Showing Spotlights 41 - 48 of 137 in category All (newest first):
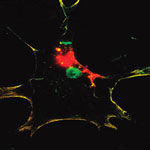 Owing to their large surface area, strong infrared photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms. However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies. Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries. In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages.
Owing to their large surface area, strong infrared photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms. However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies. Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries. In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages.
Mar 10th, 2011
 There is a need for the larger nanotechnology community synthesizing, applying or characterizing nanomaterials to have a methodology to evaluate the risk and to apply adequate protection measures to limit human exposure. Researchers in Switzerland have now taken the initiative and presented a practical, user-friendly procedure for a university-wide safety and health management of nanomaterials, developed as a multi-stakeholder effort (government, accident insurance, researchers and experts for occupational safety and health). The procedure consists of two parts: Using a decision tree, nano-labs are sorted into three hazard classes, which corresponds to analogue approaches applied to other hazard types (biohazard, radioprotection or chemistry). A list of required prevention/protection measures (safety barriers) for each hazard level is then provided.
There is a need for the larger nanotechnology community synthesizing, applying or characterizing nanomaterials to have a methodology to evaluate the risk and to apply adequate protection measures to limit human exposure. Researchers in Switzerland have now taken the initiative and presented a practical, user-friendly procedure for a university-wide safety and health management of nanomaterials, developed as a multi-stakeholder effort (government, accident insurance, researchers and experts for occupational safety and health). The procedure consists of two parts: Using a decision tree, nano-labs are sorted into three hazard classes, which corresponds to analogue approaches applied to other hazard types (biohazard, radioprotection or chemistry). A list of required prevention/protection measures (safety barriers) for each hazard level is then provided.
Feb 14th, 2011
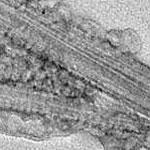 The toxicity issues surrounding carbon nanotubes (CNTs) are highly relevant for two reasons: Firstly, as more and more products containing CNTs come to market, there is a chance that free CNTs get released during their life cycles, most likely during production or disposal, and find their way through the environment into the body. Secondly, and much more pertinent with regard to potential health risks, is the use of CNTs in biological and medical settings. Some groups are using CNTs in research for vaccination as well as gene and cancer therapy. Here, the CNT applications are designed to interact directly with the immune system. Understanding the interplay between CNTs and immune proteins is therefore critical for both improving CNT applications in biology and medicine and avoiding potentially noxious immune responses.
The toxicity issues surrounding carbon nanotubes (CNTs) are highly relevant for two reasons: Firstly, as more and more products containing CNTs come to market, there is a chance that free CNTs get released during their life cycles, most likely during production or disposal, and find their way through the environment into the body. Secondly, and much more pertinent with regard to potential health risks, is the use of CNTs in biological and medical settings. Some groups are using CNTs in research for vaccination as well as gene and cancer therapy. Here, the CNT applications are designed to interact directly with the immune system. Understanding the interplay between CNTs and immune proteins is therefore critical for both improving CNT applications in biology and medicine and avoiding potentially noxious immune responses.
Jan 13th, 2011
 Although the literature grows on the use of science to inform decisions on the environmental, health and safety implications of nanotechnology, little has been published by those who make such decisions. In a recent commentary in Nature Nanotechnology, officials of the US Environmental Protection Agency (EPA), the European Commission and the Organisation for Economic Co-operation and Development (OECD), discuss the types of decision facing government regulators, the new considerations nanotechnology brings to decision-making, the role of science in informing decisions, how regulators cooperate internationally on policy issues, and the challenges that lie ahead. The authors provide an overview of key reports and regulations and then discuss the complexity of issues with regard to addressing nanomaterials within the context of existing regulations and the need to weigh nanomaterial risk and benefits.
Although the literature grows on the use of science to inform decisions on the environmental, health and safety implications of nanotechnology, little has been published by those who make such decisions. In a recent commentary in Nature Nanotechnology, officials of the US Environmental Protection Agency (EPA), the European Commission and the Organisation for Economic Co-operation and Development (OECD), discuss the types of decision facing government regulators, the new considerations nanotechnology brings to decision-making, the role of science in informing decisions, how regulators cooperate internationally on policy issues, and the challenges that lie ahead. The authors provide an overview of key reports and regulations and then discuss the complexity of issues with regard to addressing nanomaterials within the context of existing regulations and the need to weigh nanomaterial risk and benefits.
Dec 21st, 2010
 The use of minute particles as drug carriers for targeted therapy has been studied and discussed for more than 20 years. A selective accumulation of active substances in target tissues has been demonstrated for certain so-called nanocarrier systems that are administered bound to pharmaceutical drugs. Great expectations are placed on nanocarrier systems that can overcome natural barriers such as the blood-brain barrier (BBB) and transport the medication directly to the desired tissue and thus heal neurological diseases that were formerly incurable. The BBB represents the border between the circulating blood and the fluid in the central nervous system. It functions to protect the sensitive nerve cells from foreign substances and infections from the blood. Whether nanoparticles enter the central nervous system unintentionally and induce health problems is also being debated.
The use of minute particles as drug carriers for targeted therapy has been studied and discussed for more than 20 years. A selective accumulation of active substances in target tissues has been demonstrated for certain so-called nanocarrier systems that are administered bound to pharmaceutical drugs. Great expectations are placed on nanocarrier systems that can overcome natural barriers such as the blood-brain barrier (BBB) and transport the medication directly to the desired tissue and thus heal neurological diseases that were formerly incurable. The BBB represents the border between the circulating blood and the fluid in the central nervous system. It functions to protect the sensitive nerve cells from foreign substances and infections from the blood. Whether nanoparticles enter the central nervous system unintentionally and induce health problems is also being debated.
Dec 8th, 2010
 In a new research field that could be called 'experimental nanotoxicology', scientists have now, for the first time, demonstrated that biological effects of manufactured nanoparticles can be predicted using their chemical, physical, and geometrical properties. The results successfully demonstrate the high potential of cheminformatics approaches for improving the experimental design and prioritizing the biological testing of novel manufactured nanoparticles. The team modeled their approach after a process used in drug design and chemical synthesis, where the chemical structure of a new compound is quantitatively correlated with a well defined process, such as biological activity or chemical reactivity. Such a Quantitative Structure Activity Relationship (QSAR) can then be utilized to help guide chemical synthesis and drug design. They termed their approach quantitative nanostructure-activity relationship (QNAR) modeling.
In a new research field that could be called 'experimental nanotoxicology', scientists have now, for the first time, demonstrated that biological effects of manufactured nanoparticles can be predicted using their chemical, physical, and geometrical properties. The results successfully demonstrate the high potential of cheminformatics approaches for improving the experimental design and prioritizing the biological testing of novel manufactured nanoparticles. The team modeled their approach after a process used in drug design and chemical synthesis, where the chemical structure of a new compound is quantitatively correlated with a well defined process, such as biological activity or chemical reactivity. Such a Quantitative Structure Activity Relationship (QSAR) can then be utilized to help guide chemical synthesis and drug design. They termed their approach quantitative nanostructure-activity relationship (QNAR) modeling.
Nov 8th, 2010
 Understanding the behavior and impacts of nanomaterials in the environment and in human health is a daunting task. Today, we don't even know what the impact of most chemicals is, and that includes products we have been using for many years. Nevertheless, a general understanding about nanotoxicity is slowly emerging as the body of research on cytotoxicity, genotoxicity, and ecotoxicity of nanomaterials grows. Many of the published toxicity studies have limited relevance, due, in large part, to study design limitations, including inadequate justification for dose selection or route of exposure criteria. A recently published article addresses myths and misconceptions regarding nanotoxicology.
Understanding the behavior and impacts of nanomaterials in the environment and in human health is a daunting task. Today, we don't even know what the impact of most chemicals is, and that includes products we have been using for many years. Nevertheless, a general understanding about nanotoxicity is slowly emerging as the body of research on cytotoxicity, genotoxicity, and ecotoxicity of nanomaterials grows. Many of the published toxicity studies have limited relevance, due, in large part, to study design limitations, including inadequate justification for dose selection or route of exposure criteria. A recently published article addresses myths and misconceptions regarding nanotoxicology.
Nov 2nd, 2010
 Silver nanoparticles are one of the most extensively used type of nanoparticles in consumer products due to the unique antibacterial activity of silver. There have been raising environmental concerns over their adverse ecological effects, along with ionic silver potentially released from the particles. To predict the environmental impact of engineered silver nanoparticles, their characterization from environmental matrices should be pursued, yet no field-scale studies are available to date. A new research report was motivated by the fact that silver nanoparticles in consumer products are likely being released during and/or after the product's lifetime. The silver nanoparticles will likely get into wastewater streams and subsequently enter wastewater treatment plants. During wastewater treatment processes, silver nanoparticles may be incorporated into the sewage sludge matrix and concentrated over time.
Silver nanoparticles are one of the most extensively used type of nanoparticles in consumer products due to the unique antibacterial activity of silver. There have been raising environmental concerns over their adverse ecological effects, along with ionic silver potentially released from the particles. To predict the environmental impact of engineered silver nanoparticles, their characterization from environmental matrices should be pursued, yet no field-scale studies are available to date. A new research report was motivated by the fact that silver nanoparticles in consumer products are likely being released during and/or after the product's lifetime. The silver nanoparticles will likely get into wastewater streams and subsequently enter wastewater treatment plants. During wastewater treatment processes, silver nanoparticles may be incorporated into the sewage sludge matrix and concentrated over time.
Oct 4th, 2010
 Owing to their large surface area, strong infrared photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms. However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies. Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries. In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages.
Owing to their large surface area, strong infrared photoluminescence and magnetic properties, nanodiamonds are promising for various biomedical applications, including as drug/gene carriers and alternatives to the current bio-imaging platforms. However, the biomedical applications will hardly be realized unless the potential hazards of nanodiamonds to humans and other biological systems are ascertained. The biocompatibility of nanodiamonds at the cellular level has been confirmed by many independent studies. Following these earlier cytotoxicity studies, many groups have used nanodiamonds and their functionalized derivatives for drug/gene deliveries. In spite of the earlier reports that nanodiamonds are biocompatible at the cellular level, researchers have now demonstrated in a new study that nanodiamonds can activate DNA repair proteins in embryonic stem cells, suggesting possible DNA damages. 
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





