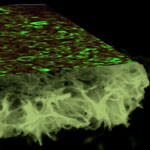 The huge economic impact of the corrosion of metallic structures is a very important issue for all modern societies. Estimates for the cost of corrosion degradation run to about 200 billion euros a year in Europe and over $270 billion a year in the U.S. The annual cost of corrosion consists of both direct costs and indirect costs. The direct costs are related to the costs of design, manufacturing, and construction in order to provide corrosion protection, and the indirect costs are concerned with corrosion-related inspection, maintenance and repairs. Researchers in Germany have now developed a novel and effective encapsulation system for metal protection against a wide range of biological and chemical aggressive agents.
The huge economic impact of the corrosion of metallic structures is a very important issue for all modern societies. Estimates for the cost of corrosion degradation run to about 200 billion euros a year in Europe and over $270 billion a year in the U.S. The annual cost of corrosion consists of both direct costs and indirect costs. The direct costs are related to the costs of design, manufacturing, and construction in order to provide corrosion protection, and the indirect costs are concerned with corrosion-related inspection, maintenance and repairs. Researchers in Germany have now developed a novel and effective encapsulation system for metal protection against a wide range of biological and chemical aggressive agents.
Jan 20th, 2012
 A University of Ulster laboratory has found a simple, low cost and environmentally friendly way to turn common graphite flakes into bulk amounts of either high quality graphene nanosheets or quantum dots. Such structures could lead to new nanoelectronics and energy conversion technologies. The scientists discovered a simple process, which is quicker and environmentally friendlier than currently established techniques for making high quality graphene nanosheets and quantum dots at an industrial scale. The most important attribute of the produced graphene nanosheets and quantum dots compared to those reported in the literature is that they are clean from any solvent contamination and possess a low concentration of oxygen, which is inherited from the starting graphite powder.
A University of Ulster laboratory has found a simple, low cost and environmentally friendly way to turn common graphite flakes into bulk amounts of either high quality graphene nanosheets or quantum dots. Such structures could lead to new nanoelectronics and energy conversion technologies. The scientists discovered a simple process, which is quicker and environmentally friendlier than currently established techniques for making high quality graphene nanosheets and quantum dots at an industrial scale. The most important attribute of the produced graphene nanosheets and quantum dots compared to those reported in the literature is that they are clean from any solvent contamination and possess a low concentration of oxygen, which is inherited from the starting graphite powder.
Jan 18th, 2012
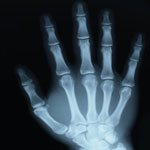 Theoretical and experimental studies over the past few years have demonstrated that carbon nanotubes (CNTs) could exhibit novel and outstanding electromagnetic effects. Researchers have used this to fabricate various types of CNT nanocomposite materials for electromagnetic interference shielding, outperforming conventional shielding. In new work, scientists have now demonstrated the enhanced X-ray shielding of CNTs. Previously, scientists believed that X-ray attenuation - the gradual loss in intensity of X-rays as they travel through a medium - was determined by the atomic number of a material and that its structure didn't matter. What the team found, though, was that that the mass attenuation coefficient of CNTs was by 20-50% higher than that observed for highly oriented pyrolytic graphite and fullerenes.
Theoretical and experimental studies over the past few years have demonstrated that carbon nanotubes (CNTs) could exhibit novel and outstanding electromagnetic effects. Researchers have used this to fabricate various types of CNT nanocomposite materials for electromagnetic interference shielding, outperforming conventional shielding. In new work, scientists have now demonstrated the enhanced X-ray shielding of CNTs. Previously, scientists believed that X-ray attenuation - the gradual loss in intensity of X-rays as they travel through a medium - was determined by the atomic number of a material and that its structure didn't matter. What the team found, though, was that that the mass attenuation coefficient of CNTs was by 20-50% higher than that observed for highly oriented pyrolytic graphite and fullerenes.
Jan 16th, 2012
 Thermal interface materials (TIMs) are essential ingredients of thermal management. TIMs are applied between the heat source, e.g. computer chips, and heat sinks and their function is to fill the voids and grooves created by imperfect surface finish of mating surfaces. Conventional TIMs filled with thermally conductive particles require high volume fractions of filler particles. But now, researchers have achieved a record enhancement of the thermal conductivity of TIMs by addition of an optimized mixture of graphene and multilayer graphene. The thermal conductivity of the epoxy matrix material was increased by an impressive factor of 23 at the 10 volume % of graphene loading. The epoxy-graphene composite preserved all the properties required for industrial TIM applications.
Thermal interface materials (TIMs) are essential ingredients of thermal management. TIMs are applied between the heat source, e.g. computer chips, and heat sinks and their function is to fill the voids and grooves created by imperfect surface finish of mating surfaces. Conventional TIMs filled with thermally conductive particles require high volume fractions of filler particles. But now, researchers have achieved a record enhancement of the thermal conductivity of TIMs by addition of an optimized mixture of graphene and multilayer graphene. The thermal conductivity of the epoxy matrix material was increased by an impressive factor of 23 at the 10 volume % of graphene loading. The epoxy-graphene composite preserved all the properties required for industrial TIM applications.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





