Showing Spotlights 57 - 64 of 77 in category All (newest first):
 Borrowing from nature's micro- and nanoscale propulsion systems, nanotechnology researchers have successfully used motor proteins to transport nanosized cargo in molecular sorting and nano-assembly devices. In so-called gliding assays, surface-attached motors propel cytoskeletal filaments, which in turn transport a cargo. However, cargo and motors both attach to the filament lattice and will affect each other. While an effect of cargo loading on transport speed has been described before, it has never been explained very well. To study this effect, scientists in Germany have observed single kinesin-1 molecules on streptavidin coated microtubules. They found that individual kinesin-1 motors frequently stopped upon encounters with attached streptavidin molecules. This work helps to understand the interactions of kinesin-1 and obstacles on the microtubule surface. An interesting, possibly even more important side result is that this understanding will not only help to optimize transport assays, balancing speed and cargo-loading, but can be used as a novel method for the detection of proteins as well.
Borrowing from nature's micro- and nanoscale propulsion systems, nanotechnology researchers have successfully used motor proteins to transport nanosized cargo in molecular sorting and nano-assembly devices. In so-called gliding assays, surface-attached motors propel cytoskeletal filaments, which in turn transport a cargo. However, cargo and motors both attach to the filament lattice and will affect each other. While an effect of cargo loading on transport speed has been described before, it has never been explained very well. To study this effect, scientists in Germany have observed single kinesin-1 molecules on streptavidin coated microtubules. They found that individual kinesin-1 motors frequently stopped upon encounters with attached streptavidin molecules. This work helps to understand the interactions of kinesin-1 and obstacles on the microtubule surface. An interesting, possibly even more important side result is that this understanding will not only help to optimize transport assays, balancing speed and cargo-loading, but can be used as a novel method for the detection of proteins as well.
Sep 4th, 2008
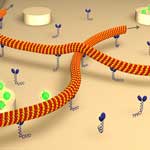 The motor proteins of the cytoskeleton accomplish nanotransport tasks by moving 'cargo' along microtubules that are about 25 nm wide but can grow up to 1,000 times as long. Nanotechnology engineers are fascinated by this transport mechanism and several efforts are underway in various labs to unravel and, researchers hope, eventually copy nature's engineering feat. A particularly promising setup consists of surface-attached linear motor proteins that drive the motion of cytoskeletal filaments. Some researchers expect that artificial molecular transport systems which utilize microtubules motility will be an alternative to pressure-driven or electrokinetic flow-based microfluidic devices. Researchers in Germany describe a novel method to characterize the rotational movement of cytoskeletal filaments gliding over motor-coated substrate surfaces. This technique allows exploring the detailed paths that motors take on cytoskeletal filaments. This is also important in understanding situations of heavy intracellular traffic, where motors might have to switch lanes.
The motor proteins of the cytoskeleton accomplish nanotransport tasks by moving 'cargo' along microtubules that are about 25 nm wide but can grow up to 1,000 times as long. Nanotechnology engineers are fascinated by this transport mechanism and several efforts are underway in various labs to unravel and, researchers hope, eventually copy nature's engineering feat. A particularly promising setup consists of surface-attached linear motor proteins that drive the motion of cytoskeletal filaments. Some researchers expect that artificial molecular transport systems which utilize microtubules motility will be an alternative to pressure-driven or electrokinetic flow-based microfluidic devices. Researchers in Germany describe a novel method to characterize the rotational movement of cytoskeletal filaments gliding over motor-coated substrate surfaces. This technique allows exploring the detailed paths that motors take on cytoskeletal filaments. This is also important in understanding situations of heavy intracellular traffic, where motors might have to switch lanes.
Aug 29th, 2008
 A fast-growing body of nanotechnology research is dedicated to nanoscale motors and molecular machinery. The results of these studies are spectacular: well-designed molecules or supramolecules show various movements upon exposure to various stimuli, such as molecular shuttles, molecular elevators and molecular motors. So far, however, nobody has been able to directly observe the movements of these molecular machines and utilize the mechanical work done by them. Now, an international group of researchers have succeeded in amplifying the minuscule change in structures at a molecular level caused by an external stimulus (light) to a macroscopic change through a cooperative effect of liquid crystals. Using liquid-crystalline elastomers (LCEs) ? unique materials having both properties of liquid crystals (LCs) and elastomers ? the scientists have successfully developed new photomechanical devices, including the first light-driven plastic motor. In other words, with this novel material the energy from light can be directly converted into mechanical work without the aid of batteries, electric wires, or gears.
A fast-growing body of nanotechnology research is dedicated to nanoscale motors and molecular machinery. The results of these studies are spectacular: well-designed molecules or supramolecules show various movements upon exposure to various stimuli, such as molecular shuttles, molecular elevators and molecular motors. So far, however, nobody has been able to directly observe the movements of these molecular machines and utilize the mechanical work done by them. Now, an international group of researchers have succeeded in amplifying the minuscule change in structures at a molecular level caused by an external stimulus (light) to a macroscopic change through a cooperative effect of liquid crystals. Using liquid-crystalline elastomers (LCEs) ? unique materials having both properties of liquid crystals (LCs) and elastomers ? the scientists have successfully developed new photomechanical devices, including the first light-driven plastic motor. In other words, with this novel material the energy from light can be directly converted into mechanical work without the aid of batteries, electric wires, or gears.
Jun 20th, 2008
 The concept of a 'machine' - a mechanical or electrical device that transmits or modifies energy to perform a certain task - can be extended to the nano world as well. On the nanoscale, the nanomachine components would be molecular structures each designed to perform a specific task which, all taken together, would result in a complex function. Nanoscientists have already built molecular motors, wheels, and gears for powering nanomachines. The ability to control nanoscale motors, more specifically, to control the motion of molecular components of such motors, doesn't only involve acceleration and movement but, equally important, deceleration and stopping. So far, the development of a practical braking system for nanomotors remains a challenge. Researchers in Taiwan now have reported development of a light-driven molecular brake that could provide on-demand stopping power for futuristic nanotechnology machines.
The concept of a 'machine' - a mechanical or electrical device that transmits or modifies energy to perform a certain task - can be extended to the nano world as well. On the nanoscale, the nanomachine components would be molecular structures each designed to perform a specific task which, all taken together, would result in a complex function. Nanoscientists have already built molecular motors, wheels, and gears for powering nanomachines. The ability to control nanoscale motors, more specifically, to control the motion of molecular components of such motors, doesn't only involve acceleration and movement but, equally important, deceleration and stopping. So far, the development of a practical braking system for nanomotors remains a challenge. Researchers in Taiwan now have reported development of a light-driven molecular brake that could provide on-demand stopping power for futuristic nanotechnology machines.
Jun 4th, 2008
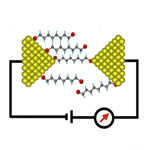 One of the many fascinating concepts in nanotechnology is the vision of molecular electronics where tomorrow's engineers might use individual molecules to perform the functions in an electronic circuit that are performed by semiconductor devices today. This is just another example of scientists taking a cue from nature's playbook, where essentially all electronic processes, from photosynthesis to signal transduction, occur in molecular structures. The basic science on which molecular electronics technology would be built is now unfolding but researchers are still struggling with the most basic requirements for molecular electronics, for instance, how to precisely position individual molecules on a surface or how to reliably measure the resistance of a single molecule. A tremendous amount of painstaking work goes into developing the kind of ultraprecise and ultrasensitive instruments that are required to develop electronics at the nanoscale. A recent example is a new device for measuring the conductance values of single-molecule junctions which are covalently bound to two electrodes.
One of the many fascinating concepts in nanotechnology is the vision of molecular electronics where tomorrow's engineers might use individual molecules to perform the functions in an electronic circuit that are performed by semiconductor devices today. This is just another example of scientists taking a cue from nature's playbook, where essentially all electronic processes, from photosynthesis to signal transduction, occur in molecular structures. The basic science on which molecular electronics technology would be built is now unfolding but researchers are still struggling with the most basic requirements for molecular electronics, for instance, how to precisely position individual molecules on a surface or how to reliably measure the resistance of a single molecule. A tremendous amount of painstaking work goes into developing the kind of ultraprecise and ultrasensitive instruments that are required to develop electronics at the nanoscale. A recent example is a new device for measuring the conductance values of single-molecule junctions which are covalently bound to two electrodes.
May 27th, 2008
 The catalytic conversion of chemical to mechanical energy is ubiquitous in biology, powering such important and diverse processes as cell division, skeletal muscle movement, protein synthesis, and transport of cargo within cells. Catalytic 'engines' will be key components of active micron- and sub-micron scale systems for controlled movement, particle assembly, and separations. A few days ago we took a look at catalytic nanomotors - sophisticated molecular-size biomotors have evolved in nature - and described an example where researchers supercharged their nanomotors by inserting carbon nanotubes into the gold and platinum nanowires (Speeding up catalytic nanomotors with carbon nanotubes). Today we show an example where catalytic nanomotors can, in principle, be tethered or coupled to other objects to act as the engines of nanoscale assemblies. Additionally, an object that moves by generating a continuous surface force in a fluid can, in principle, be used to pump the fluid by the same catalytic mechanism. Thus, by immobilizing these nanomotors, a group of scientists have developed micro/nanofluidic pumps that transduce energy catalytically.
The catalytic conversion of chemical to mechanical energy is ubiquitous in biology, powering such important and diverse processes as cell division, skeletal muscle movement, protein synthesis, and transport of cargo within cells. Catalytic 'engines' will be key components of active micron- and sub-micron scale systems for controlled movement, particle assembly, and separations. A few days ago we took a look at catalytic nanomotors - sophisticated molecular-size biomotors have evolved in nature - and described an example where researchers supercharged their nanomotors by inserting carbon nanotubes into the gold and platinum nanowires (Speeding up catalytic nanomotors with carbon nanotubes). Today we show an example where catalytic nanomotors can, in principle, be tethered or coupled to other objects to act as the engines of nanoscale assemblies. Additionally, an object that moves by generating a continuous surface force in a fluid can, in principle, be used to pump the fluid by the same catalytic mechanism. Thus, by immobilizing these nanomotors, a group of scientists have developed micro/nanofluidic pumps that transduce energy catalytically.
May 7th, 2008
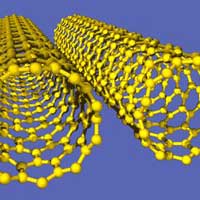 Sophisticated molecular-size motors have evolved in nature, where they are used in virtually every important biological process. In contrast, the development of synthetic nanomotors that mimic the function of these amazing natural systems and could be used in man-made nanodevices is in its infancy. Building nanoscale motors is not just an exercise in scaling down the design of a macroworld engine to nanoscale dimensions. Many factors such as friction, heat dissipation and many other mechanical behaviors are just very different at this scale - everything is constantly moving (under kinetic energy supplied by the heat of the surroundings) and being buffeted by other atoms and molecules (Brownian motion). In nature, biological motors use catalytic reactions to create forces based on chemical changes. These motors do not require external energy sources such as electric or magnetic fields. Instead, the input energy is supplied locally and chemically. Despite impressive progress over the past years, man-made nanomachines lack the efficiency and speed of their biological counterparts. New research has demonstrated that the incorporation of carbon nanotubes (CNT) into the platinum component of asymmetric metal nanowire motors leads to dramatically accelerated movement in hydrogen peroxide solutions, with average speeds of 50-60 micrometers per second.
Sophisticated molecular-size motors have evolved in nature, where they are used in virtually every important biological process. In contrast, the development of synthetic nanomotors that mimic the function of these amazing natural systems and could be used in man-made nanodevices is in its infancy. Building nanoscale motors is not just an exercise in scaling down the design of a macroworld engine to nanoscale dimensions. Many factors such as friction, heat dissipation and many other mechanical behaviors are just very different at this scale - everything is constantly moving (under kinetic energy supplied by the heat of the surroundings) and being buffeted by other atoms and molecules (Brownian motion). In nature, biological motors use catalytic reactions to create forces based on chemical changes. These motors do not require external energy sources such as electric or magnetic fields. Instead, the input energy is supplied locally and chemically. Despite impressive progress over the past years, man-made nanomachines lack the efficiency and speed of their biological counterparts. New research has demonstrated that the incorporation of carbon nanotubes (CNT) into the platinum component of asymmetric metal nanowire motors leads to dramatically accelerated movement in hydrogen peroxide solutions, with average speeds of 50-60 micrometers per second.
May 1st, 2008
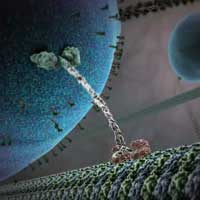 In case you haven't seen the absolutely amazing animation 'Cellular Visions: The Inner Life of a Cell' yet, go watch it now. In it, there is a sequence where a motor protein is sort of 'walking' along a filament, dragging this round sphere of lipids behind it. This kind of nanoscale biological motor is able to load/unload particular types of cargo without external stimuli, and transport them along cytoskeletal filaments by using the energy of adenosine triphosphate (ATP) hydrolysis within cells. Nanotechnology researchers are fascinated by the various molecular delivery systems that have evolved in nature and they are receiving increasing attention as blueprints for nanoscale actuators and building blocks to construct artificially-engineered bio-hybrid systems. Some researchers expect that artificial molecular transport systems which utilize microtubules motility will be an alternative way to pressure-driven or electrokinetic flow-based microfluidic devices. Researchers in Japan propose a molecular transport system that can achieve autonomous loading/unloading of specified cargoes. This system loads a cargo molecule through DNA hybridization.
In case you haven't seen the absolutely amazing animation 'Cellular Visions: The Inner Life of a Cell' yet, go watch it now. In it, there is a sequence where a motor protein is sort of 'walking' along a filament, dragging this round sphere of lipids behind it. This kind of nanoscale biological motor is able to load/unload particular types of cargo without external stimuli, and transport them along cytoskeletal filaments by using the energy of adenosine triphosphate (ATP) hydrolysis within cells. Nanotechnology researchers are fascinated by the various molecular delivery systems that have evolved in nature and they are receiving increasing attention as blueprints for nanoscale actuators and building blocks to construct artificially-engineered bio-hybrid systems. Some researchers expect that artificial molecular transport systems which utilize microtubules motility will be an alternative way to pressure-driven or electrokinetic flow-based microfluidic devices. Researchers in Japan propose a molecular transport system that can achieve autonomous loading/unloading of specified cargoes. This system loads a cargo molecule through DNA hybridization.
Apr 28th, 2008
 Borrowing from nature's micro- and nanoscale propulsion systems, nanotechnology researchers have successfully used motor proteins to transport nanosized cargo in molecular sorting and nano-assembly devices. In so-called gliding assays, surface-attached motors propel cytoskeletal filaments, which in turn transport a cargo. However, cargo and motors both attach to the filament lattice and will affect each other. While an effect of cargo loading on transport speed has been described before, it has never been explained very well. To study this effect, scientists in Germany have observed single kinesin-1 molecules on streptavidin coated microtubules. They found that individual kinesin-1 motors frequently stopped upon encounters with attached streptavidin molecules. This work helps to understand the interactions of kinesin-1 and obstacles on the microtubule surface. An interesting, possibly even more important side result is that this understanding will not only help to optimize transport assays, balancing speed and cargo-loading, but can be used as a novel method for the detection of proteins as well.
Borrowing from nature's micro- and nanoscale propulsion systems, nanotechnology researchers have successfully used motor proteins to transport nanosized cargo in molecular sorting and nano-assembly devices. In so-called gliding assays, surface-attached motors propel cytoskeletal filaments, which in turn transport a cargo. However, cargo and motors both attach to the filament lattice and will affect each other. While an effect of cargo loading on transport speed has been described before, it has never been explained very well. To study this effect, scientists in Germany have observed single kinesin-1 molecules on streptavidin coated microtubules. They found that individual kinesin-1 motors frequently stopped upon encounters with attached streptavidin molecules. This work helps to understand the interactions of kinesin-1 and obstacles on the microtubule surface. An interesting, possibly even more important side result is that this understanding will not only help to optimize transport assays, balancing speed and cargo-loading, but can be used as a novel method for the detection of proteins as well.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





