Showing Spotlights 1673 - 1680 of 2784 in category All (newest first):
 Life cycle assessment is an essential tool for ensuring the safe, responsible, and sustainable commercialization of a new technology. With missing data about the large scale impact of nanotechnology, life cycle assessments of potential nanoproducts should form an integral part of nanotechnology research at early stages of decision making as it can help in the screening of different process alternatives. Part of any meaningful results from a life cycle assessment is the total quantity of the material under investigation. Especially exposure assessments often begin with estimates based on total amounts of a material produced with the assumption that some fraction of the material in question will ultimately released to the environment. As it turns out, nobody - no research institution, no government agency, no industry association - knows even vaguely how much nanomaterials are manufactured today.
Life cycle assessment is an essential tool for ensuring the safe, responsible, and sustainable commercialization of a new technology. With missing data about the large scale impact of nanotechnology, life cycle assessments of potential nanoproducts should form an integral part of nanotechnology research at early stages of decision making as it can help in the screening of different process alternatives. Part of any meaningful results from a life cycle assessment is the total quantity of the material under investigation. Especially exposure assessments often begin with estimates based on total amounts of a material produced with the assumption that some fraction of the material in question will ultimately released to the environment. As it turns out, nobody - no research institution, no government agency, no industry association - knows even vaguely how much nanomaterials are manufactured today.
Apr 11th, 2011
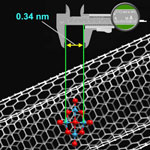 Metrology is the science of measurements, and nanometrology is that part of metrology that relates to measurements at the nanoscale. Many governments worldwide have existing nanotechnology policies and are taking the preliminary steps towards nanometrology strategies, for example in support of pre-normative R+D and standardization work. In this Nanowerk Spotlight, we look at the European Commission funded project Co-Nanomet as an example of the importance of nanometrology as a key enabling technology for quality control at the nanoscale. While a first and obvious benefit of metrology is its potential to improve scientific understanding, a second, equally important, but less obvious benefit of metrology is closely linked to the concepts of quality control or conformity assessment, which means making a decision about whether a product or service conforms to specifications.
Metrology is the science of measurements, and nanometrology is that part of metrology that relates to measurements at the nanoscale. Many governments worldwide have existing nanotechnology policies and are taking the preliminary steps towards nanometrology strategies, for example in support of pre-normative R+D and standardization work. In this Nanowerk Spotlight, we look at the European Commission funded project Co-Nanomet as an example of the importance of nanometrology as a key enabling technology for quality control at the nanoscale. While a first and obvious benefit of metrology is its potential to improve scientific understanding, a second, equally important, but less obvious benefit of metrology is closely linked to the concepts of quality control or conformity assessment, which means making a decision about whether a product or service conforms to specifications.
Apr 8th, 2011
 Many strategies to develop stretchable electronics rely on engineering new constructs from existing materials, e.g. ultrathin, stretchable silicon structures. Another approach is to fabricate ultrathin CMOS circuits on stretchable materials such as polymers. Nanotechnology allows a novel route to materials and structures that can be used to develop human-friendly devices with realistic functions and abilities that would not be feasible by mere extension of conventional technology. New research suggests devices that can act as part of human skin or clothing, and can therefore be used ubiquitously. Such devices could eventually find a wide range of applications in recreation, virtual reality, robotics and health care.
Many strategies to develop stretchable electronics rely on engineering new constructs from existing materials, e.g. ultrathin, stretchable silicon structures. Another approach is to fabricate ultrathin CMOS circuits on stretchable materials such as polymers. Nanotechnology allows a novel route to materials and structures that can be used to develop human-friendly devices with realistic functions and abilities that would not be feasible by mere extension of conventional technology. New research suggests devices that can act as part of human skin or clothing, and can therefore be used ubiquitously. Such devices could eventually find a wide range of applications in recreation, virtual reality, robotics and health care.
Apr 6th, 2011
 A few years ago, researchers determined that the stiffness of cancer cells affects the way they spread. When cancer is becoming metastatic, or invading other organs, the diseased cells must travel throughout the body. Because the cells need to enter the bloodstream and maneuver through tight anatomical spaces, cancer cells are much more flexible, or softer, than normal cells. With this knowledge, researchers wanted to understand the cell mechanics associated with the anticancer treatment of cells from patient samples; in particular they were interested in reporting the effects of green tea extract due to the fact that is was a natural product, it has know anti-cancer effects and it is widely consumed in beverage form around the world.
A few years ago, researchers determined that the stiffness of cancer cells affects the way they spread. When cancer is becoming metastatic, or invading other organs, the diseased cells must travel throughout the body. Because the cells need to enter the bloodstream and maneuver through tight anatomical spaces, cancer cells are much more flexible, or softer, than normal cells. With this knowledge, researchers wanted to understand the cell mechanics associated with the anticancer treatment of cells from patient samples; in particular they were interested in reporting the effects of green tea extract due to the fact that is was a natural product, it has know anti-cancer effects and it is widely consumed in beverage form around the world.
Apr 5th, 2011
 In order to find replacement materials for ITO, scientists have been working with carbon nanotubes, graphene, and other nanoscale materials such as nanowires. While many of these approaches work fine in the lab, upscaleability usually has been an issue. Researchers at Empa, the Swiss Federal Laboratories for Material Science and Technology, have now demonstrated another solution: they presented a transparent and flexible electrode based on a precision fabric with metal and polymer fibers woven into a mesh. The team demonstrated organic solar cells fabricated on their flexible precision fabrics as well as on conventional glass/ITO substrates and found very similar performance characteristics.
In order to find replacement materials for ITO, scientists have been working with carbon nanotubes, graphene, and other nanoscale materials such as nanowires. While many of these approaches work fine in the lab, upscaleability usually has been an issue. Researchers at Empa, the Swiss Federal Laboratories for Material Science and Technology, have now demonstrated another solution: they presented a transparent and flexible electrode based on a precision fabric with metal and polymer fibers woven into a mesh. The team demonstrated organic solar cells fabricated on their flexible precision fabrics as well as on conventional glass/ITO substrates and found very similar performance characteristics.
Apr 4th, 2011
 It started innocently enough with isolated instances of smoke coming out of computers. Then networks crashed. Now, programs are malfunctioning on a large scale, shutting down the Vatican's huge computer infrastructure which it depends on to manage its billions upon billions of investment dollars, real estate portfolios, and art collections. It is difficult to obtain all the details, but it appears that some form of nanotechnology got out of control. Surprisingly, and against its deeply ingrained reflexes of total openness and transparency, the Vatican initially tried to cover the whole thing up. Until a tabloid reporter got wind of what had happened and the whole thing became public with an article today (April 1) in an Italian tabloid that had this sensation-seeking headline splashed all over the front page: "Gay nanobots ballano Bunga-Bunga in Vaticano" - Gay nanobots dance Bunga-Bunga in the Vatican.
It started innocently enough with isolated instances of smoke coming out of computers. Then networks crashed. Now, programs are malfunctioning on a large scale, shutting down the Vatican's huge computer infrastructure which it depends on to manage its billions upon billions of investment dollars, real estate portfolios, and art collections. It is difficult to obtain all the details, but it appears that some form of nanotechnology got out of control. Surprisingly, and against its deeply ingrained reflexes of total openness and transparency, the Vatican initially tried to cover the whole thing up. Until a tabloid reporter got wind of what had happened and the whole thing became public with an article today (April 1) in an Italian tabloid that had this sensation-seeking headline splashed all over the front page: "Gay nanobots ballano Bunga-Bunga in Vaticano" - Gay nanobots dance Bunga-Bunga in the Vatican.
Apr 1st, 2011
 Brain-computer interfaces, neural probes, brain implants - they all require intensive in vivo studies on how to best combine inorganic electronics with organic neurons. Currently, most neural culture studies suffer from the fact that their cells migrate on a flat surface and are directly exposed to the culture solution, which do not reflect the microenvironment in vivo - neurons, though, can alter their behaviors dramatically in response to the environment change. Ideal cultures, therefore, should mimic the native neural microenvironment to capture the normal cell behavior. This has motivated a group of researchers to come up with this a semiconductor nanomembrane tube approach, which provides a 3D confinement that can potentially isolate the cells from the culture solution. Since these tubes are made from a semiconductor material, it means that they can be integrated with electronic functionalities such as voltage sensors.
Brain-computer interfaces, neural probes, brain implants - they all require intensive in vivo studies on how to best combine inorganic electronics with organic neurons. Currently, most neural culture studies suffer from the fact that their cells migrate on a flat surface and are directly exposed to the culture solution, which do not reflect the microenvironment in vivo - neurons, though, can alter their behaviors dramatically in response to the environment change. Ideal cultures, therefore, should mimic the native neural microenvironment to capture the normal cell behavior. This has motivated a group of researchers to come up with this a semiconductor nanomembrane tube approach, which provides a 3D confinement that can potentially isolate the cells from the culture solution. Since these tubes are made from a semiconductor material, it means that they can be integrated with electronic functionalities such as voltage sensors.
Mar 31st, 2011
 Regenerative medicine, in particular the area of tissue regeneration, is seeing a rapidly growing field of novel biomaterials that can act as bioactive scaffolds that induce tissue regeneration; that is in contrast to the more traditional concept of passively accepted implant materials. In order to present biological stimuli to the physiological environment and trigger tissue repair, optimal integration of synthetic biomaterials within the surrounding tissue is of paramount importance. In that respect, hydrogels made from biodegradable polymers are ideal candidates since they are generally biocompatible, biodegradable, and, in some cases, injectable. New research has provided firm evidence for a feasible bottom-up approach for the preparation of injectable gels by employing oppositely charged gelatin nanospheres as building blocks.
Regenerative medicine, in particular the area of tissue regeneration, is seeing a rapidly growing field of novel biomaterials that can act as bioactive scaffolds that induce tissue regeneration; that is in contrast to the more traditional concept of passively accepted implant materials. In order to present biological stimuli to the physiological environment and trigger tissue repair, optimal integration of synthetic biomaterials within the surrounding tissue is of paramount importance. In that respect, hydrogels made from biodegradable polymers are ideal candidates since they are generally biocompatible, biodegradable, and, in some cases, injectable. New research has provided firm evidence for a feasible bottom-up approach for the preparation of injectable gels by employing oppositely charged gelatin nanospheres as building blocks.
Mar 30th, 2011
 Life cycle assessment is an essential tool for ensuring the safe, responsible, and sustainable commercialization of a new technology. With missing data about the large scale impact of nanotechnology, life cycle assessments of potential nanoproducts should form an integral part of nanotechnology research at early stages of decision making as it can help in the screening of different process alternatives. Part of any meaningful results from a life cycle assessment is the total quantity of the material under investigation. Especially exposure assessments often begin with estimates based on total amounts of a material produced with the assumption that some fraction of the material in question will ultimately released to the environment. As it turns out, nobody - no research institution, no government agency, no industry association - knows even vaguely how much nanomaterials are manufactured today.
Life cycle assessment is an essential tool for ensuring the safe, responsible, and sustainable commercialization of a new technology. With missing data about the large scale impact of nanotechnology, life cycle assessments of potential nanoproducts should form an integral part of nanotechnology research at early stages of decision making as it can help in the screening of different process alternatives. Part of any meaningful results from a life cycle assessment is the total quantity of the material under investigation. Especially exposure assessments often begin with estimates based on total amounts of a material produced with the assumption that some fraction of the material in question will ultimately released to the environment. As it turns out, nobody - no research institution, no government agency, no industry association - knows even vaguely how much nanomaterials are manufactured today. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





