Showing Spotlights 41 - 48 of 235 in category All (newest first):
 Conventional skin electronics are fixed designs that lack consideration for the individual characteristics of the wearer as well as the ability to actively adapt to various user specifications. In order to manage different sensor specifications and body impedances, design adjustments are inevitable during fabrication of these devices. To address this issue, researchers have developed a real-time fabrication platform that can adapt to arbitrary environments by freely drawing or erasing paths in a printed circuit.
Conventional skin electronics are fixed designs that lack consideration for the individual characteristics of the wearer as well as the ability to actively adapt to various user specifications. In order to manage different sensor specifications and body impedances, design adjustments are inevitable during fabrication of these devices. To address this issue, researchers have developed a real-time fabrication platform that can adapt to arbitrary environments by freely drawing or erasing paths in a printed circuit.
Feb 3rd, 2022
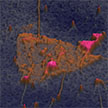 A novel multidimensional characterization approach that combines several characterization techniques, allowing to get materials properties of the heterostructure with (almost) nanometer resolution. The method combines several microscopy tools into a multidimensional imaging tool that allows the inspection of materials' optical properties at a resolution much higher than with regular optical microscopy. At the core of this method are nanometer-resolution optical maps that we obtained with scattering Scanning Near-field Optical Microscopy (sSNOM).
A novel multidimensional characterization approach that combines several characterization techniques, allowing to get materials properties of the heterostructure with (almost) nanometer resolution. The method combines several microscopy tools into a multidimensional imaging tool that allows the inspection of materials' optical properties at a resolution much higher than with regular optical microscopy. At the core of this method are nanometer-resolution optical maps that we obtained with scattering Scanning Near-field Optical Microscopy (sSNOM).
Jan 22nd, 2022
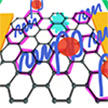 The use of graphene-based field-effect transistors is one of the most powerful biosensing units for the detection of numerous biological and biochemical analytes. In new work, researchers propose a microscopic model of mycotoxin detection via graphene-based aptasensors enabling improvement of biosensor sensitivity, which is based on the investigation of aptamer interaction mechanism with graphene channel during binding of targeted molecules. The limit of detection is estimated at only 1 pM and time response in just 10 seconds, making the developed platform a high-performance device.
The use of graphene-based field-effect transistors is one of the most powerful biosensing units for the detection of numerous biological and biochemical analytes. In new work, researchers propose a microscopic model of mycotoxin detection via graphene-based aptasensors enabling improvement of biosensor sensitivity, which is based on the investigation of aptamer interaction mechanism with graphene channel during binding of targeted molecules. The limit of detection is estimated at only 1 pM and time response in just 10 seconds, making the developed platform a high-performance device.
Dec 22nd, 2021
 Photodetectors with blackbody response show significant applications in remote sensing and infrared imaging. However, up to now, few works have demonstrated excellent response to blackbody radiation (weak irregular radiation from a real object), which is essential to reliably evaluate their potential in practical detections. Researchers now have demonstrated van der Waals unipolar barrier photodetectors with nBn and pBp heterostructures. Designing unipolar barriers with conventional materials is challenging due to the strict requirements of lattice and band matching.
Photodetectors with blackbody response show significant applications in remote sensing and infrared imaging. However, up to now, few works have demonstrated excellent response to blackbody radiation (weak irregular radiation from a real object), which is essential to reliably evaluate their potential in practical detections. Researchers now have demonstrated van der Waals unipolar barrier photodetectors with nBn and pBp heterostructures. Designing unipolar barriers with conventional materials is challenging due to the strict requirements of lattice and band matching.
Jun 24th, 2021
 Researchers have significantly enhanced the performance of a surface plasmon resonance (SPR) biosensing platform by adding an atomically thin phase change material to the sensor. This plasmonic sensing device exhibits a detection limit of 1 femtomol for tumor necrosis factor (TNF alpha) biomarkers , which is more than three orders of magnitude compared to current state-of-art nanomaterial-enhanced plasmonic sensors. It also detects small biotin molecules with a sensitivity of 10 femtomols. This sub-attomole detection level is a significant improvement compared to other SPR designs.
Researchers have significantly enhanced the performance of a surface plasmon resonance (SPR) biosensing platform by adding an atomically thin phase change material to the sensor. This plasmonic sensing device exhibits a detection limit of 1 femtomol for tumor necrosis factor (TNF alpha) biomarkers , which is more than three orders of magnitude compared to current state-of-art nanomaterial-enhanced plasmonic sensors. It also detects small biotin molecules with a sensitivity of 10 femtomols. This sub-attomole detection level is a significant improvement compared to other SPR designs.
Apr 19th, 2021
 Wearable flexible sensors have recently emerged as promising alternatives for human healthcare monitoring due to their lightweight and thin-film-based nature. However, few of these flexible sensor systems are applicable to neonatal monitoring, especially sleeping posture detection. Researchers now report a versatile laser-induced-graphene-based integrated sensor system, which can wirelessly monitor sleeping postures, respiration rate, and diaper wetness with feedback alarm functions.
Wearable flexible sensors have recently emerged as promising alternatives for human healthcare monitoring due to their lightweight and thin-film-based nature. However, few of these flexible sensor systems are applicable to neonatal monitoring, especially sleeping posture detection. Researchers now report a versatile laser-induced-graphene-based integrated sensor system, which can wirelessly monitor sleeping postures, respiration rate, and diaper wetness with feedback alarm functions.
Mar 31st, 2021
 Researchers demonstrate label-free chiral detection of metabolic molecules at picomolar level through microbubble-induced rapid accumulation of biomolecules on plasmonic chiral sensors, which shows a 10-million times enhancement in sensitivity comparing to state-of-the-art plasmonic chiral sensors. The researchers achieved their ultrahigh sensitivity in chiral sensing of biomolecules by utilizing two enhancement mechanisms: the microbubble-induced accumulation of biomolecules onto the chiral plasmonic substrates; and the subsequent plasmon-enhanced chiral sensing.
Researchers demonstrate label-free chiral detection of metabolic molecules at picomolar level through microbubble-induced rapid accumulation of biomolecules on plasmonic chiral sensors, which shows a 10-million times enhancement in sensitivity comparing to state-of-the-art plasmonic chiral sensors. The researchers achieved their ultrahigh sensitivity in chiral sensing of biomolecules by utilizing two enhancement mechanisms: the microbubble-induced accumulation of biomolecules onto the chiral plasmonic substrates; and the subsequent plasmon-enhanced chiral sensing.
Mar 30th, 2021
 Researchers have demonstrated the large-scale fabrication of skin-interfaced printed microfluidic patches, capable of multiplexed electrochemical detection of biomarkers from human sweat. This sensor continuously and simultaneously measures the level of biomarkers such as lactate, Na+, K+, and pH during sweating. The skin-worn sensor can selectively measure analytes even in the presence of other interfering species present in sweat. The patch is integrated with a custom-made miniature printed circuit board that enables multiplexed decoding of sweat and wireless signal transduction to the host devices and is completely free from any sensor-to-sensor transfer of signals.
Researchers have demonstrated the large-scale fabrication of skin-interfaced printed microfluidic patches, capable of multiplexed electrochemical detection of biomarkers from human sweat. This sensor continuously and simultaneously measures the level of biomarkers such as lactate, Na+, K+, and pH during sweating. The skin-worn sensor can selectively measure analytes even in the presence of other interfering species present in sweat. The patch is integrated with a custom-made miniature printed circuit board that enables multiplexed decoding of sweat and wireless signal transduction to the host devices and is completely free from any sensor-to-sensor transfer of signals.
Mar 29th, 2021
 Conventional skin electronics are fixed designs that lack consideration for the individual characteristics of the wearer as well as the ability to actively adapt to various user specifications. In order to manage different sensor specifications and body impedances, design adjustments are inevitable during fabrication of these devices. To address this issue, researchers have developed a real-time fabrication platform that can adapt to arbitrary environments by freely drawing or erasing paths in a printed circuit.
Conventional skin electronics are fixed designs that lack consideration for the individual characteristics of the wearer as well as the ability to actively adapt to various user specifications. In order to manage different sensor specifications and body impedances, design adjustments are inevitable during fabrication of these devices. To address this issue, researchers have developed a real-time fabrication platform that can adapt to arbitrary environments by freely drawing or erasing paths in a printed circuit.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





