Showing Spotlights 57 - 64 of 199 in category All (newest first):
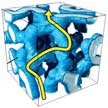 Finding low-cost solid materials capable of efficiently and safely replacing liquid electrolytes in lithium-ion batteries has been a considerable research interest over the past years. Of the various types of solid electrolytes that have been developed so far, composite polymer electrolytes exhibit acceptable Li-ion conductivity due to the interaction between nanofillers and polymer. By fabricating a pre-percolated network of ceramic filler instead of distributing particles in polymer, a 3D interconnected ceramic framework provides continuous pathways for ion conduction. This novel method will help to develop composite materials in a different but much improved way than conventional particle distributions.
Finding low-cost solid materials capable of efficiently and safely replacing liquid electrolytes in lithium-ion batteries has been a considerable research interest over the past years. Of the various types of solid electrolytes that have been developed so far, composite polymer electrolytes exhibit acceptable Li-ion conductivity due to the interaction between nanofillers and polymer. By fabricating a pre-percolated network of ceramic filler instead of distributing particles in polymer, a 3D interconnected ceramic framework provides continuous pathways for ion conduction. This novel method will help to develop composite materials in a different but much improved way than conventional particle distributions.
Feb 28th, 2018
 Scientists report a systematic study involving theoretical and experimental approaches to evaluate the Li-ion storage capability in 2D atomic sheets of nonlayered MoO2. They describe a new process - polymer-assisted reduction - to make atomically flat 2D sheets of MoO2. Since MoO2 is not a 2D material, this process opens a new pathway to make 2D nanostructures from non-layered materials. By successfully making 2D atomic sheets of MoO2, the researchers fabricated a battery electrode in which the Li ion diffusion and electron transport are exceptionally fast.
Scientists report a systematic study involving theoretical and experimental approaches to evaluate the Li-ion storage capability in 2D atomic sheets of nonlayered MoO2. They describe a new process - polymer-assisted reduction - to make atomically flat 2D sheets of MoO2. Since MoO2 is not a 2D material, this process opens a new pathway to make 2D nanostructures from non-layered materials. By successfully making 2D atomic sheets of MoO2, the researchers fabricated a battery electrode in which the Li ion diffusion and electron transport are exceptionally fast.
Feb 15th, 2018
 Inspired by the designs printed on T-shirts, researchers recently reported a new class of wearable power sources. To explore the feasibility of power sources directly printed on cotton T-shirts, which look like letters or symbols, they chose electric double layer supercapacitors based on activated carbon materials as a model electrochemical system. These T-shirts look and behave like a normal T-shirt but feature printed supercapacitors in the shape of letters and symbols.
Inspired by the designs printed on T-shirts, researchers recently reported a new class of wearable power sources. To explore the feasibility of power sources directly printed on cotton T-shirts, which look like letters or symbols, they chose electric double layer supercapacitors based on activated carbon materials as a model electrochemical system. These T-shirts look and behave like a normal T-shirt but feature printed supercapacitors in the shape of letters and symbols.
Jan 19th, 2018
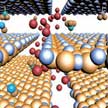 Motived by the large number of non-layered materials, e.g. transition metal oxides, which hold great promise in battery applications, scientists aim to extend the concept of nanofluidic channels into intrinsically non-layered materials and improve their electrochemical properties. In a new study, researchers have developed a Co3O4-based two-dimensional nano-architecture possessing nanofluidic channels with specially designed interlayer characteristics for fast lithium ion transport, leading to exceptional performance in lithium ion batteries ever reported for this material.
Motived by the large number of non-layered materials, e.g. transition metal oxides, which hold great promise in battery applications, scientists aim to extend the concept of nanofluidic channels into intrinsically non-layered materials and improve their electrochemical properties. In a new study, researchers have developed a Co3O4-based two-dimensional nano-architecture possessing nanofluidic channels with specially designed interlayer characteristics for fast lithium ion transport, leading to exceptional performance in lithium ion batteries ever reported for this material.
Dec 8th, 2017
 So far, most of the developed self-powered piezoelectric devices are rigid or have limited lateral stretchability and could not be used to harvest energy from lateral strain, which greatly limits their applications on large strain deformation. In new work, researchers have successfully fabricated a piezoelectric nanocomposite device with good transparency, high stretchability, and self-powered sensing characteristics. Attached to the human body, it can harvest biomechanical energy and monitor physiological signals.
So far, most of the developed self-powered piezoelectric devices are rigid or have limited lateral stretchability and could not be used to harvest energy from lateral strain, which greatly limits their applications on large strain deformation. In new work, researchers have successfully fabricated a piezoelectric nanocomposite device with good transparency, high stretchability, and self-powered sensing characteristics. Attached to the human body, it can harvest biomechanical energy and monitor physiological signals.
Dec 4th, 2017
 The wearable power sources required for wearable and implantable electronic devices are limited by the size of the gadgets they power. Microsupercapacitors are newly emerging miniaturized high-power microelectrochemical energy-storage devices that can deliver high power density, fast charge and discharge, and a superior cycling lifetime. A new study shows that electrode fractal design is a viable strategy for improving the performance of integrated microsupercapacitors that use thin-film electrodes at no extra processing or fabrication cost.
The wearable power sources required for wearable and implantable electronic devices are limited by the size of the gadgets they power. Microsupercapacitors are newly emerging miniaturized high-power microelectrochemical energy-storage devices that can deliver high power density, fast charge and discharge, and a superior cycling lifetime. A new study shows that electrode fractal design is a viable strategy for improving the performance of integrated microsupercapacitors that use thin-film electrodes at no extra processing or fabrication cost.
Nov 20th, 2017
 Self-powered nanotechnology based on one type of nanogenerators - piezoelectric nanogenerators - aims at powering nanodevices and nanosystems using the energy harvested from the environment in which these systems are suppose to operate. This offers a completely new approach for harvesting mechanical energy using organic and inorganic materials. Researchers have now reported a novel bio-piezoelectric nanogenerator using naturally abundant, self-aligned cellulose fibrous untreated onion skin as efficient piezoelectric material.
Self-powered nanotechnology based on one type of nanogenerators - piezoelectric nanogenerators - aims at powering nanodevices and nanosystems using the energy harvested from the environment in which these systems are suppose to operate. This offers a completely new approach for harvesting mechanical energy using organic and inorganic materials. Researchers have now reported a novel bio-piezoelectric nanogenerator using naturally abundant, self-aligned cellulose fibrous untreated onion skin as efficient piezoelectric material.
Nov 15th, 2017
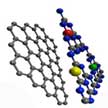 Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Nov 13th, 2017
 Finding low-cost solid materials capable of efficiently and safely replacing liquid electrolytes in lithium-ion batteries has been a considerable research interest over the past years. Of the various types of solid electrolytes that have been developed so far, composite polymer electrolytes exhibit acceptable Li-ion conductivity due to the interaction between nanofillers and polymer. By fabricating a pre-percolated network of ceramic filler instead of distributing particles in polymer, a 3D interconnected ceramic framework provides continuous pathways for ion conduction. This novel method will help to develop composite materials in a different but much improved way than conventional particle distributions.
Finding low-cost solid materials capable of efficiently and safely replacing liquid electrolytes in lithium-ion batteries has been a considerable research interest over the past years. Of the various types of solid electrolytes that have been developed so far, composite polymer electrolytes exhibit acceptable Li-ion conductivity due to the interaction between nanofillers and polymer. By fabricating a pre-percolated network of ceramic filler instead of distributing particles in polymer, a 3D interconnected ceramic framework provides continuous pathways for ion conduction. This novel method will help to develop composite materials in a different but much improved way than conventional particle distributions.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





