Showing Spotlights 65 - 72 of 200 in category All (newest first):
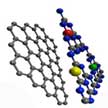 Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Nov 13th, 2017
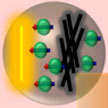 Chemical engineering researchers have reported the usage of activated carbon prepared from tea leaves, improving the mass transport phenomenon (33 % performance improvement) in an operating direct methanol fuel cell, owing to its pore structure characteristics. The cell performance underwent drastic changes in the mass transport region of the fuel cell polarization curve, comparable to the standard membrane electrode assembly. This is attributed to the pore structure of this framework aiding in enhanced water removal, as a result more air molecules react with the platinum catalyst sites finally improving the fuel cell performance.
Chemical engineering researchers have reported the usage of activated carbon prepared from tea leaves, improving the mass transport phenomenon (33 % performance improvement) in an operating direct methanol fuel cell, owing to its pore structure characteristics. The cell performance underwent drastic changes in the mass transport region of the fuel cell polarization curve, comparable to the standard membrane electrode assembly. This is attributed to the pore structure of this framework aiding in enhanced water removal, as a result more air molecules react with the platinum catalyst sites finally improving the fuel cell performance.
Oct 3rd, 2017
 Electrocatalysis offers important opportunities for clean fuel production, but uncovering the chemistry at the electrode surface remains a challenge. Here, this work exploits a single-nanosheet device to perform in-situ measurements of water oxidation electrocatalysis and reveal a crucial interaction with oxygen. The obtained in-depth understanding could provide valuable clues for catalysis system design and the in-situ measurement could be also useful to analyze other interfacial reaction processes.
Electrocatalysis offers important opportunities for clean fuel production, but uncovering the chemistry at the electrode surface remains a challenge. Here, this work exploits a single-nanosheet device to perform in-situ measurements of water oxidation electrocatalysis and reveal a crucial interaction with oxygen. The obtained in-depth understanding could provide valuable clues for catalysis system design and the in-situ measurement could be also useful to analyze other interfacial reaction processes.
Sep 29th, 2017
 Two-dimensional (2D) energy materials have outstanding physical and chemical properties in contrast to their bulk counterparts. This is particularly true for charge storage devices such as lithium-ion batteries and supercapacitors. Unfortunately, when directly applying these 2D nanostructured materials for energy storage, there is still a significant challenge as they may have serious self-restacking leading to decreased active surface areas and sluggish ion transport kinetics. Researchers have now developed an effective interlayer engineering strategy to improve sodium ion transport in 2D nanosheets via controlled organic intercalation.
Two-dimensional (2D) energy materials have outstanding physical and chemical properties in contrast to their bulk counterparts. This is particularly true for charge storage devices such as lithium-ion batteries and supercapacitors. Unfortunately, when directly applying these 2D nanostructured materials for energy storage, there is still a significant challenge as they may have serious self-restacking leading to decreased active surface areas and sluggish ion transport kinetics. Researchers have now developed an effective interlayer engineering strategy to improve sodium ion transport in 2D nanosheets via controlled organic intercalation.
Sep 13th, 2017
 Sodium-ion battery, as an emerging battery technology beyond lithium-ion battery, has attracted great research interests recent years. Sodium-ion batteries have a similar configuration and electrochemical reaction processes with lithium-ion batteries. But the Na resources are much more abundant and cost-effective than Li resources, which makes sodium-ion batteries highly promising as next-generation energy storage devices, especially for large-scale energy storage. However, the practical application of Na-ion batteries is still not currently realized.
Sodium-ion battery, as an emerging battery technology beyond lithium-ion battery, has attracted great research interests recent years. Sodium-ion batteries have a similar configuration and electrochemical reaction processes with lithium-ion batteries. But the Na resources are much more abundant and cost-effective than Li resources, which makes sodium-ion batteries highly promising as next-generation energy storage devices, especially for large-scale energy storage. However, the practical application of Na-ion batteries is still not currently realized.
Sep 12th, 2017
 Wearable energy harvesters are greatly attractive and receive intensive research efforts in recent years, aiming at powering various emerging flexible and wearable electronics to meet the requirements of smart fabrics, motion tracking and health monitoring. Researchers now have developed a coating based on cellulose-derived hydrophobic nanoparticles and demonstrated its application as a wearable water triboelectric generator that harvests energy from water flow. This innovative fabric-based TEG has self-cleaning and antifouling properties.
Wearable energy harvesters are greatly attractive and receive intensive research efforts in recent years, aiming at powering various emerging flexible and wearable electronics to meet the requirements of smart fabrics, motion tracking and health monitoring. Researchers now have developed a coating based on cellulose-derived hydrophobic nanoparticles and demonstrated its application as a wearable water triboelectric generator that harvests energy from water flow. This innovative fabric-based TEG has self-cleaning and antifouling properties.
Aug 7th, 2017
 Graphene currently is the most studied material on the planet - this is especially true for charge storage and the results from many laboratories confirm its potential to change today's energy-storage landscape. Specifically, graphene could present several new features for energy-storage devices, such as smaller capacitors, completely flexible and even rollable energy-storage devices, transparent batteries, and high-capacity and fast-charging devices.
Graphene currently is the most studied material on the planet - this is especially true for charge storage and the results from many laboratories confirm its potential to change today's energy-storage landscape. Specifically, graphene could present several new features for energy-storage devices, such as smaller capacitors, completely flexible and even rollable energy-storage devices, transparent batteries, and high-capacity and fast-charging devices.
Jul 20th, 2017
 Developing highly active electrocatalysts for photoelectrochemical water splitting is critical to bringing solar/electrical-to-hydrogen energy conversion processes into reality. Researchers have developed a novel 3D hierarchical hybrid electrocatalyst grown on electrochemically exfoliated graphene. The researchers then further integrated the hybrid nanosheets with a macroporous silicon photocathode, and the results show that it can enable highly active solar-driven photoelectrochemical water splitting in both basic media and real river water.
Developing highly active electrocatalysts for photoelectrochemical water splitting is critical to bringing solar/electrical-to-hydrogen energy conversion processes into reality. Researchers have developed a novel 3D hierarchical hybrid electrocatalyst grown on electrochemically exfoliated graphene. The researchers then further integrated the hybrid nanosheets with a macroporous silicon photocathode, and the results show that it can enable highly active solar-driven photoelectrochemical water splitting in both basic media and real river water.
Jul 20th, 2017
 Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
Efficient electrocatalysts lie at the heart of a series of significant energy conversion and storage technologies, and atomically precise understanding of the influences of component dopants is crucial for looking into the reaction mechanism and controlled synthesis of the desired electrocatalysts. Graphitic carbon nitride is a promising electrocatalytic material owing to its intrinsically high N content and abundant edge sites. This material has been researched towards some of the most significant electrocatalytic reactions including oxygen reduction/evolution reaction and hydro evolution reaction. New work has comprehensively explored the influences of component elements within graphitic carbon nitride motiety for electrocatalytic reactions.
 Subscribe to our Nanotechnology Spotlight feed
Subscribe to our Nanotechnology Spotlight feed





