Showing Spotlights 17 - 24 of 137 in category All (newest first):
 As nanotechnologies are beginning to empower our lives in so many ways, understanding the environmental health and safety aspect of nanotechnology has become a crucial issue. The lack of information on the impact of engineered nanomaterials on organisms and the environment motivates researchers all over the world to strive for a better understanding of the implications of nanotechnology applications. Researchers have now provided a mechanistic understanding on how nanomaterials affect zebrafish embryos development and specifically answers the question on what causes the embryos to fail hatching at due time.
As nanotechnologies are beginning to empower our lives in so many ways, understanding the environmental health and safety aspect of nanotechnology has become a crucial issue. The lack of information on the impact of engineered nanomaterials on organisms and the environment motivates researchers all over the world to strive for a better understanding of the implications of nanotechnology applications. Researchers have now provided a mechanistic understanding on how nanomaterials affect zebrafish embryos development and specifically answers the question on what causes the embryos to fail hatching at due time.
Dec 20th, 2012
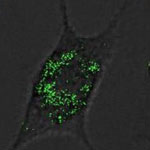 Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae. Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. Researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles.
Silver nanoparticles are among the most commercialized nanomaterials due to their use as antibacterial agent in consumer products and surface coatings. Several reports have shown that silver ions from silver compounds or those that develop from nanosilver particles through contact with water are highly toxic to microorganisms such as bacteria, fungi and algae. Contributing to an incomplete and confusing picture, in the literature, silver nanoparticles are claimed as nontoxic or toxic depending on their size, concentration and surface functionalization. Researchers have now reported on possible adverse effects of the silver nanoparticles upon their release into the environment and provided novel insights into the possible applications of silver nanoparticles in nanomedicine by discussing their p53 gene related cell death profiles.
Sep 12th, 2012
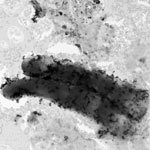 Aquatic ecotoxicity test methods, which are routinely applied to testing of nanomaterials, were originally developed for water soluble chemicals. Nanomaterials are fundamentally different from many 'conventional' chemicals as they often have limited or no solubility at all and are potentially released to the environment in a particulate form (e.g. carbon nanotubes). Only limited nano-specific guidance on ecotoxicity testing is currently available, for instance existing OECD ecotoxicity test guidelines to nanomaterials. These guidelines exhibit a number of specific shortcomings mainly related to characterization, exposure preparation, quantification and monitoring concentrations, and dose-metrics. A new paper from scientists in Denmark contributes to the progress in algae testing of nanomaterials. This work also aids in the development of additional guidance as it adds to the understanding of pros and cons of different techniques for biomass quantification.
Aquatic ecotoxicity test methods, which are routinely applied to testing of nanomaterials, were originally developed for water soluble chemicals. Nanomaterials are fundamentally different from many 'conventional' chemicals as they often have limited or no solubility at all and are potentially released to the environment in a particulate form (e.g. carbon nanotubes). Only limited nano-specific guidance on ecotoxicity testing is currently available, for instance existing OECD ecotoxicity test guidelines to nanomaterials. These guidelines exhibit a number of specific shortcomings mainly related to characterization, exposure preparation, quantification and monitoring concentrations, and dose-metrics. A new paper from scientists in Denmark contributes to the progress in algae testing of nanomaterials. This work also aids in the development of additional guidance as it adds to the understanding of pros and cons of different techniques for biomass quantification.
Aug 31st, 2012
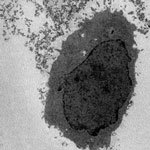 When a nanomaterial enters a physiological environment, it often adsorbs suspended biomolecules including proteins, lipids, small molecules, saccharides, and nucleic acids. These biomolecules define a new interface between the nanomaterial and its surroundings that mediates cellular association and response. In a recent article appearing in ACS Nano, Drs. Anna Salvati, Kenneth Dawson, and their colleagues at the University College in Dublin, Ireland, show that if nanoparticles are exposed directly to cells in the absence of suspended biomolecules, the nanoparticles will extract biomolecules from cells themselves. These findings suggest that in vitro toxicological or cell uptake studies performed in the absence of added serum may not be relevant in vivo.
When a nanomaterial enters a physiological environment, it often adsorbs suspended biomolecules including proteins, lipids, small molecules, saccharides, and nucleic acids. These biomolecules define a new interface between the nanomaterial and its surroundings that mediates cellular association and response. In a recent article appearing in ACS Nano, Drs. Anna Salvati, Kenneth Dawson, and their colleagues at the University College in Dublin, Ireland, show that if nanoparticles are exposed directly to cells in the absence of suspended biomolecules, the nanoparticles will extract biomolecules from cells themselves. These findings suggest that in vitro toxicological or cell uptake studies performed in the absence of added serum may not be relevant in vivo.
Jul 24th, 2012
 There is currently no clear evidence that engineered nanoparticles pose a significant threat to the environment. Nonetheless, major gaps in our knowledge exist. The present dossier illustrates the problems in the field of environmental analytics, presents the current state of knowledge on the fate and behavior of ENPs in various environmental compartments and provides an overview of the preliminary results from ecotoxicological research and from model calculations of exposure assessments. At present, ecotoxicological research focuses primarily on controlled laboratory studies involving cell cultures or model organisms. One of the major critiques here is the use of unrealistically high dosages1 Overall, no definitive conclusions can be drawn on whether environmental damage can be expected or not.
There is currently no clear evidence that engineered nanoparticles pose a significant threat to the environment. Nonetheless, major gaps in our knowledge exist. The present dossier illustrates the problems in the field of environmental analytics, presents the current state of knowledge on the fate and behavior of ENPs in various environmental compartments and provides an overview of the preliminary results from ecotoxicological research and from model calculations of exposure assessments. At present, ecotoxicological research focuses primarily on controlled laboratory studies involving cell cultures or model organisms. One of the major critiques here is the use of unrealistically high dosages1 Overall, no definitive conclusions can be drawn on whether environmental damage can be expected or not.
Jul 12th, 2012
 Understanding the health and environmental impact of nanomaterials is vital to the sustainable and responsible development of nanotechnology. Currently, small animal experiments are the 'gold standard' for nanomaterial toxicity testing. However, a detailed understanding often requires dozens of animals and can take many months to complete. Dr. Andre Nel and his coworkers at the California NanoSystems Institute (CNSI) and the University of California Los Angeles (UCLA) are taking a fundamentally different approach to nanomaterial toxicity testing. Nel believes that, under the right circumstances, resource-intensive animal experiments can be replaced or adjusted with comparatively simple in vitro assays. This article explores his approach and its implications for nanomaterial design and development.
Understanding the health and environmental impact of nanomaterials is vital to the sustainable and responsible development of nanotechnology. Currently, small animal experiments are the 'gold standard' for nanomaterial toxicity testing. However, a detailed understanding often requires dozens of animals and can take many months to complete. Dr. Andre Nel and his coworkers at the California NanoSystems Institute (CNSI) and the University of California Los Angeles (UCLA) are taking a fundamentally different approach to nanomaterial toxicity testing. Nel believes that, under the right circumstances, resource-intensive animal experiments can be replaced or adjusted with comparatively simple in vitro assays. This article explores his approach and its implications for nanomaterial design and development.
Jul 9th, 2012
 A commentary by Steffen Foss Hansen and Anders Baun in this week's Nature Nanotechnology pointedly asks "when will governments and regulatory agencies stop asking for more reports and reviews, and start taking regulatory action?" The two scientists take issue with yet another scientific opinion on nanosilver that has been requested by the European Commission in late 2011: "SCENIHR - Request for a scientific opinion on Nanosilver: safety, health and environmental effects and role in antimicrobial resistance". Specifically, the EC wants SCENIHR to answer four questions under the general heading of 'Nanosilver: safety, health and environmental effects, and role in antimicrobial resistance'. These questions, however, have already been addressed by no less than 18 review articles in scientific journals.
A commentary by Steffen Foss Hansen and Anders Baun in this week's Nature Nanotechnology pointedly asks "when will governments and regulatory agencies stop asking for more reports and reviews, and start taking regulatory action?" The two scientists take issue with yet another scientific opinion on nanosilver that has been requested by the European Commission in late 2011: "SCENIHR - Request for a scientific opinion on Nanosilver: safety, health and environmental effects and role in antimicrobial resistance". Specifically, the EC wants SCENIHR to answer four questions under the general heading of 'Nanosilver: safety, health and environmental effects, and role in antimicrobial resistance'. These questions, however, have already been addressed by no less than 18 review articles in scientific journals.
Jul 4th, 2012
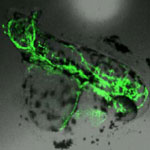 A researcher team at the California NanoSystems Institute and the University of California, Los Angeles, have found that the crystal structure of silver nanoparticles is an important determinant of their toxicity to aquatic life. The study comes amid growing concern that the proliferation of nanotechnology will result in the inadvertent release of nanomaterials into the environment. Release of silver nanoparticles is a particular concern given that over 30% of the roughly 800 nano-enabled products currently on the market contain silver nanoparticles. Prior to this study, the environmental toxicity of silver nanoparticles was thought to result mainly from release of silver ions. Nel and his team have now found that defects on the surface of silver nanoparticles can catalyze the production of reactive oxygen species that damage biomolecules within cells. This new mechanism of toxicity may be applicable to non-aquatic organisms, and extend beyond silver nanoparticles.
A researcher team at the California NanoSystems Institute and the University of California, Los Angeles, have found that the crystal structure of silver nanoparticles is an important determinant of their toxicity to aquatic life. The study comes amid growing concern that the proliferation of nanotechnology will result in the inadvertent release of nanomaterials into the environment. Release of silver nanoparticles is a particular concern given that over 30% of the roughly 800 nano-enabled products currently on the market contain silver nanoparticles. Prior to this study, the environmental toxicity of silver nanoparticles was thought to result mainly from release of silver ions. Nel and his team have now found that defects on the surface of silver nanoparticles can catalyze the production of reactive oxygen species that damage biomolecules within cells. This new mechanism of toxicity may be applicable to non-aquatic organisms, and extend beyond silver nanoparticles.
Jun 20th, 2012
 As nanotechnologies are beginning to empower our lives in so many ways, understanding the environmental health and safety aspect of nanotechnology has become a crucial issue. The lack of information on the impact of engineered nanomaterials on organisms and the environment motivates researchers all over the world to strive for a better understanding of the implications of nanotechnology applications. Researchers have now provided a mechanistic understanding on how nanomaterials affect zebrafish embryos development and specifically answers the question on what causes the embryos to fail hatching at due time.
As nanotechnologies are beginning to empower our lives in so many ways, understanding the environmental health and safety aspect of nanotechnology has become a crucial issue. The lack of information on the impact of engineered nanomaterials on organisms and the environment motivates researchers all over the world to strive for a better understanding of the implications of nanotechnology applications. Researchers have now provided a mechanistic understanding on how nanomaterials affect zebrafish embryos development and specifically answers the question on what causes the embryos to fail hatching at due time. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





