Showing Spotlights 1649 - 1656 of 2785 in category All (newest first):
 Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations. Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Researchers in India have recently come up with an innovative method for organic pollutant removal from waste water.
Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations. Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Researchers in India have recently come up with an innovative method for organic pollutant removal from waste water.
Jun 7th, 2011
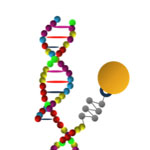 MicroRNAs (miRNAs) are short ribonucleic acid molecules, consisting of 21-25 nucleotide bases, that negatively regulate gene expression, also termed as gene silencing. Each miRNA is thought to regulate multiple genes, and since the human genome encodes hundreds of miRNAs, the potential regulatory circuitry afforded by miRNA is enormous. Recent discoveries suggest the association of specific miRNA sequences with a spectrum of diseases including cardiovascular and autoimmune diseases, as well as with a variety of cancers. It is therefore imperative, for diagnostics and prognostics, to accurately measure the expression levels of target miRNA molecules in patients' tissue samples or body fluids. To that end, researchers have developed an alternative way for the direct analysis of miRNAs in an array format, demonstrating fast and ultrasensitive detection of specific miRNAs.
MicroRNAs (miRNAs) are short ribonucleic acid molecules, consisting of 21-25 nucleotide bases, that negatively regulate gene expression, also termed as gene silencing. Each miRNA is thought to regulate multiple genes, and since the human genome encodes hundreds of miRNAs, the potential regulatory circuitry afforded by miRNA is enormous. Recent discoveries suggest the association of specific miRNA sequences with a spectrum of diseases including cardiovascular and autoimmune diseases, as well as with a variety of cancers. It is therefore imperative, for diagnostics and prognostics, to accurately measure the expression levels of target miRNA molecules in patients' tissue samples or body fluids. To that end, researchers have developed an alternative way for the direct analysis of miRNAs in an array format, demonstrating fast and ultrasensitive detection of specific miRNAs.
Jun 6th, 2011
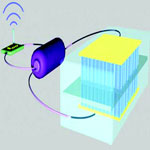 Fabrication of a single nanodevice is no longer the state of the art in nanotechnology. The leading edge - and also currently the most challenging area in nanotechnology - is research that leads to a self-powered nanoscale system that is driven by the energy harvested from its environment and that can perform its work independently and sustainable. This is a key step toward self-powered nanotechnology, which is vitally important for medical science, environmental monitoring, defence technology and even personal electronics. A research team has now provided the first demonstration that a nanogenerator can be strong enough to power a device with the capability of sensing, data processing and wireless data transmission. This is a powerful demonstration of the self-powered nanosystem and its potential applications.
Fabrication of a single nanodevice is no longer the state of the art in nanotechnology. The leading edge - and also currently the most challenging area in nanotechnology - is research that leads to a self-powered nanoscale system that is driven by the energy harvested from its environment and that can perform its work independently and sustainable. This is a key step toward self-powered nanotechnology, which is vitally important for medical science, environmental monitoring, defence technology and even personal electronics. A research team has now provided the first demonstration that a nanogenerator can be strong enough to power a device with the capability of sensing, data processing and wireless data transmission. This is a powerful demonstration of the self-powered nanosystem and its potential applications.
Jun 1st, 2011
 The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
The small particles in volcanic ash can damage jet aircraft engines. During jet engine operation, large amounts of air are sucked in. If this air contains ash particles, especially glass-rich silicate ash, they will melt in the jet turbine's operating temperature of 1400-1500C and subsequently fuse to the turbine blades. This will drive the finely tuned blades out-of-balance and could stall the engine. In today's jet engines, a thermal barrier coating (TBC) insulates metallic engine parts from the heat generated by the combustion process. If ingested ash melts onto this coating it will penetrate it and, upon cooling, the molten ash forms a brittle glass that flakes off, taking the coating with it. A team of researchers have now examined a new class of ceramic TBC that could offer jet engines special protection against volcanic ash damage in the future.
May 31st, 2011
 Groundbreaking research has shown a quantum atom has been tracked inside a living human cell and may lead to improvements in the testing and development of new drugs. Researchers conducted studies that confirm that non-invasive quantum measurement is possible on nanodiamonds containing a single nitrogen-vacancy (NV) spin moving within living cells. Studying the quantum properties of a single NV defect within a diamond nanocrystal, the researchers demonstrate a new technique which enables the orientation of a nanoparticle to be determined to an accuracy of less than one degree in an acquisition time of 89 milliseconds. This new technique offers biologists an extra degree of freedom when studying the translational motion of nanoparticles. Monitoring the coherence from a single electron spin paves the ways for nanoscale bio-magnetometry allowing scientists to probe changes in the cell's electromagnetic environment.
Groundbreaking research has shown a quantum atom has been tracked inside a living human cell and may lead to improvements in the testing and development of new drugs. Researchers conducted studies that confirm that non-invasive quantum measurement is possible on nanodiamonds containing a single nitrogen-vacancy (NV) spin moving within living cells. Studying the quantum properties of a single NV defect within a diamond nanocrystal, the researchers demonstrate a new technique which enables the orientation of a nanoparticle to be determined to an accuracy of less than one degree in an acquisition time of 89 milliseconds. This new technique offers biologists an extra degree of freedom when studying the translational motion of nanoparticles. Monitoring the coherence from a single electron spin paves the ways for nanoscale bio-magnetometry allowing scientists to probe changes in the cell's electromagnetic environment.
May 26th, 2011
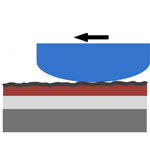 Interaction forces between contact surfaces, such as the adhesion and friction forces, are crucial in many applications at the nanoscale because of the high surface-to-volume ratio of nanomaterials and nanodevices. Over the last decade, various solid lubricant materials, micro/nano patterns, and surface treatment processes have been developed for efficient operation and extended lifetime in MEMS/NEMS applications, and for various fabrication processes such as nanoimprint lithography and transfer printing. From a practical viewpoint, large-area CVD-graphene holds the greatest promise as a solid lubricant because of its excellent scalability and transferability. So far, the tribological characteristics of CVD-graphene at the micro- and nanoscale have not been reported yet. Researchers have now demonstrated the feasibility of using large-area CVD-graphene as a solid lubricant.
Interaction forces between contact surfaces, such as the adhesion and friction forces, are crucial in many applications at the nanoscale because of the high surface-to-volume ratio of nanomaterials and nanodevices. Over the last decade, various solid lubricant materials, micro/nano patterns, and surface treatment processes have been developed for efficient operation and extended lifetime in MEMS/NEMS applications, and for various fabrication processes such as nanoimprint lithography and transfer printing. From a practical viewpoint, large-area CVD-graphene holds the greatest promise as a solid lubricant because of its excellent scalability and transferability. So far, the tribological characteristics of CVD-graphene at the micro- and nanoscale have not been reported yet. Researchers have now demonstrated the feasibility of using large-area CVD-graphene as a solid lubricant.
May 25th, 2011
 It is widely believed that stem cell therapies have the potential to revolutionize the treatment of human diseases. Key to the success of such therapies are two crucial properties: the ability of stem cells to develop into any specialized cell type depending on the specific need at hand; and the ability to guide the fate of the stem cells by various external factors. Researchers in Asia have now demonstrated that graphene provides a promising biocompatible scaffold that does not hamper the proliferation of human mesenchymal stem cells and accelerates their specific differentiation into bone cells. The differentiation rate is comparable to the one achieved with common growth factors, demonstrating graphene's potential for stem cell research.
It is widely believed that stem cell therapies have the potential to revolutionize the treatment of human diseases. Key to the success of such therapies are two crucial properties: the ability of stem cells to develop into any specialized cell type depending on the specific need at hand; and the ability to guide the fate of the stem cells by various external factors. Researchers in Asia have now demonstrated that graphene provides a promising biocompatible scaffold that does not hamper the proliferation of human mesenchymal stem cells and accelerates their specific differentiation into bone cells. The differentiation rate is comparable to the one achieved with common growth factors, demonstrating graphene's potential for stem cell research.
May 24th, 2011
 Germanium was the basic material of first-generation transistors in the late 1940s and early 1950s before it was replaced by silicon (the first silicon transistor was produced by Texas Instruments in 1954). Using germanium instead of silicon as transistor material would enable faster chips containing smaller transistors because higher switching speeds than in silicon could be achieved using germanium. A novel fabrication route demonstrated by a research team in Australia, using a combination of scanning tunneling microscope (STM) lithography and high-quality crystal growth, opens up an entire new area where quantum behavior of highly confined electrons in germanium can be studied for the first time.
Germanium was the basic material of first-generation transistors in the late 1940s and early 1950s before it was replaced by silicon (the first silicon transistor was produced by Texas Instruments in 1954). Using germanium instead of silicon as transistor material would enable faster chips containing smaller transistors because higher switching speeds than in silicon could be achieved using germanium. A novel fabrication route demonstrated by a research team in Australia, using a combination of scanning tunneling microscope (STM) lithography and high-quality crystal growth, opens up an entire new area where quantum behavior of highly confined electrons in germanium can be studied for the first time.
May 20th, 2011
 Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations. Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Researchers in India have recently come up with an innovative method for organic pollutant removal from waste water.
Water treatment is important for human consumption and environmental protection. Non-trivial purification of water involves removal of toxic ions, organic impurities, microbes and their by-products as well as scooping oil spills. The removal of organic contaminants from water is a major industrial concern. The challenging goal here is to detect, decompose and remove contaminants present usually in low concentrations. Towards this end, different types of sorbent materials have been developed to date, the most common being activated carbon. Though the use of activated carbon is still considered to be one of the best method, the disposal of adsorbed contaminants along with the adsorbent is a major concern. Researchers in India have recently come up with an innovative method for organic pollutant removal from waste water.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





