Showing Spotlights 281 - 288 of 624 in category All (newest first):
 One of the most restricting parameters in nanofabrication is the difficulty involved with controllably patterning materials at precise locations in a repeatable manner over relatively large areas. A novel microelectromechanical system (MEMS)-based mask writer has now been developed by a team of researchers. The device allows to directly write structures at the nanoscale without the need to use photoresist, lift-off techniques or other complex and expensive approaches. The technique uses a MEMS plate with apertures drilled into it and a shutter so that one can, in effect, spray paint with atoms.
One of the most restricting parameters in nanofabrication is the difficulty involved with controllably patterning materials at precise locations in a repeatable manner over relatively large areas. A novel microelectromechanical system (MEMS)-based mask writer has now been developed by a team of researchers. The device allows to directly write structures at the nanoscale without the need to use photoresist, lift-off techniques or other complex and expensive approaches. The technique uses a MEMS plate with apertures drilled into it and a shutter so that one can, in effect, spray paint with atoms.
Jul 3rd, 2013
 While researchers are working on developing more cost-effective nanolithographic tools such as for instance superlens lithography, one of the key problems with nanofabrication is how to generate ever-challenging patterns with high resolution - especially for 3D nanostructures - and at the same time substantially reduce the cost of the process. A novel nanolithography method is based on light scattering from nanoparticles, which can generate 3D hollow-core structures that resembles 'nano-volcanoes'. Different from traditional lithography methods that are typically based on complex systems, this process relies solely on the light interaction with a single nanoparticle. No masks and external optics are needed in this approach, and light is manipulated into the desired optical pattern solely by the colloids.
While researchers are working on developing more cost-effective nanolithographic tools such as for instance superlens lithography, one of the key problems with nanofabrication is how to generate ever-challenging patterns with high resolution - especially for 3D nanostructures - and at the same time substantially reduce the cost of the process. A novel nanolithography method is based on light scattering from nanoparticles, which can generate 3D hollow-core structures that resembles 'nano-volcanoes'. Different from traditional lithography methods that are typically based on complex systems, this process relies solely on the light interaction with a single nanoparticle. No masks and external optics are needed in this approach, and light is manipulated into the desired optical pattern solely by the colloids.
Jun 24th, 2013
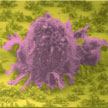 Fractals are structures built up from repeated sizings of a simple shape to make a complex one. A fractal is a geometric structure that can repeat itself towards infinity. Zooming in on a fragment of it, the original structure becomes visible again. In biological systems, fractal structures can be found everywhere - bronchial trees, vasculature, and nerve cells. These amazing structures can provide a specific interfacial contact mode that is highly efficient for absorbing sunlight, transporting nutrition, exchanging oxygen and carbon dioxide, and signal transduction. Researchers have now demonstrated the fabrication of programmable fractal gold nanostructured interfaces and their outstanding specific recognition of rare cancer cells from whole blood samples along with their effective release capability.
Fractals are structures built up from repeated sizings of a simple shape to make a complex one. A fractal is a geometric structure that can repeat itself towards infinity. Zooming in on a fragment of it, the original structure becomes visible again. In biological systems, fractal structures can be found everywhere - bronchial trees, vasculature, and nerve cells. These amazing structures can provide a specific interfacial contact mode that is highly efficient for absorbing sunlight, transporting nutrition, exchanging oxygen and carbon dioxide, and signal transduction. Researchers have now demonstrated the fabrication of programmable fractal gold nanostructured interfaces and their outstanding specific recognition of rare cancer cells from whole blood samples along with their effective release capability.
Jun 13th, 2013
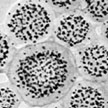 One major challenge in contemporary science is to accomplish with synthetic building blocks what nature does so well, that is, creating complex and functional structures through multiple levels of assembly of biomolecules. Bottom-up engineering of nanostructures that assemble themselves from polymer molecules are bound to become useful tools in chemistry. To that end, researchers are using block copolymer based micellar architectures to form hierarchical superstructures with defined shape and geometry. Researchers have now demonstrate that nanoparticles tethered with block copolymers resemble micelles that can assemble into well-ordered higher level mesostructures.
One major challenge in contemporary science is to accomplish with synthetic building blocks what nature does so well, that is, creating complex and functional structures through multiple levels of assembly of biomolecules. Bottom-up engineering of nanostructures that assemble themselves from polymer molecules are bound to become useful tools in chemistry. To that end, researchers are using block copolymer based micellar architectures to form hierarchical superstructures with defined shape and geometry. Researchers have now demonstrate that nanoparticles tethered with block copolymers resemble micelles that can assemble into well-ordered higher level mesostructures.
Jun 12th, 2013
 Atomically precise manufacturing (APM) can be understood through physics, engineering design principles, proof-of-concept examples, computational modeling, and parallels with familiar technologies. APM is a prospective production technology based on guiding the motion of reactive molecules to build progressively larger components and systems. Bottom-up atomic precision can enable production with unprecedented scope (in terms of product materials, components, systems, and performance), while fundamental mechanical scaling laws can enable unprecedented productivity.
Atomically precise manufacturing (APM) can be understood through physics, engineering design principles, proof-of-concept examples, computational modeling, and parallels with familiar technologies. APM is a prospective production technology based on guiding the motion of reactive molecules to build progressively larger components and systems. Bottom-up atomic precision can enable production with unprecedented scope (in terms of product materials, components, systems, and performance), while fundamental mechanical scaling laws can enable unprecedented productivity.
Jun 11th, 2013
 Researchers have come up with various electrode materials to improve the performance of supercapacitors, focussing mostly on porous carbon due to its high surface areas, tunable structures, good conductivities, and low cost. Graphene and carbon nanotubes show great potential but are costly. Researchers in Canada have now reported the successful hydrothermal-based synthesis of two-dimensional, yet interconnected, carbon nanosheets with superior electrochemical storage properties comparable to those of state-of-the-art graphene-based electrodes.
Researchers have come up with various electrode materials to improve the performance of supercapacitors, focussing mostly on porous carbon due to its high surface areas, tunable structures, good conductivities, and low cost. Graphene and carbon nanotubes show great potential but are costly. Researchers in Canada have now reported the successful hydrothermal-based synthesis of two-dimensional, yet interconnected, carbon nanosheets with superior electrochemical storage properties comparable to those of state-of-the-art graphene-based electrodes.
May 15th, 2013
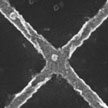 Ferromagnetic materials exhibit the so-called anomalous Hall effect (AHE), whereby the electrons flowing through the material experience a lateral force pushing them to one side as a result of the material's intrinsic magnetization. Although the AHE has been used in the field on nanotechnology to measure the magnetic behavior of nanoparticles (with sizes larger than 50 nm), nobody so far had tried to separate the signals of the individual particles. Researchers in Germany have now developed a simple technique which allows to measure the magnetic response of single ferromagnetic nanoparticles down to a radius of about 3.3 nm.
Ferromagnetic materials exhibit the so-called anomalous Hall effect (AHE), whereby the electrons flowing through the material experience a lateral force pushing them to one side as a result of the material's intrinsic magnetization. Although the AHE has been used in the field on nanotechnology to measure the magnetic behavior of nanoparticles (with sizes larger than 50 nm), nobody so far had tried to separate the signals of the individual particles. Researchers in Germany have now developed a simple technique which allows to measure the magnetic response of single ferromagnetic nanoparticles down to a radius of about 3.3 nm.
May 14th, 2013
 The boundaries of electron beam lithography (EBL), the workhorse of current nanofabrication processes, is constantly being pushed further down into the single nanometer range by researchers' efforts to overcome the various limitations of EBL resolution - spot size, electron scattering, secondary-electron range, resist development, and mechanical stability of the resist. A team of scientists has now achieved the EBL fabrication of 2 nm feature size and 10 nm periodic dense structures, which are the highest resolution patterns ever achieved with common resists. The minimum feature size, 2 nm, is composed of roughly 10 atoms wide, and with just a few atoms of standard deviation.
The boundaries of electron beam lithography (EBL), the workhorse of current nanofabrication processes, is constantly being pushed further down into the single nanometer range by researchers' efforts to overcome the various limitations of EBL resolution - spot size, electron scattering, secondary-electron range, resist development, and mechanical stability of the resist. A team of scientists has now achieved the EBL fabrication of 2 nm feature size and 10 nm periodic dense structures, which are the highest resolution patterns ever achieved with common resists. The minimum feature size, 2 nm, is composed of roughly 10 atoms wide, and with just a few atoms of standard deviation.
Mar 26th, 2013
 One of the most restricting parameters in nanofabrication is the difficulty involved with controllably patterning materials at precise locations in a repeatable manner over relatively large areas. A novel microelectromechanical system (MEMS)-based mask writer has now been developed by a team of researchers. The device allows to directly write structures at the nanoscale without the need to use photoresist, lift-off techniques or other complex and expensive approaches. The technique uses a MEMS plate with apertures drilled into it and a shutter so that one can, in effect, spray paint with atoms.
One of the most restricting parameters in nanofabrication is the difficulty involved with controllably patterning materials at precise locations in a repeatable manner over relatively large areas. A novel microelectromechanical system (MEMS)-based mask writer has now been developed by a team of researchers. The device allows to directly write structures at the nanoscale without the need to use photoresist, lift-off techniques or other complex and expensive approaches. The technique uses a MEMS plate with apertures drilled into it and a shutter so that one can, in effect, spray paint with atoms.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





