Showing Spotlights 225 - 232 of 544 in category All (newest first):
 On September 15-16, 2014, come explore ways in which Alternative Testing Strategies (ATS) may be combined to create a Weight of Evidence (WOE) or 'multiple models' approach to inform context-specific decisions about risk from exposure to novel nanoscale materials. The goal is to advance a common understanding of the state of the science, early lessons, current opportunities, and next steps for developing ATS for use in decision making for nanoscale materials.
On September 15-16, 2014, come explore ways in which Alternative Testing Strategies (ATS) may be combined to create a Weight of Evidence (WOE) or 'multiple models' approach to inform context-specific decisions about risk from exposure to novel nanoscale materials. The goal is to advance a common understanding of the state of the science, early lessons, current opportunities, and next steps for developing ATS for use in decision making for nanoscale materials.
Jul 9th, 2014
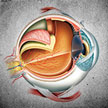 Defined as a clouding of the lens of the eye, cataracts affect more than 20 million people worldwide and accounts for 51 per cent of world blindness. In fact, this debilitating eye disease has been identified as the leading cause of blindness today. A multidisciplinary team of researchers is busy trying to understand the fundamental mechanisms of how the aggregates that cause cataracts form, and how nanotechnology may be used to prevent or at least inhibit them.
Defined as a clouding of the lens of the eye, cataracts affect more than 20 million people worldwide and accounts for 51 per cent of world blindness. In fact, this debilitating eye disease has been identified as the leading cause of blindness today. A multidisciplinary team of researchers is busy trying to understand the fundamental mechanisms of how the aggregates that cause cataracts form, and how nanotechnology may be used to prevent or at least inhibit them.
Jul 4th, 2014
 Cellular functions within living organisms are extremely complex processes and researchers have been using nanopatterned substrates to control and monitor cellular functions in order to design and fabricate nanoscale biotechnological systems. Especially stem cell research has benefitted from nanopatterned surfaces. Nevertheless, despite the intense scientific efforts to achieve precise control of stem cell fates with engineered nanopatterned substrates, reliable and cost effective control of stem cell behavior remains a challenge. Researchers have now fabricated biomimetic substrates that are similar to that of the native extracellular matrix in the epidermis which assists proliferation, differentiation, and biosynthesis of the keratinocyte (i.e. human outer skin) cells.
Cellular functions within living organisms are extremely complex processes and researchers have been using nanopatterned substrates to control and monitor cellular functions in order to design and fabricate nanoscale biotechnological systems. Especially stem cell research has benefitted from nanopatterned surfaces. Nevertheless, despite the intense scientific efforts to achieve precise control of stem cell fates with engineered nanopatterned substrates, reliable and cost effective control of stem cell behavior remains a challenge. Researchers have now fabricated biomimetic substrates that are similar to that of the native extracellular matrix in the epidermis which assists proliferation, differentiation, and biosynthesis of the keratinocyte (i.e. human outer skin) cells.
Jul 3rd, 2014
 There are a wide range of passive devices such as beads, wells and tubes that can be used to capture and confine single cells. Previous active cell grippers with moving parts have relied on electrical modalities which can be challenging to implement off-chip and in a highly parallel manner. Researchers have now, for the first time, demonstrated an untethered active microgripper that can be used to capture and contain single cells.
There are a wide range of passive devices such as beads, wells and tubes that can be used to capture and confine single cells. Previous active cell grippers with moving parts have relied on electrical modalities which can be challenging to implement off-chip and in a highly parallel manner. Researchers have now, for the first time, demonstrated an untethered active microgripper that can be used to capture and contain single cells.
Jun 25th, 2014
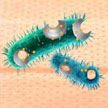 A growing body of medical nanotechnology research deals with the development of antibacterial applications, ranging from nanotechnology-based approaches for diagnosing superbugs to antimicrobial surface coatings and wound treatment with antibacterial nanomaterials. Especially silver nanomaterials have been used effectively against different bacteria, fungi and viruses but also carbon nanomaterials like nanotubes and graphene. In new work, researchers have now designed an antibacterial system combining graphene quantum dots with a low dose of a common medical reagent, hydrogen peroxide H2O2.
A growing body of medical nanotechnology research deals with the development of antibacterial applications, ranging from nanotechnology-based approaches for diagnosing superbugs to antimicrobial surface coatings and wound treatment with antibacterial nanomaterials. Especially silver nanomaterials have been used effectively against different bacteria, fungi and viruses but also carbon nanomaterials like nanotubes and graphene. In new work, researchers have now designed an antibacterial system combining graphene quantum dots with a low dose of a common medical reagent, hydrogen peroxide H2O2.
Jun 12th, 2014
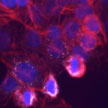 Numerous nanotoxicological studies reporting effects of nanomaterials typically address a single exposure at high dosages that are irrelevant to realistic human exposure. Recognizing that acute in vitro work had extremely low correlation to in vivo nanomaterial studies, coupled with the recognition that the unique characteristics that distinguish nanomaterials vary as a function of time, researchers sought to identify a model that would allow for the evaluation of nanomaterial behavior over a 3-month period, but be carried out in an in vitro model.
Numerous nanotoxicological studies reporting effects of nanomaterials typically address a single exposure at high dosages that are irrelevant to realistic human exposure. Recognizing that acute in vitro work had extremely low correlation to in vivo nanomaterial studies, coupled with the recognition that the unique characteristics that distinguish nanomaterials vary as a function of time, researchers sought to identify a model that would allow for the evaluation of nanomaterial behavior over a 3-month period, but be carried out in an in vitro model.
Jun 4th, 2014
 The formation of protein corona is a continuous state of flux in which many proteins compete to bind to the nanoparticle surface, each with their own characteristics such as concentration, structure and solubility determining their final affinity to the nanoparticle surface. This is the reason why biological responses to nanoparticles are strongly dependent to the type and amount of associated proteins in the composition of the protein corona. The protein corona determines the biological fate of nanoparticles and physiological responses. New research findings now show that the plasma protein alterations associated with different diseases, medical conditions, or even lifestyle, can affect the protein composition and content of the hard corona composition.
The formation of protein corona is a continuous state of flux in which many proteins compete to bind to the nanoparticle surface, each with their own characteristics such as concentration, structure and solubility determining their final affinity to the nanoparticle surface. This is the reason why biological responses to nanoparticles are strongly dependent to the type and amount of associated proteins in the composition of the protein corona. The protein corona determines the biological fate of nanoparticles and physiological responses. New research findings now show that the plasma protein alterations associated with different diseases, medical conditions, or even lifestyle, can affect the protein composition and content of the hard corona composition.
Jun 3rd, 2014
 Nanobioreactors are emerging as advanced bio-devices, which fuse the advantages of nanomaterials with those of nanobiotechnology. Due to their ultimately small size, high surface area and simulation capacity, they are set to become to be a versatile tool to fabricate ultra-sensitive and selective novel nanobio-devices, which offer us new platform to tackle key energy, medical and environmental issues. Now, a novel two-dimensional bioreactor offers a simple and effective way to overcome many limitations that have been faced by previous designs.
Nanobioreactors are emerging as advanced bio-devices, which fuse the advantages of nanomaterials with those of nanobiotechnology. Due to their ultimately small size, high surface area and simulation capacity, they are set to become to be a versatile tool to fabricate ultra-sensitive and selective novel nanobio-devices, which offer us new platform to tackle key energy, medical and environmental issues. Now, a novel two-dimensional bioreactor offers a simple and effective way to overcome many limitations that have been faced by previous designs.
May 21st, 2014
 On September 15-16, 2014, come explore ways in which Alternative Testing Strategies (ATS) may be combined to create a Weight of Evidence (WOE) or 'multiple models' approach to inform context-specific decisions about risk from exposure to novel nanoscale materials. The goal is to advance a common understanding of the state of the science, early lessons, current opportunities, and next steps for developing ATS for use in decision making for nanoscale materials.
On September 15-16, 2014, come explore ways in which Alternative Testing Strategies (ATS) may be combined to create a Weight of Evidence (WOE) or 'multiple models' approach to inform context-specific decisions about risk from exposure to novel nanoscale materials. The goal is to advance a common understanding of the state of the science, early lessons, current opportunities, and next steps for developing ATS for use in decision making for nanoscale materials.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





