Showing Spotlights 249 - 256 of 624 in category All (newest first):
 3D-printing processes are engineered to use material more efficiently, give designs more flexibility and produce objects more precisely. These 3D printing techniques are reaching a stage where desired products and structures can be made independent of the complexity of their shapes. Applying 3D printing concepts to nanotechnology could bring similar advantages to nanofabrication - speed, less waste, economic viability - than it is expected to bring to manufacturing technologies. here we show examples of current research into 3D printing in nanotechnology.
3D-printing processes are engineered to use material more efficiently, give designs more flexibility and produce objects more precisely. These 3D printing techniques are reaching a stage where desired products and structures can be made independent of the complexity of their shapes. Applying 3D printing concepts to nanotechnology could bring similar advantages to nanofabrication - speed, less waste, economic viability - than it is expected to bring to manufacturing technologies. here we show examples of current research into 3D printing in nanotechnology.
Sep 26th, 2014
 Electrochromic devices are some of the most attractive candidates for paper-like displays, so called electronic paper, which will be the next generation display. Researchers have now demonstrated solid state flexible polymer based electrochromic devices are fabricated continuously by stacking layers in one direction. This novel bottom-up approach with no need for a lamination step enables fully printed and 2D patterned organic electrochromics.
Electrochromic devices are some of the most attractive candidates for paper-like displays, so called electronic paper, which will be the next generation display. Researchers have now demonstrated solid state flexible polymer based electrochromic devices are fabricated continuously by stacking layers in one direction. This novel bottom-up approach with no need for a lamination step enables fully printed and 2D patterned organic electrochromics.
Sep 17th, 2014
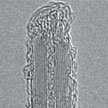 So far, it has been generally accepted knowledge that boron nitride nanotubes (BNNTs) are highly inert to oxidative treatments and can only be covalently modified by highly reactive species. By contrast, oxidation of carbon nanotubes has been proven very convenient and fundamentally important to modify the nanotube structure and morphology via controlled corrosive effects. Now, researchers have discovered a convenient method to disperse and chemically modify the morphology of BNNTs by sonication in aqueous ammonia solutions.
So far, it has been generally accepted knowledge that boron nitride nanotubes (BNNTs) are highly inert to oxidative treatments and can only be covalently modified by highly reactive species. By contrast, oxidation of carbon nanotubes has been proven very convenient and fundamentally important to modify the nanotube structure and morphology via controlled corrosive effects. Now, researchers have discovered a convenient method to disperse and chemically modify the morphology of BNNTs by sonication in aqueous ammonia solutions.
Aug 13th, 2014
 Researchers have now shown that, by varying the shape of magnetite nanoparticles, they can control the nature of the self-assembled structures as the nanoparticles assemble. This new work provides guidelines for the design of new self-assembled materials. Self-assembly of nanoparticles driven by competing forces can result in truly unique structures, the diversity and complexity of which could be particularly striking if the building blocks were simultaneously coupled by short- and long-range forces of different symmetries.
Researchers have now shown that, by varying the shape of magnetite nanoparticles, they can control the nature of the self-assembled structures as the nanoparticles assemble. This new work provides guidelines for the design of new self-assembled materials. Self-assembly of nanoparticles driven by competing forces can result in truly unique structures, the diversity and complexity of which could be particularly striking if the building blocks were simultaneously coupled by short- and long-range forces of different symmetries.
Aug 4th, 2014
 Conjugated polymer based organic photovoltaic (OPV) devices have been the subject of increasing research interest over the past years due to their potential of being light weight, mechanically flexible, semitransparent. To increase the efficiency of OPV, it is necessary to achieve a precisely controlled donor-acceptor phase separation within the short exciton diffusion length without dead ends, as well as a high hole mobility within the polymer. Now, researchers have demonstrated the effects of nanostructure geometry on the nanoimprint induced P3HT chain alignment and the performance of nanoimprinted photovoltaic devices.
Conjugated polymer based organic photovoltaic (OPV) devices have been the subject of increasing research interest over the past years due to their potential of being light weight, mechanically flexible, semitransparent. To increase the efficiency of OPV, it is necessary to achieve a precisely controlled donor-acceptor phase separation within the short exciton diffusion length without dead ends, as well as a high hole mobility within the polymer. Now, researchers have demonstrated the effects of nanostructure geometry on the nanoimprint induced P3HT chain alignment and the performance of nanoimprinted photovoltaic devices.
Jul 22nd, 2014
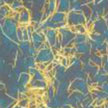 Many nanofabrication techniques depend on creating a structure on one substrate and then transferring it via various processes onto another, desired, substrate. Often, these methods are not generally applicable as they suffer from the process-specific drawbacks, such as for instance intolerance of transferred nanostructures to chemical etchant, and the harsh thermal environment needed. A novel universal and rapid method allows transferring nanostructures with various dimensions onto diverse substrates with high fidelity.
Many nanofabrication techniques depend on creating a structure on one substrate and then transferring it via various processes onto another, desired, substrate. Often, these methods are not generally applicable as they suffer from the process-specific drawbacks, such as for instance intolerance of transferred nanostructures to chemical etchant, and the harsh thermal environment needed. A novel universal and rapid method allows transferring nanostructures with various dimensions onto diverse substrates with high fidelity.
Jul 8th, 2014
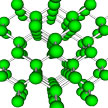 Thermoelectric materials hold great promise for turning waste heat back into useful power and are touted for use in hybrid cars, new and efficient refrigerators, and other cooling or heating applications. But they have one big drawback: they are very inefficient. Since thermoelectric devices work by maintaining a temperature difference between their different sides, it is important that the used materials have low conductivity, i.e. are good thermal insulators.
Thermoelectric materials hold great promise for turning waste heat back into useful power and are touted for use in hybrid cars, new and efficient refrigerators, and other cooling or heating applications. But they have one big drawback: they are very inefficient. Since thermoelectric devices work by maintaining a temperature difference between their different sides, it is important that the used materials have low conductivity, i.e. are good thermal insulators.
Jul 7th, 2014
 The microstructures of carbon nanotube assemblies determine their properties, for example, highly graphitized CNTs exhibit excellent mechanical and electrical properties; while CNTs with defects and poor crystallinity are beneficial for research on field emission property and hydrogen storage capacity. Therefore, it is of vital importance to control the CNT microstructures effectively for desired applications. A new technique can solve a problem of three-dimensional orientation control of CNTs in microscopic scale.
The microstructures of carbon nanotube assemblies determine their properties, for example, highly graphitized CNTs exhibit excellent mechanical and electrical properties; while CNTs with defects and poor crystallinity are beneficial for research on field emission property and hydrogen storage capacity. Therefore, it is of vital importance to control the CNT microstructures effectively for desired applications. A new technique can solve a problem of three-dimensional orientation control of CNTs in microscopic scale.
Jun 30th, 2014
 3D-printing processes are engineered to use material more efficiently, give designs more flexibility and produce objects more precisely. These 3D printing techniques are reaching a stage where desired products and structures can be made independent of the complexity of their shapes. Applying 3D printing concepts to nanotechnology could bring similar advantages to nanofabrication - speed, less waste, economic viability - than it is expected to bring to manufacturing technologies. here we show examples of current research into 3D printing in nanotechnology.
3D-printing processes are engineered to use material more efficiently, give designs more flexibility and produce objects more precisely. These 3D printing techniques are reaching a stage where desired products and structures can be made independent of the complexity of their shapes. Applying 3D printing concepts to nanotechnology could bring similar advantages to nanofabrication - speed, less waste, economic viability - than it is expected to bring to manufacturing technologies. here we show examples of current research into 3D printing in nanotechnology.

 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





