Showing Spotlights 9 - 16 of 137 in category All (newest first):
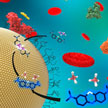 Meeting the need for a reliable, sensitive, and accurate methodology for the detection of nanoparticles in complex samples, using low-cost and portable instrumentation, scientists have developed a novel methodology to quickly screen for the presence and reactivity of nanoparticles in commercial, environmental, and biological samples. A colorimetric assay - similar to a swimming pool test kit - tests for the presence or absence of nanoparticles in biological and environmental relevant samples with sufficient sensitivity at part per billion concentration levels.
Meeting the need for a reliable, sensitive, and accurate methodology for the detection of nanoparticles in complex samples, using low-cost and portable instrumentation, scientists have developed a novel methodology to quickly screen for the presence and reactivity of nanoparticles in commercial, environmental, and biological samples. A colorimetric assay - similar to a swimming pool test kit - tests for the presence or absence of nanoparticles in biological and environmental relevant samples with sufficient sensitivity at part per billion concentration levels.
Feb 20th, 2015
 Insurance companies are major stakeholders capable of contributing to the safer and more sustainable development of nanotechnologies and nanomaterials. This is owed to the fact that the insurance industry is one of the bearers of potential losses that can arise from the production and use of nanomaterials and nanotechnology applications. Researchers have examined how the insurance market perception of nanotechnology can influence the sustainability of technological advances and insurers? concern for nanotechnology risks. They claim that, despite its role in sustaining technology development in modern society, insurers' perception on nanomaterials has been largely overlooked by researchers and regulators alike.
Insurance companies are major stakeholders capable of contributing to the safer and more sustainable development of nanotechnologies and nanomaterials. This is owed to the fact that the insurance industry is one of the bearers of potential losses that can arise from the production and use of nanomaterials and nanotechnology applications. Researchers have examined how the insurance market perception of nanotechnology can influence the sustainability of technological advances and insurers? concern for nanotechnology risks. They claim that, despite its role in sustaining technology development in modern society, insurers' perception on nanomaterials has been largely overlooked by researchers and regulators alike.
Dec 16th, 2014
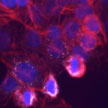 Numerous nanotoxicological studies reporting effects of nanomaterials typically address a single exposure at high dosages that are irrelevant to realistic human exposure. Recognizing that acute in vitro work had extremely low correlation to in vivo nanomaterial studies, coupled with the recognition that the unique characteristics that distinguish nanomaterials vary as a function of time, researchers sought to identify a model that would allow for the evaluation of nanomaterial behavior over a 3-month period, but be carried out in an in vitro model.
Numerous nanotoxicological studies reporting effects of nanomaterials typically address a single exposure at high dosages that are irrelevant to realistic human exposure. Recognizing that acute in vitro work had extremely low correlation to in vivo nanomaterial studies, coupled with the recognition that the unique characteristics that distinguish nanomaterials vary as a function of time, researchers sought to identify a model that would allow for the evaluation of nanomaterial behavior over a 3-month period, but be carried out in an in vitro model.
Jun 4th, 2014
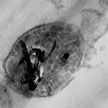 The investigation of effects of engineered nanomaterials on endothelial cells - which form the inner lining of blood vessels - is a critical safety issue. Already, various engineered nanomaterials are being designed for biomedical applications for intravascular use and other nanomaterials may reach the vasculature as a result of occupational, environmental, or other types of exposure. Researchers have now elucidated the mechanism of cytotoxicity of carboxylated MWCNTs on cultured endothelial cells and they show a new potential way of pharmacological cytoprotection against cytotoxic effect of carboxylated MWCNTs.
The investigation of effects of engineered nanomaterials on endothelial cells - which form the inner lining of blood vessels - is a critical safety issue. Already, various engineered nanomaterials are being designed for biomedical applications for intravascular use and other nanomaterials may reach the vasculature as a result of occupational, environmental, or other types of exposure. Researchers have now elucidated the mechanism of cytotoxicity of carboxylated MWCNTs on cultured endothelial cells and they show a new potential way of pharmacological cytoprotection against cytotoxic effect of carboxylated MWCNTs.
Mar 12th, 2014
 Assessing the potential effects of nanomaterials on environment and human health consists of two distinct aspects: To what degree are nanoparticles released from products; and how and to what degree do the released nanoparticles affect organisms? The first aspect is centered on a field called exposure science, the study of human contact to agents - such as chemicals or microbes - found in their surroundings. A new study looks at the release of nanosilver from consumer products for children. The core finding is that the release of silver from nanosilver-containing products depends heavily on how the product is used. The total amount of silver released by a consumer product is likely to be very low and, for the products tested, happened only in the beginning of product life.
Assessing the potential effects of nanomaterials on environment and human health consists of two distinct aspects: To what degree are nanoparticles released from products; and how and to what degree do the released nanoparticles affect organisms? The first aspect is centered on a field called exposure science, the study of human contact to agents - such as chemicals or microbes - found in their surroundings. A new study looks at the release of nanosilver from consumer products for children. The core finding is that the release of silver from nanosilver-containing products depends heavily on how the product is used. The total amount of silver released by a consumer product is likely to be very low and, for the products tested, happened only in the beginning of product life.
Jul 30th, 2013
 The European Commission acknowledges that nanomaterials are revolutionary materials and that important challenges exist in regard to hazard and exposure assessments. Yet, they conclude that current risk-assessment methods are applicable to nanomaterials. Scientists argue that significant changes to REACH and the accompanying annexes are required to answer the call made by the public, downstream users and progressive businesses for clearer and more definite regulatory rules specific to nanomaterials.
The European Commission acknowledges that nanomaterials are revolutionary materials and that important challenges exist in regard to hazard and exposure assessments. Yet, they conclude that current risk-assessment methods are applicable to nanomaterials. Scientists argue that significant changes to REACH and the accompanying annexes are required to answer the call made by the public, downstream users and progressive businesses for clearer and more definite regulatory rules specific to nanomaterials.
May 28th, 2013
 The purpose of the emerging field of nanotoxicity is to recognize and evaluate the hazards and risks of engineered nanomaterials and evaluate safety. Today, we don't even know what the impact of most chemicals is, and that includes products that have been produced by industry for many years. Nevertheless, a general understanding about nanotoxicity is slowly emerging as the body of research on cytotoxicity, genotoxicity, and ecotoxicity of nanomaterials grows. A new review summarizes and discusses recent reports derived from cell lines or animal models concerning the effects of nanomaterials on, and their application in, the endocrine system of mammalian and other species.
The purpose of the emerging field of nanotoxicity is to recognize and evaluate the hazards and risks of engineered nanomaterials and evaluate safety. Today, we don't even know what the impact of most chemicals is, and that includes products that have been produced by industry for many years. Nevertheless, a general understanding about nanotoxicity is slowly emerging as the body of research on cytotoxicity, genotoxicity, and ecotoxicity of nanomaterials grows. A new review summarizes and discusses recent reports derived from cell lines or animal models concerning the effects of nanomaterials on, and their application in, the endocrine system of mammalian and other species.
Feb 18th, 2013
 To date, the predominant focus of the nanotechnology risk research endeavor has been defining the fate, transport, and toxic properties of pristine or "as manufactured" nanomaterials. However, the high surface to volume ratio and reactivity of nanoparticles makes them highly dynamic in environmental systems. The resulting transformations of the nanomaterials will affect their fate, transport, and toxic properties. A recent review summarizes what is known about chemical, physical, and biologically mediated transformations of nanomaterials in natural systems and their effects on the resulting nanomaterial behavior.
To date, the predominant focus of the nanotechnology risk research endeavor has been defining the fate, transport, and toxic properties of pristine or "as manufactured" nanomaterials. However, the high surface to volume ratio and reactivity of nanoparticles makes them highly dynamic in environmental systems. The resulting transformations of the nanomaterials will affect their fate, transport, and toxic properties. A recent review summarizes what is known about chemical, physical, and biologically mediated transformations of nanomaterials in natural systems and their effects on the resulting nanomaterial behavior.
Jan 10th, 2013
 Meeting the need for a reliable, sensitive, and accurate methodology for the detection of nanoparticles in complex samples, using low-cost and portable instrumentation, scientists have developed a novel methodology to quickly screen for the presence and reactivity of nanoparticles in commercial, environmental, and biological samples. A colorimetric assay - similar to a swimming pool test kit - tests for the presence or absence of nanoparticles in biological and environmental relevant samples with sufficient sensitivity at part per billion concentration levels.
Meeting the need for a reliable, sensitive, and accurate methodology for the detection of nanoparticles in complex samples, using low-cost and portable instrumentation, scientists have developed a novel methodology to quickly screen for the presence and reactivity of nanoparticles in commercial, environmental, and biological samples. A colorimetric assay - similar to a swimming pool test kit - tests for the presence or absence of nanoparticles in biological and environmental relevant samples with sufficient sensitivity at part per billion concentration levels.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





