Showing Spotlights 1593 - 1600 of 2780 in category All (newest first):
 Harvesting unexploited energy in the living environment to power small electronic devices and systems is increasingly attracting the attention of research groups around the world. As device sizes shrink to the micro- and even nanoscale, power consumption also decreases to ever lower levels, i.e. microwatts to milliwatts range. And as research findings have shown over the past few years, it is entirely possible to drive such minuscule devices by directly scavenging energy from their working environment. A recent example is the integration of nanogenerators into the inner surface of tires, demonstrating the possibility of energy harvesting from the motion of automobiles.
Harvesting unexploited energy in the living environment to power small electronic devices and systems is increasingly attracting the attention of research groups around the world. As device sizes shrink to the micro- and even nanoscale, power consumption also decreases to ever lower levels, i.e. microwatts to milliwatts range. And as research findings have shown over the past few years, it is entirely possible to drive such minuscule devices by directly scavenging energy from their working environment. A recent example is the integration of nanogenerators into the inner surface of tires, demonstrating the possibility of energy harvesting from the motion of automobiles.
Sep 14th, 2011
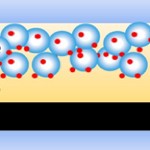 Dye-sensitized solar cells (DSSCs) are among the most promising photovoltaic devices for low-cost light-to-energy conversion with relatively high efficiency. While the DSSC is a fairly mature design, researchers are still trying to improve its efficiency with various techniques. To date, the most commonly used counter electrode in DSSC is fluorine doped tin oxide glass coated with a thin layer of platinum. However, as a noble metal, the low abundance and high cost hinder platinum from being used for large-scale manufacturing. In the quest to seek alternative counter electrode materials for expensive and scarce platinum, a group of scientists has now explored the use of abundant ternary or quaternary materials as potential substitutes for platinum.
Dye-sensitized solar cells (DSSCs) are among the most promising photovoltaic devices for low-cost light-to-energy conversion with relatively high efficiency. While the DSSC is a fairly mature design, researchers are still trying to improve its efficiency with various techniques. To date, the most commonly used counter electrode in DSSC is fluorine doped tin oxide glass coated with a thin layer of platinum. However, as a noble metal, the low abundance and high cost hinder platinum from being used for large-scale manufacturing. In the quest to seek alternative counter electrode materials for expensive and scarce platinum, a group of scientists has now explored the use of abundant ternary or quaternary materials as potential substitutes for platinum.
Sep 13th, 2011
 Self-propelled motion of engineered nanomaterials can be useful in applications such as bottom-up assembly of structures, pattern formation, microfluidic diagnostic systems, or drug delivery at specific locations. While nature has perfected nano- and microscale motor systems, movement at the nanoscale is still a massively challenging problem for nanotechnology researchers. There are various approaches to creating self-powered micro- and nanosized motors and many researchers have focused on catalytic conversion of chemical to mechanical energy. Whereas the catalytic reactions of small molecules have been the focus of recent nanomotor research efforts, polymerization has not attracted any interest; until now. Researchers describe a new type of micromotor that is powered by a polymerization reaction and deposits tiny threads along its trail like a microspider.
Self-propelled motion of engineered nanomaterials can be useful in applications such as bottom-up assembly of structures, pattern formation, microfluidic diagnostic systems, or drug delivery at specific locations. While nature has perfected nano- and microscale motor systems, movement at the nanoscale is still a massively challenging problem for nanotechnology researchers. There are various approaches to creating self-powered micro- and nanosized motors and many researchers have focused on catalytic conversion of chemical to mechanical energy. Whereas the catalytic reactions of small molecules have been the focus of recent nanomotor research efforts, polymerization has not attracted any interest; until now. Researchers describe a new type of micromotor that is powered by a polymerization reaction and deposits tiny threads along its trail like a microspider.
Sep 12th, 2011
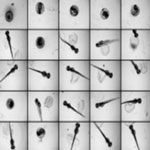 With the mass production of engineered nanoparticles, risk assessment efforts are in need of platforms that offer predictive value to human health and environment, and also possess high throughput screening capacity. Scientists, when turning to a model-organism to help answer genetic questions that cannot be easily addressed in humans, often chose the zebrafish. However, the current screening process in zebrafish involves mostly counting the survival rate, hatching and developmental abnormalities etc. through visual examination of each embryo and/or larvae under a dissecting microscope. Such process is time-consuming, labor-intensive and has limitations on data acquisition as well as statistics analysis. Researchers have now successfully demonstrated two high content imaging platforms to enhance the ability to screen the toxicological effects of nanoparticles in zebrafish embryos.
With the mass production of engineered nanoparticles, risk assessment efforts are in need of platforms that offer predictive value to human health and environment, and also possess high throughput screening capacity. Scientists, when turning to a model-organism to help answer genetic questions that cannot be easily addressed in humans, often chose the zebrafish. However, the current screening process in zebrafish involves mostly counting the survival rate, hatching and developmental abnormalities etc. through visual examination of each embryo and/or larvae under a dissecting microscope. Such process is time-consuming, labor-intensive and has limitations on data acquisition as well as statistics analysis. Researchers have now successfully demonstrated two high content imaging platforms to enhance the ability to screen the toxicological effects of nanoparticles in zebrafish embryos.
Sep 9th, 2011
 We are experiencing an unprecedented resurgence of interest in herbal healing, and 'herbal renaissance' is happening all over the globe. The Western world has begun to acknowledge the importance of traditional medicines as they symbolize safety in contrast to the allopathic medicines, which tend to produce undesirable side effects and are lacking in curative value. In the realm of medicine, nanotechnology holds enormous promise for benefitting society by potentially reducing the miseries of people suffering from grave illnesses and save a great number of lives. Traditional Oriental medicine would greatly benefit by integrating with the scientific advancements in medical science and diagnostics in concert with nanotechnology. This trinity may usher in a new era of affordable, safe and effective medicinal system.
We are experiencing an unprecedented resurgence of interest in herbal healing, and 'herbal renaissance' is happening all over the globe. The Western world has begun to acknowledge the importance of traditional medicines as they symbolize safety in contrast to the allopathic medicines, which tend to produce undesirable side effects and are lacking in curative value. In the realm of medicine, nanotechnology holds enormous promise for benefitting society by potentially reducing the miseries of people suffering from grave illnesses and save a great number of lives. Traditional Oriental medicine would greatly benefit by integrating with the scientific advancements in medical science and diagnostics in concert with nanotechnology. This trinity may usher in a new era of affordable, safe and effective medicinal system.
Sep 8th, 2011
 Electronic memory devices are increasingly expected to provide not only greater storage density, but also faster access to information. As storage density increases, however, power consumption and unwanted heat generation also increase, and the fidelity of accessing the memory is frequently diminished. Various platforms exist to overcome these hurdles and, increasingly, graphene finds it way into computer memory technology. The most recent example are experiments that demonstrate the benefits of graphene as a platform for flash memory which show the potential to exceed the performance of current flash memory technology by utilizing the intrinsic properties of graphene.
Electronic memory devices are increasingly expected to provide not only greater storage density, but also faster access to information. As storage density increases, however, power consumption and unwanted heat generation also increase, and the fidelity of accessing the memory is frequently diminished. Various platforms exist to overcome these hurdles and, increasingly, graphene finds it way into computer memory technology. The most recent example are experiments that demonstrate the benefits of graphene as a platform for flash memory which show the potential to exceed the performance of current flash memory technology by utilizing the intrinsic properties of graphene.
Sep 5th, 2011
 In the last couple of years, there has been particularly growing interest worldwide in exploring ways of finding suitable solutions to clean up oil spills and deal with industrial oily wastewater through use of nanomaterials. Key for the success of these materials is a high separation capacity, with resistance to oil fouling, and that are easily recyclable. Oil/water separation is an interfacial challenge, and novel materials designed to possess special wettability have different interaction and affinity for oil and water, thus can realize the separation. Until now, researches in this field all focus on materials with both hydrophobic and oleophilic properties. However, the oil-removing type of materials is easily fouled even blocked up by oils because of their intrinsic oleophilic property. A novel superhydrophilic and underwater superoleophobic hydrogel coated mesh can selectively separate water from oil/water mixtures effectively and without any extra power.
In the last couple of years, there has been particularly growing interest worldwide in exploring ways of finding suitable solutions to clean up oil spills and deal with industrial oily wastewater through use of nanomaterials. Key for the success of these materials is a high separation capacity, with resistance to oil fouling, and that are easily recyclable. Oil/water separation is an interfacial challenge, and novel materials designed to possess special wettability have different interaction and affinity for oil and water, thus can realize the separation. Until now, researches in this field all focus on materials with both hydrophobic and oleophilic properties. However, the oil-removing type of materials is easily fouled even blocked up by oils because of their intrinsic oleophilic property. A novel superhydrophilic and underwater superoleophobic hydrogel coated mesh can selectively separate water from oil/water mixtures effectively and without any extra power.
Sep 1st, 2011
 Most molecular probes used in biomedical research require dyes or fluorescence in order to obtain meaningful signals. These probes usually are quite limited with regard to the complexity of what they can image - be it the measurable concentration range or the number of molecules that can be simultaneously detected. This is an issue that is particularly relevant when it comes to track the simultaneous multiple molecular transformations that dictate complicated diseases like cancer. Scientists now have come up with an intriguing new class of molecular probes to solve this problem. They took an existing spectroscopic technique - surface-enhanced Raman scattering (SERS) - and developed a unique class of nanoparticle labels that provide for different responses when excited by laser light.
Most molecular probes used in biomedical research require dyes or fluorescence in order to obtain meaningful signals. These probes usually are quite limited with regard to the complexity of what they can image - be it the measurable concentration range or the number of molecules that can be simultaneously detected. This is an issue that is particularly relevant when it comes to track the simultaneous multiple molecular transformations that dictate complicated diseases like cancer. Scientists now have come up with an intriguing new class of molecular probes to solve this problem. They took an existing spectroscopic technique - surface-enhanced Raman scattering (SERS) - and developed a unique class of nanoparticle labels that provide for different responses when excited by laser light.
Aug 30th, 2011
 Harvesting unexploited energy in the living environment to power small electronic devices and systems is increasingly attracting the attention of research groups around the world. As device sizes shrink to the micro- and even nanoscale, power consumption also decreases to ever lower levels, i.e. microwatts to milliwatts range. And as research findings have shown over the past few years, it is entirely possible to drive such minuscule devices by directly scavenging energy from their working environment. A recent example is the integration of nanogenerators into the inner surface of tires, demonstrating the possibility of energy harvesting from the motion of automobiles.
Harvesting unexploited energy in the living environment to power small electronic devices and systems is increasingly attracting the attention of research groups around the world. As device sizes shrink to the micro- and even nanoscale, power consumption also decreases to ever lower levels, i.e. microwatts to milliwatts range. And as research findings have shown over the past few years, it is entirely possible to drive such minuscule devices by directly scavenging energy from their working environment. A recent example is the integration of nanogenerators into the inner surface of tires, demonstrating the possibility of energy harvesting from the motion of automobiles.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





