Showing Spotlights 25 - 32 of 198 in category All (newest first):
 Researchers report an in vitro and in vivo proof-of-concept for the capacity of triboelectric nanogenerator (TENG) technology to function as a simple, scalable, inexpensive, and self-powered device for tactile sensory restoration. This integrated tactile sensory restoration device powers itself and is suitable for implantation. It bypasses damaged nerves and activate sensory neurons at various levels of electrical potential, generated by different levels of tactile pressure on the implanted device.
Researchers report an in vitro and in vivo proof-of-concept for the capacity of triboelectric nanogenerator (TENG) technology to function as a simple, scalable, inexpensive, and self-powered device for tactile sensory restoration. This integrated tactile sensory restoration device powers itself and is suitable for implantation. It bypasses damaged nerves and activate sensory neurons at various levels of electrical potential, generated by different levels of tactile pressure on the implanted device.
Jul 1st, 2021
 In general, there are three types of driving force for ion transport: electric field; mechanical pressure; and concentration gradient. Recently, light has been propsoed as a fourth. Now, researchers report the incorporation of a transition metal dichalcogenides based van der Waals multilayer heterostructure into nanofluidic materials, and demonstrate a new photo-induced active ion transport phenomenon. This will inspire a broad range of fundamental research and practical application for light-controlled ionic circuits, artificial photosynthesis, biomimetic energy conversion, and so on.
In general, there are three types of driving force for ion transport: electric field; mechanical pressure; and concentration gradient. Recently, light has been propsoed as a fourth. Now, researchers report the incorporation of a transition metal dichalcogenides based van der Waals multilayer heterostructure into nanofluidic materials, and demonstrate a new photo-induced active ion transport phenomenon. This will inspire a broad range of fundamental research and practical application for light-controlled ionic circuits, artificial photosynthesis, biomimetic energy conversion, and so on.
Mar 4th, 2021
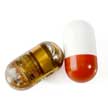 A major challenge in the development of implantable and ingestible biomedical electronic devices is the limited lifetime of their power sources. The energy requirements of these devices are highly dependent on their application and the complexity of the required electrical systems. The power unit, which is composed of one or more energy sources - batteries, energy-harvesting, and energy transfer - as well as power management circuits, supplies electrical energy to the whole system.
A major challenge in the development of implantable and ingestible biomedical electronic devices is the limited lifetime of their power sources. The energy requirements of these devices are highly dependent on their application and the complexity of the required electrical systems. The power unit, which is composed of one or more energy sources - batteries, energy-harvesting, and energy transfer - as well as power management circuits, supplies electrical energy to the whole system.
Feb 16th, 2021
 In the search for battery technologies that one day could replace lithium-ion batteries and meet power demands within the size and cost constraints of car makers, aerospace and other industries, researchers are exploring lithium-air batteries (LAB). Generally, to make LAB technology practical, two goals are highly important: a novel cathode that uses a highly active and stable catalysts to enhance ORR and OER kinetics; and a proper electrolyte design that can promote the solvent-based growth mechanism for the discharge products.
In the search for battery technologies that one day could replace lithium-ion batteries and meet power demands within the size and cost constraints of car makers, aerospace and other industries, researchers are exploring lithium-air batteries (LAB). Generally, to make LAB technology practical, two goals are highly important: a novel cathode that uses a highly active and stable catalysts to enhance ORR and OER kinetics; and a proper electrolyte design that can promote the solvent-based growth mechanism for the discharge products.
Dec 29th, 2020
 In order to make dynamic written content, for instance a news reader, available for blind or vision-impaired people, scientists have come up with various designs of sheet-type, refreshable Braille displays. Expanding the use of nanotechnologies in designing next-generation Braille readers, researchers now have successfully demonstrated a refreshable Braille display system by using a safe high-voltage power source - a triboelectric nanogenerator.
In order to make dynamic written content, for instance a news reader, available for blind or vision-impaired people, scientists have come up with various designs of sheet-type, refreshable Braille displays. Expanding the use of nanotechnologies in designing next-generation Braille readers, researchers now have successfully demonstrated a refreshable Braille display system by using a safe high-voltage power source - a triboelectric nanogenerator.
Nov 5th, 2020
 Researchers have discovered a novel 'sandwiched' silicon electrode structure that can withstand 500 cycles and deliver capacities three times larger than graphite. They used freestanding sheets made of carbon nanotubes - bucky papers - for sandwiching silicon nanoparticles. These nanotubes form a quasi-three-dimensional structure and hold silicon nanoparticles together even after 100 cycles and mitigate electrical resistance arising from breaking of particles. The sandwiched silicon anode was able to withstand discharging rates as high as 4C.
Researchers have discovered a novel 'sandwiched' silicon electrode structure that can withstand 500 cycles and deliver capacities three times larger than graphite. They used freestanding sheets made of carbon nanotubes - bucky papers - for sandwiching silicon nanoparticles. These nanotubes form a quasi-three-dimensional structure and hold silicon nanoparticles together even after 100 cycles and mitigate electrical resistance arising from breaking of particles. The sandwiched silicon anode was able to withstand discharging rates as high as 4C.
Jul 22nd, 2020
 The concept of nanostructuring of battery electrodes is not new; it has been frequently employed to improve the charging speed and, in some cases, stability of Li-ion electrodes. Researchers now demonstrated 3D-interconnected Ni nanowire meshes as current collectors and structural scaffolds for building nanostructured Li-ion electrodes. This material exhibits a semi-ordered structure and is characterized by some of the highest combined porosity and surface-to-volume ratio among macroporous metals.
The concept of nanostructuring of battery electrodes is not new; it has been frequently employed to improve the charging speed and, in some cases, stability of Li-ion electrodes. Researchers now demonstrated 3D-interconnected Ni nanowire meshes as current collectors and structural scaffolds for building nanostructured Li-ion electrodes. This material exhibits a semi-ordered structure and is characterized by some of the highest combined porosity and surface-to-volume ratio among macroporous metals.
Jul 6th, 2020
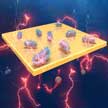 Researchers have identified the potential of using nanoscale carbon dots on bacteria for augmented bioelectricity production. They used this to demonstrate a novel method for significantly increasing the bacterial bioelectricity generation and applied it to set up microbial fuel cells. The results show that carbon dots' highly conductive carbon core not only improved overall systemic electrical conductivity but could also increase the packing density of the electron transfer path. This significantly improves the bioelectricity production of microbial fuel cells.
Researchers have identified the potential of using nanoscale carbon dots on bacteria for augmented bioelectricity production. They used this to demonstrate a novel method for significantly increasing the bacterial bioelectricity generation and applied it to set up microbial fuel cells. The results show that carbon dots' highly conductive carbon core not only improved overall systemic electrical conductivity but could also increase the packing density of the electron transfer path. This significantly improves the bioelectricity production of microbial fuel cells.
Mar 19th, 2020
 Researchers report an in vitro and in vivo proof-of-concept for the capacity of triboelectric nanogenerator (TENG) technology to function as a simple, scalable, inexpensive, and self-powered device for tactile sensory restoration. This integrated tactile sensory restoration device powers itself and is suitable for implantation. It bypasses damaged nerves and activate sensory neurons at various levels of electrical potential, generated by different levels of tactile pressure on the implanted device.
Researchers report an in vitro and in vivo proof-of-concept for the capacity of triboelectric nanogenerator (TENG) technology to function as a simple, scalable, inexpensive, and self-powered device for tactile sensory restoration. This integrated tactile sensory restoration device powers itself and is suitable for implantation. It bypasses damaged nerves and activate sensory neurons at various levels of electrical potential, generated by different levels of tactile pressure on the implanted device. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





