Showing Spotlights 297 - 304 of 624 in category All (newest first):
 In order to fabricate stimuli-responsive materials, researchers have shown a lot of interest in asymmetric materials such as modulated gels which consist of a controlled layer that is responsive to an environmental stimuli and a nonresponsive substrate layer. And while much effort has gone into creating free-standing films through layer-by-layer (LbL) assembly, relatively little attention has been paid to the asymmetric properties or functionalization of the two surfaces of such free-standing layer-by-layer films. In new work, researchers have now reported the fabrication of asymmetric free-standing layer-by-layer film with asymmetric wettability - one surface is superhydrophobic and the other one is hydrophilic. The superhydrophobic side is water-repellent while the hydrophilic side can absorb/desorb water easily.
In order to fabricate stimuli-responsive materials, researchers have shown a lot of interest in asymmetric materials such as modulated gels which consist of a controlled layer that is responsive to an environmental stimuli and a nonresponsive substrate layer. And while much effort has gone into creating free-standing films through layer-by-layer (LbL) assembly, relatively little attention has been paid to the asymmetric properties or functionalization of the two surfaces of such free-standing layer-by-layer films. In new work, researchers have now reported the fabrication of asymmetric free-standing layer-by-layer film with asymmetric wettability - one surface is superhydrophobic and the other one is hydrophilic. The superhydrophobic side is water-repellent while the hydrophilic side can absorb/desorb water easily.
Oct 29th, 2012
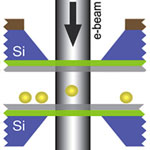 The idea of using laser light to trap or levitate small particles goes back to the pioneering work by Arthur Ashkin of Bell Laboratories in the 1970s and 1980s. Ashkin found that radiation pressure - the ability of light to exert pressure to move small objects - could be harnessed to constrain small particles. This discovery has since formed the basis for scientific advances such as the development of optical tweezers, which are frequently used to control the motion of small biological objects. However, optical trapping of nanoparticles remains a challenging task because the forces are often too small when the sizes of the objects are reduced to the nanometer scale. New findings from scientists at Lawrence Berkeley National Laboratory and National University of Singapore fill a gap and also open the door to new discoveries by demonstrating trapping and manipulating nanometer size particles using an electron beam instead of optical forces. It could also lead to new force spectroscopy where nanostructures can be assembled one nanoparticle at a time.
The idea of using laser light to trap or levitate small particles goes back to the pioneering work by Arthur Ashkin of Bell Laboratories in the 1970s and 1980s. Ashkin found that radiation pressure - the ability of light to exert pressure to move small objects - could be harnessed to constrain small particles. This discovery has since formed the basis for scientific advances such as the development of optical tweezers, which are frequently used to control the motion of small biological objects. However, optical trapping of nanoparticles remains a challenging task because the forces are often too small when the sizes of the objects are reduced to the nanometer scale. New findings from scientists at Lawrence Berkeley National Laboratory and National University of Singapore fill a gap and also open the door to new discoveries by demonstrating trapping and manipulating nanometer size particles using an electron beam instead of optical forces. It could also lead to new force spectroscopy where nanostructures can be assembled one nanoparticle at a time.
Oct 16th, 2012
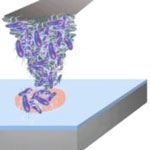 Dip-Pen Nanolithography (DPN) is a scanning probe lithography technique in which the tip of an atomic force microscope is used to 'write' molecules directly onto a substrate, allowing nanostructured surface patterning on scales of under 100 nm. Since the driving force of DPN for transporting materials is molecular diffusion through the sub-micrometer sized water meniscus formed between the AFM tip and the surface, large-sized ink materials are not efficiently transported through this water meniscus. For this reason, bacterial cells (1-2 micrometer or larger in length) patterning with DPN technique is often considered to be impossible. Overcoming this limitation, researchers have developed a 'stamp-on' DPN method that uses a previously developed hydrogel-coated tip and carrier agents to generate micrometer-sized bacterial cells.
Dip-Pen Nanolithography (DPN) is a scanning probe lithography technique in which the tip of an atomic force microscope is used to 'write' molecules directly onto a substrate, allowing nanostructured surface patterning on scales of under 100 nm. Since the driving force of DPN for transporting materials is molecular diffusion through the sub-micrometer sized water meniscus formed between the AFM tip and the surface, large-sized ink materials are not efficiently transported through this water meniscus. For this reason, bacterial cells (1-2 micrometer or larger in length) patterning with DPN technique is often considered to be impossible. Overcoming this limitation, researchers have developed a 'stamp-on' DPN method that uses a previously developed hydrogel-coated tip and carrier agents to generate micrometer-sized bacterial cells.
Oct 11th, 2012
 One way to use engineered nanoparticles in the real world is in thin films. Such nanoparticulate films are thin layers, sometimes only a few nanometers thick, of composite materials that contain nanoparticles. These new materials have a wide range of applications in drug delivery, nanoelectronics, magnetic storage devices, sensors, or optical coating. However, most processes used to fabricate thin nanocomposite films with high nanoparticle fillings suffer from random nanoparticle agglomeration causing formation of irregularly shaped nanostructured features within the composite. Another complication arises from cracks that develop during the fabrication of the films. Researchers have now described a simple method for fabricating thick, crack-free silica nanoparticle films by subsequent deposition of thin, crack-free silica nanoparticle multilayers.
One way to use engineered nanoparticles in the real world is in thin films. Such nanoparticulate films are thin layers, sometimes only a few nanometers thick, of composite materials that contain nanoparticles. These new materials have a wide range of applications in drug delivery, nanoelectronics, magnetic storage devices, sensors, or optical coating. However, most processes used to fabricate thin nanocomposite films with high nanoparticle fillings suffer from random nanoparticle agglomeration causing formation of irregularly shaped nanostructured features within the composite. Another complication arises from cracks that develop during the fabrication of the films. Researchers have now described a simple method for fabricating thick, crack-free silica nanoparticle films by subsequent deposition of thin, crack-free silica nanoparticle multilayers.
Sep 26th, 2012
 The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
The remarkable properties of some natural materials have motivated many researchers to synthesize biomimetic nanocomposites that attempt to reproduce Nature's achievements and to understand the toughening and deformation mechanisms of natural nanocomposite materials. One of the best examples is nacre, the pearly internal layer of many mollusc shells. CaCO3 has long been considered to be a genuine chemical component to approximate nacre. However, only supported polycrystalline CaCO3 (calcite) films have been reported so far. With rapid advancement of nanotechnology, it would be highly desirable to synthesize freestanding CaCO3 tablet building blocks in large quantities, ideally identical or similar to those in natural nacre, using solution-based methods. Now, for the first time, scientists have devised a facile chemical method to synthesize single-crystalline CaCO3 nanotablets in large quantities and provided genuine primary building blocks for the fabrication of nacreous inorganic-organic hybrids.
Sep 20th, 2012
 Multiphoton lithography (MPL) is a microfabrication technique used to create three-dimensional microscale objects with complex geometrical arrangements. Of the various chemistries used to produce solid forms in MPL, protein photocrosslinking has been of particular value in biological applications. In new work, researchers have now described a strategy for creating a nearly unlimited range of microforms from crosslinked protein, including structures composed of multiple proteins. They also describe MPL microfabrication of complex unconstrained objects using high-viscosity protein-based reagents. To avoid drift during fabrication of microforms that are not in integral contact with a surface, the team developed a methodology for producing high-viscosity protein-based reagents, or "protogels". These materials allow the fabrication of protein-based objects that retain rotational and translational degrees of freedom.
Multiphoton lithography (MPL) is a microfabrication technique used to create three-dimensional microscale objects with complex geometrical arrangements. Of the various chemistries used to produce solid forms in MPL, protein photocrosslinking has been of particular value in biological applications. In new work, researchers have now described a strategy for creating a nearly unlimited range of microforms from crosslinked protein, including structures composed of multiple proteins. They also describe MPL microfabrication of complex unconstrained objects using high-viscosity protein-based reagents. To avoid drift during fabrication of microforms that are not in integral contact with a surface, the team developed a methodology for producing high-viscosity protein-based reagents, or "protogels". These materials allow the fabrication of protein-based objects that retain rotational and translational degrees of freedom.
Sep 6th, 2012
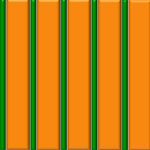 Lithography based on block copolymer self-assembly has received significant attention due to the ability to achieve morphologies with dimensions in the range of 10 to 20 nm or even below. Block copolymer lithography is a cost-effective, parallel, and scalable nanolithography for densely packed periodic arrays of nanoscale features, whose typical dimension scale is beyond the resolution limit of conventional photolithography. Researchers have now introduced a conceptually new and versatile strategy to achieve asymmetric line patterns. This is the first work to demonstrate that highly asymmetric line nanopatterning is possible even though a block copolymer self-assembly technique is used.
Lithography based on block copolymer self-assembly has received significant attention due to the ability to achieve morphologies with dimensions in the range of 10 to 20 nm or even below. Block copolymer lithography is a cost-effective, parallel, and scalable nanolithography for densely packed periodic arrays of nanoscale features, whose typical dimension scale is beyond the resolution limit of conventional photolithography. Researchers have now introduced a conceptually new and versatile strategy to achieve asymmetric line patterns. This is the first work to demonstrate that highly asymmetric line nanopatterning is possible even though a block copolymer self-assembly technique is used.
Sep 5th, 2012
 Ultrasonic spray pyrolysis (USP) has been widely used in industry for spherical solid powder production, particularly of metal oxides. For some applications, though, porous particles are more desirable than dense ones. Back in 2005, researchers developed a technique to synthesize porous micro- and nanoparticles via USP. This method has since been expanded to prepare porous carbon microspheres. The high surface area and unique porous structures suggest that porous carbon spheres can be useful for electrode materials, adsorbents, and catalyst supports. Researchers at the University of Illinois already demonstrated the use of carbon microspheres as supercapacitors. Now, the team has expanded the aerosol synthesis of porous carbon materials by the use of energetic carbon precursors. Some of the resulting porous carbon spheres exhibit unique and unprecedented morphologies.
Ultrasonic spray pyrolysis (USP) has been widely used in industry for spherical solid powder production, particularly of metal oxides. For some applications, though, porous particles are more desirable than dense ones. Back in 2005, researchers developed a technique to synthesize porous micro- and nanoparticles via USP. This method has since been expanded to prepare porous carbon microspheres. The high surface area and unique porous structures suggest that porous carbon spheres can be useful for electrode materials, adsorbents, and catalyst supports. Researchers at the University of Illinois already demonstrated the use of carbon microspheres as supercapacitors. Now, the team has expanded the aerosol synthesis of porous carbon materials by the use of energetic carbon precursors. Some of the resulting porous carbon spheres exhibit unique and unprecedented morphologies.
Sep 3rd, 2012
 In order to fabricate stimuli-responsive materials, researchers have shown a lot of interest in asymmetric materials such as modulated gels which consist of a controlled layer that is responsive to an environmental stimuli and a nonresponsive substrate layer. And while much effort has gone into creating free-standing films through layer-by-layer (LbL) assembly, relatively little attention has been paid to the asymmetric properties or functionalization of the two surfaces of such free-standing layer-by-layer films. In new work, researchers have now reported the fabrication of asymmetric free-standing layer-by-layer film with asymmetric wettability - one surface is superhydrophobic and the other one is hydrophilic. The superhydrophobic side is water-repellent while the hydrophilic side can absorb/desorb water easily.
In order to fabricate stimuli-responsive materials, researchers have shown a lot of interest in asymmetric materials such as modulated gels which consist of a controlled layer that is responsive to an environmental stimuli and a nonresponsive substrate layer. And while much effort has gone into creating free-standing films through layer-by-layer (LbL) assembly, relatively little attention has been paid to the asymmetric properties or functionalization of the two surfaces of such free-standing layer-by-layer films. In new work, researchers have now reported the fabrication of asymmetric free-standing layer-by-layer film with asymmetric wettability - one surface is superhydrophobic and the other one is hydrophilic. The superhydrophobic side is water-repellent while the hydrophilic side can absorb/desorb water easily. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





