| Posted: Jul 13, 2015 |
Heat buckyballs to help environment
|
|
(Nanowerk News) Rice University scientists are forging toward tunable carbon-capture materials with a new study that shows how chemical changes affect the abilities of enhanced buckyballs to confine greenhouse gases.
|
|
The lab of Rice chemist Andrew Barron found last year that carbon-60 molecules (aka buckyballs, discovered at Rice in the 1980s) gain the ability to sequester carbon dioxide when combined with a polymer known as polyethyleneimine (PEI).
Enrico Andreoli, left, and Andrew Barron of Rice University are studying the use of enhanced carbon-60 molecules to capture carbon dioxide that would otherwise be released as a greenhouse gas.
|
|
Enrico Andreoli, left, and Andrew Barron of Rice University are studying the use of enhanced carbon-60 molecules to capture carbon dioxide that would otherwise be released as a greenhouse gas. Courtesy of the Welsh Government
|
|
Two critical questions – how and how well – are addressed in a new paper in the American Chemical Society journal Energy and Fuels ("Correlating Carbon Dioxide Capture and Chemical Changes in Pyrolyzed Polyethylenimine-C60").
|
 |
|
The amine-rich combination of C60 and PEI showed its potential in the previous study to capture emissions of carbon dioxide, a greenhouse gas, from such sources as industrial flue gases and natural-gas wells.
|
|
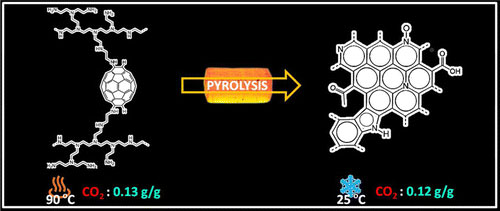
In the new study, the researchers found pyrolyzing the material – heating it in an oxygen-free environment – changes its chemical composition in ways that may someday be used to tune what the scientists call PEI-C60 for specific carbon-capture applications.
|
|
“One of the things we wanted to see is at what point, chemically, it converts from being something that absorbed best at high temperature to something that absorbed best at low temperature,” Barron said. “In other words, at what point does the chemistry change from one to the other?”
|
|
Lead author Enrico Andreoli pyrolyzed PEI-C60 in argon at various temperatures from 100 to 1,000 degrees Celsius (212 to 1,832 degrees Fahrenheit) and then evaluated each batch for carbon uptake.
|
|
He discovered the existence of a transition point at 200 C, a boundary between the material’s ability to soak in carbon dioxide through chemical means as opposed to physical absorption.
|
|
The material that was pyrolyzed at low temperatures became gooey and failed at pulling in carbon from high-temperature sources by chemical means. The opposite was true for PEI-C60 pyrolyzed at high heat. The now-porous, brittle material became better in low-temperature environments, physically soaking up carbon dioxide molecules.
|
|
At 200 C, they found the heat treatment breaks the polymer’s carbon-nitrogen bonds, leading to a drastic decrease in carbon capture by any means.
|
|
“One of the goals was to see if can we make this a little less gooey and still have chemical uptake, and the answer is, not really,” Barron said. “It flips from one process to the other. But this does give us a nice continuum of how to get from one to the other.”
|
|
Andreoli found that at its peak, untreated PEI-C60 absorbed more than a 10th of its weight in carbon dioxide at high temperatures (0.13 grams per gram of material at 90 C). Pyrolyzed PEI-C60 did nearly as well at low temperatures (0.12 grams at 25 C).
|
|
The researchers, with an eye on potential environmental benefits, continue to refine their process. “This has definitely pointed us in the right direction,” Barron said.
|

