Showing Spotlights 393 - 400 of 624 in category All (newest first):
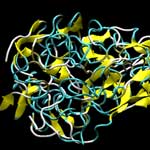 Material scientists have been fascinated by spider silks for a long time - ultra-strong and extensible self-assembling biopolymers that outperform the mechanical characteristics of many synthetic materials, including steel. While the source of these unique material properties are thought to lie in the distinct protein structures found in spider silk, the physical mechanisms behind them have remained poorly understood for decades. This is partly due to the fact that structural models of spider silk with atomic-level resolution have not been available, preventing a molecular-level analysis. So far, only small models of isolated crystalline domains of silk have been reported. Yet, the integrated structure of the semi-amorphous regions combined with crystalline domains in silk is critical for properties such as toughness and fracture mechanisms. To unravel silk's secrets and ultimately be able to create synthetic materials that duplicate, or even exceed, the extraordinary properties of natural silk, researchers need to fully understand the links between genetic makeup, chemical interactions, and structure, as well as its macroscale mechanical properties.
Material scientists have been fascinated by spider silks for a long time - ultra-strong and extensible self-assembling biopolymers that outperform the mechanical characteristics of many synthetic materials, including steel. While the source of these unique material properties are thought to lie in the distinct protein structures found in spider silk, the physical mechanisms behind them have remained poorly understood for decades. This is partly due to the fact that structural models of spider silk with atomic-level resolution have not been available, preventing a molecular-level analysis. So far, only small models of isolated crystalline domains of silk have been reported. Yet, the integrated structure of the semi-amorphous regions combined with crystalline domains in silk is critical for properties such as toughness and fracture mechanisms. To unravel silk's secrets and ultimately be able to create synthetic materials that duplicate, or even exceed, the extraordinary properties of natural silk, researchers need to fully understand the links between genetic makeup, chemical interactions, and structure, as well as its macroscale mechanical properties.
Apr 20th, 2010
 Controllable fabrication of complex, three-dimensional (3D) nanoscale structures remains a difficult challenge. Researchers are experimenting with a wide range of nanofabrication techniques, from top-down approaches such as mechanical machining to biomimetic replication of complex biotemplates to bottom-up fabrication using anisotropic self-assembly systems - to just name a few examples. New work at Johns Hopkins University utilizes the extrinsic stresses that develop during thin-film deposition - but can also be induced by external forces post-deposition - for the self-assembly of 3D curved and simultaneously patterned structures. This technique is fairly simple and low-cost since it requires only thermal evaporation and low temperature processing; the stress for self-assembly can be controlled to occur only when required. Furthermore, the layers that are selected for 3D structuring can also easily be patterned with conventional electron-beam lithographic processing.
Controllable fabrication of complex, three-dimensional (3D) nanoscale structures remains a difficult challenge. Researchers are experimenting with a wide range of nanofabrication techniques, from top-down approaches such as mechanical machining to biomimetic replication of complex biotemplates to bottom-up fabrication using anisotropic self-assembly systems - to just name a few examples. New work at Johns Hopkins University utilizes the extrinsic stresses that develop during thin-film deposition - but can also be induced by external forces post-deposition - for the self-assembly of 3D curved and simultaneously patterned structures. This technique is fairly simple and low-cost since it requires only thermal evaporation and low temperature processing; the stress for self-assembly can be controlled to occur only when required. Furthermore, the layers that are selected for 3D structuring can also easily be patterned with conventional electron-beam lithographic processing.
Apr 16th, 2010
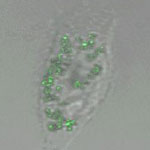 Silver nanoparticles can now be found in all kinds of products, from socks to food containers to coatings for medical devices. Valued for its infection-fighting, antimicrobial properties, silver, in its modern incarnation as silver nanoparticles, has become the promising antimicrobial material in a variety of applications because the nanoparticles can damage bacterial cells. Due to their plasmonic properties and easy surface chemistry silver nanoparticles are also beginning to attract interest among nanomedicine researchers. However, the surface chemistry of nanoparticles that governs their interactions with other constituents in their environment has critical importance. Therefore, chemically altering the surface properties of nanoparticles with polymers, biological ligands and macromolecules is actively being explored.
Silver nanoparticles can now be found in all kinds of products, from socks to food containers to coatings for medical devices. Valued for its infection-fighting, antimicrobial properties, silver, in its modern incarnation as silver nanoparticles, has become the promising antimicrobial material in a variety of applications because the nanoparticles can damage bacterial cells. Due to their plasmonic properties and easy surface chemistry silver nanoparticles are also beginning to attract interest among nanomedicine researchers. However, the surface chemistry of nanoparticles that governs their interactions with other constituents in their environment has critical importance. Therefore, chemically altering the surface properties of nanoparticles with polymers, biological ligands and macromolecules is actively being explored.
Apr 9th, 2010
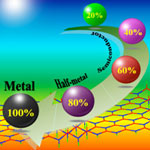 Researchers have been exploring boron nitride (BN) nanomaterials - from nanotubes to nanosheets and nanoribbons - and found similarities, but also differences to the the properties of the corresponding carbon nanomaterials. Motivated by the outstanding properties of graphene, the boron nitride single layer - a structural analogy of graphene - has been extensively studied both theoretically and experimentally, and has been experimentally realized. However, BN nanomaterials are wide-band-gap semiconductors, and their band structures are rather robust, and difficult to modulate, which is a substantial obstacle for their applications in nanoelectronic devices. Researchers have now shown that hydrogenation might be a simple approach to tune the band structure of graphene-like boron nitride structures.
Researchers have been exploring boron nitride (BN) nanomaterials - from nanotubes to nanosheets and nanoribbons - and found similarities, but also differences to the the properties of the corresponding carbon nanomaterials. Motivated by the outstanding properties of graphene, the boron nitride single layer - a structural analogy of graphene - has been extensively studied both theoretically and experimentally, and has been experimentally realized. However, BN nanomaterials are wide-band-gap semiconductors, and their band structures are rather robust, and difficult to modulate, which is a substantial obstacle for their applications in nanoelectronic devices. Researchers have now shown that hydrogenation might be a simple approach to tune the band structure of graphene-like boron nitride structures.
Apr 6th, 2010
 The concept of self-healing has become a popular theme in the field of material science. The whole concept of 'smart' materials that react on external impact - pH, humidity changes, or distortion of the coating integrity - and repair themselves has experienced a tremendous boost with the advent of nanotechnology. The nanoscale multilayer structure of a coating, in which the components are integrated and mutually reactive, is a main point in sophisticated and strong corrosion protection. Researchers have now proposed a new approach to self-healing polymer coating systems based on an electrospun coaxial healing agent. Electrospinning offers a number of unique opportunities. Most significantly, the location and concentration of the healing component can be spatially varied.
The concept of self-healing has become a popular theme in the field of material science. The whole concept of 'smart' materials that react on external impact - pH, humidity changes, or distortion of the coating integrity - and repair themselves has experienced a tremendous boost with the advent of nanotechnology. The nanoscale multilayer structure of a coating, in which the components are integrated and mutually reactive, is a main point in sophisticated and strong corrosion protection. Researchers have now proposed a new approach to self-healing polymer coating systems based on an electrospun coaxial healing agent. Electrospinning offers a number of unique opportunities. Most significantly, the location and concentration of the healing component can be spatially varied.
Mar 29th, 2010
 It has proven difficult to directly manufacture functional nanostructures and nanodevices with predetermined designs using bottom-up processes alone. So far, developing top-down machining techniques capable of fabricating structural/functional nanostructures and nanodevices appears to be indispensable, but mechanical machining tools with nanometer precision are still lacking. A grand challenge in nanotechnology is to machine three-dimensional nanostructures in a controllable and reproducible fashion. That begs the question if traditional top-down mechanical machining can also be realized at the nanoscale. So far, the conventional wisdom has been that traditional top-down mechanical machining like cutting and milling using a lathe is impossible at the nanoscale. Nanotechnologists considered most of the traditional top-down approaches as not applicable for fabricating nanostructure and nanodevices. However, as it turns out, there still is room at the bottom for traditional mechanical machining.
It has proven difficult to directly manufacture functional nanostructures and nanodevices with predetermined designs using bottom-up processes alone. So far, developing top-down machining techniques capable of fabricating structural/functional nanostructures and nanodevices appears to be indispensable, but mechanical machining tools with nanometer precision are still lacking. A grand challenge in nanotechnology is to machine three-dimensional nanostructures in a controllable and reproducible fashion. That begs the question if traditional top-down mechanical machining can also be realized at the nanoscale. So far, the conventional wisdom has been that traditional top-down mechanical machining like cutting and milling using a lathe is impossible at the nanoscale. Nanotechnologists considered most of the traditional top-down approaches as not applicable for fabricating nanostructure and nanodevices. However, as it turns out, there still is room at the bottom for traditional mechanical machining.
Mar 26th, 2010
 Freshwater could become the oil of the 21st century - scarce, expensive and fought over. While over 70 per cent of the Earth's surface is covered by water, most of it is unusable for human consumption. Technological advances have made desalination and demineralization feasible - albeit expensive - solutions for increasing the world's supply of freshwater. However, nanotechnology- based water purification devices have the potential to transform the field of desalination. Researchers have now demonstrated a new, efficient and fouling-free desalination process based on the ion concentration polarization (ICP) phenomenon - a fundamental electrochemical transport phenomenon that occurs when an ion current is passed through ion-selective membranes - for direct desalination of sea water.
Freshwater could become the oil of the 21st century - scarce, expensive and fought over. While over 70 per cent of the Earth's surface is covered by water, most of it is unusable for human consumption. Technological advances have made desalination and demineralization feasible - albeit expensive - solutions for increasing the world's supply of freshwater. However, nanotechnology- based water purification devices have the potential to transform the field of desalination. Researchers have now demonstrated a new, efficient and fouling-free desalination process based on the ion concentration polarization (ICP) phenomenon - a fundamental electrochemical transport phenomenon that occurs when an ion current is passed through ion-selective membranes - for direct desalination of sea water.
Mar 22nd, 2010
 Surface energy is ubiquitous in nature and it plays an important role in many scientific areas such as for instance surface physics, biophysics, surface chemistry, or catalysis. So far it has been impractical to consider utilizing surface energy as an energy source because there are few molecules or atoms involved in the surface interaction and the density of surface energy is low. However, due to the lower power consumption requirements of nanotechnology devices and the higher specific surface area for nanomaterials it appears attractive to use surface energy at the nanoscale. Researchers in China have now demonstrated that an effective design of single-walled carbon nanotubes can be used to convert the surface energy of liquids into electricity.
Surface energy is ubiquitous in nature and it plays an important role in many scientific areas such as for instance surface physics, biophysics, surface chemistry, or catalysis. So far it has been impractical to consider utilizing surface energy as an energy source because there are few molecules or atoms involved in the surface interaction and the density of surface energy is low. However, due to the lower power consumption requirements of nanotechnology devices and the higher specific surface area for nanomaterials it appears attractive to use surface energy at the nanoscale. Researchers in China have now demonstrated that an effective design of single-walled carbon nanotubes can be used to convert the surface energy of liquids into electricity.
Mar 5th, 2010
 Material scientists have been fascinated by spider silks for a long time - ultra-strong and extensible self-assembling biopolymers that outperform the mechanical characteristics of many synthetic materials, including steel. While the source of these unique material properties are thought to lie in the distinct protein structures found in spider silk, the physical mechanisms behind them have remained poorly understood for decades. This is partly due to the fact that structural models of spider silk with atomic-level resolution have not been available, preventing a molecular-level analysis. So far, only small models of isolated crystalline domains of silk have been reported. Yet, the integrated structure of the semi-amorphous regions combined with crystalline domains in silk is critical for properties such as toughness and fracture mechanisms. To unravel silk's secrets and ultimately be able to create synthetic materials that duplicate, or even exceed, the extraordinary properties of natural silk, researchers need to fully understand the links between genetic makeup, chemical interactions, and structure, as well as its macroscale mechanical properties.
Material scientists have been fascinated by spider silks for a long time - ultra-strong and extensible self-assembling biopolymers that outperform the mechanical characteristics of many synthetic materials, including steel. While the source of these unique material properties are thought to lie in the distinct protein structures found in spider silk, the physical mechanisms behind them have remained poorly understood for decades. This is partly due to the fact that structural models of spider silk with atomic-level resolution have not been available, preventing a molecular-level analysis. So far, only small models of isolated crystalline domains of silk have been reported. Yet, the integrated structure of the semi-amorphous regions combined with crystalline domains in silk is critical for properties such as toughness and fracture mechanisms. To unravel silk's secrets and ultimately be able to create synthetic materials that duplicate, or even exceed, the extraordinary properties of natural silk, researchers need to fully understand the links between genetic makeup, chemical interactions, and structure, as well as its macroscale mechanical properties.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





