Showing Spotlights 33 - 40 of 315 in category All (newest first):
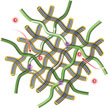 Researchers have created a free-standing carbon nanotube paper electrode with high sulfur loading for lithium-sulfur batteries employing a bottom-up strategy to design and fabricate a hierarchical structure. This new fabrication method does not employ aluminum foil or binders, thereby fully utilizing the advantage of a Li-S system with high specific capacity. This proof-of-concept experiment indicates that the rational design of the nanostructured electrode offers the possibility to efficiently use the active materials at practical loading.
Researchers have created a free-standing carbon nanotube paper electrode with high sulfur loading for lithium-sulfur batteries employing a bottom-up strategy to design and fabricate a hierarchical structure. This new fabrication method does not employ aluminum foil or binders, thereby fully utilizing the advantage of a Li-S system with high specific capacity. This proof-of-concept experiment indicates that the rational design of the nanostructured electrode offers the possibility to efficiently use the active materials at practical loading.
Oct 17th, 2014
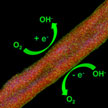 Researchers have demonstrated a unique coaxial carbon nanocable material with pristine carbon nanotubes as the core and nitrogen-doped wrinkled carbon layer as the shell. The active sites rendered by the surface enriched dopant atoms on the carbon nanocables are accessible and effective to catalyze the oxygen involved electrochemical reactions. These coaxial nanocables afford higher ORR/OER current compared with the routine bulk doped nitrogen-doped carbon nanotubes.
Researchers have demonstrated a unique coaxial carbon nanocable material with pristine carbon nanotubes as the core and nitrogen-doped wrinkled carbon layer as the shell. The active sites rendered by the surface enriched dopant atoms on the carbon nanocables are accessible and effective to catalyze the oxygen involved electrochemical reactions. These coaxial nanocables afford higher ORR/OER current compared with the routine bulk doped nitrogen-doped carbon nanotubes.
Oct 10th, 2014
 Researchers consider the rational combination of carbon nanotubes (CNTs) and graphene into three-dimensional hybrids an effective route to amplify the inherent physical properties at the macroscale. By in situ nitrogen doping and structural hybridization of carbon nanotubes and graphene, researchers have now successfully fabricated nitrogen-doped aligned carbon nanotube/graphene sandwiches. In this work, aligned CNTs and graphene layers were anchored to each other, constructing a sandwich-like hierarchical architecture with efficient 3D electron transfer pathways and ion diffusion channels.
Researchers consider the rational combination of carbon nanotubes (CNTs) and graphene into three-dimensional hybrids an effective route to amplify the inherent physical properties at the macroscale. By in situ nitrogen doping and structural hybridization of carbon nanotubes and graphene, researchers have now successfully fabricated nitrogen-doped aligned carbon nanotube/graphene sandwiches. In this work, aligned CNTs and graphene layers were anchored to each other, constructing a sandwich-like hierarchical architecture with efficient 3D electron transfer pathways and ion diffusion channels.
Sep 12th, 2014
 The microstructures of carbon nanotube assemblies determine their properties, for example, highly graphitized CNTs exhibit excellent mechanical and electrical properties; while CNTs with defects and poor crystallinity are beneficial for research on field emission property and hydrogen storage capacity. Therefore, it is of vital importance to control the CNT microstructures effectively for desired applications. A new technique can solve a problem of three-dimensional orientation control of CNTs in microscopic scale.
The microstructures of carbon nanotube assemblies determine their properties, for example, highly graphitized CNTs exhibit excellent mechanical and electrical properties; while CNTs with defects and poor crystallinity are beneficial for research on field emission property and hydrogen storage capacity. Therefore, it is of vital importance to control the CNT microstructures effectively for desired applications. A new technique can solve a problem of three-dimensional orientation control of CNTs in microscopic scale.
Jun 30th, 2014
 Researchers report for the first time the fabrication and measurement of all-inkjet-printed, all-air-processed organic solar cells. Organic photovoltaic technologies have the potential to become a thin-film alternative to inorganic silicon photovoltaics due to their intrinsic potential for low-cost print processing from solution - high-speed and at low temperature. Organic solar cells can be integrated into building facades and windows because they are optically translucent and can be manufactured on large areas at high throughput.
Researchers report for the first time the fabrication and measurement of all-inkjet-printed, all-air-processed organic solar cells. Organic photovoltaic technologies have the potential to become a thin-film alternative to inorganic silicon photovoltaics due to their intrinsic potential for low-cost print processing from solution - high-speed and at low temperature. Organic solar cells can be integrated into building facades and windows because they are optically translucent and can be manufactured on large areas at high throughput.
Jun 16th, 2014
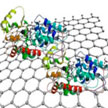 Studies have shown that in a favorable nano environment, enzyme immobilization onto nanosupports could lead to increased enzyme stability and improved specificity, and could allow for prolonged enzyme functionality through chemical and physical treatment. Researchers also have shown that immobilization onto carbon-based nanosupports can increase the enzyme turnover and allow for prolonged enzyme-based conjugates isolation and usage. In new work, researchers have now taken another step towards the detailed characterization and optimization of enzyme-nanosupport interface reactions.
Studies have shown that in a favorable nano environment, enzyme immobilization onto nanosupports could lead to increased enzyme stability and improved specificity, and could allow for prolonged enzyme functionality through chemical and physical treatment. Researchers also have shown that immobilization onto carbon-based nanosupports can increase the enzyme turnover and allow for prolonged enzyme-based conjugates isolation and usage. In new work, researchers have now taken another step towards the detailed characterization and optimization of enzyme-nanosupport interface reactions.
May 14th, 2014
 Taking the approach of flexible electronics one step further, researchers now have integrated all-carbon based electronic devices to live plants and insects. They developed an unconventional approach for the in situ synthesis of monolithically integrated electronic devices based on single-walled carbon nanotube channels and graphitic electrodes. The highly flexible transistors were formed directly by the in situ synthesis using patterned metal catalyst films and subsequently could be transferred to both planar and nonplanar substrates, including papers, clothes, and fingernails.
Taking the approach of flexible electronics one step further, researchers now have integrated all-carbon based electronic devices to live plants and insects. They developed an unconventional approach for the in situ synthesis of monolithically integrated electronic devices based on single-walled carbon nanotube channels and graphitic electrodes. The highly flexible transistors were formed directly by the in situ synthesis using patterned metal catalyst films and subsequently could be transferred to both planar and nonplanar substrates, including papers, clothes, and fingernails.
May 2nd, 2014
 Most of the accomplishments in building carbon nanotube circuits have come at the single-nanotube level. Researchers have been struggling with two major obstacles in building CNT-based circuits: the presence of metallic CNTs and a 'perfect' alignment of nanotubes. In new work, researchers have now demonstrated the ability to fabricate, in a scalable manner, larger-scale CNFET circuits at highly scaled technology nodes. The channel lengths are ranging from 90 nm to sub-20 nm.
Most of the accomplishments in building carbon nanotube circuits have come at the single-nanotube level. Researchers have been struggling with two major obstacles in building CNT-based circuits: the presence of metallic CNTs and a 'perfect' alignment of nanotubes. In new work, researchers have now demonstrated the ability to fabricate, in a scalable manner, larger-scale CNFET circuits at highly scaled technology nodes. The channel lengths are ranging from 90 nm to sub-20 nm.
Apr 29th, 2014
 Researchers have created a free-standing carbon nanotube paper electrode with high sulfur loading for lithium-sulfur batteries employing a bottom-up strategy to design and fabricate a hierarchical structure. This new fabrication method does not employ aluminum foil or binders, thereby fully utilizing the advantage of a Li-S system with high specific capacity. This proof-of-concept experiment indicates that the rational design of the nanostructured electrode offers the possibility to efficiently use the active materials at practical loading.
Researchers have created a free-standing carbon nanotube paper electrode with high sulfur loading for lithium-sulfur batteries employing a bottom-up strategy to design and fabricate a hierarchical structure. This new fabrication method does not employ aluminum foil or binders, thereby fully utilizing the advantage of a Li-S system with high specific capacity. This proof-of-concept experiment indicates that the rational design of the nanostructured electrode offers the possibility to efficiently use the active materials at practical loading.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





