| Posted: Aug 01, 2017 |
Foldable like an accordion: Bending individual nanostructures
(Nanowerk News) Since a research group at Kiel University (CAU) and the Hamburg University of Technology (TUHH) in Hamburg-Harburg has developed aerographite – one of the most light weight materials in the world – in the year 2012 -, they have continued researching about it. Its complex tetrapodal architecture gives the carbon-based 3D material very unique properties, such as extremely high elasticity and electrical conductivity.
|
|
Now, for the first time, as part of an international research team, materials scientists from the CAU were able to fold the individual hollow tetrapods, each measuring only a few micrometers in size.
|
 |
| The black aerographite is the lightest material in the world. It is constructed from tiny tetrapod structures. (Image: Siekmann/CAU)
|
|
After bending, the tetrapods automatically retain its original shape, without suffering any damage. This makes advanced applications conceivable, both in materials science as well as in the field of regenerative medicine. The research team published their results in Nature Communications ("Nanomechanics of individual aerographite tetrapods").
|
|
Regarding new materials, scientists are primarily interested in one thing: What properties do they have, and how do they behave under different conditions? This also determines the new possible uses of the materials.
|
|
"In order to predict the overall mechanical behaviour of a network material, we must investigate the individual building block structures with which it is constructed," explained Dr. Yogendra Mishra, materials scientist in the working group “Functional Nanomaterials” at the CAU.
|
|
Aerographite is constructed of tetrapods, carbon-based 3D nanostructure which consist of four hollow arms. When combined together, they form a porous, extremely lightweight network, and bring the weight of aerographite down to just 0.2 milligrams per cubic centimetre.
|
|
"Because of this unique structure, the material exhibits a high mechanical strength as well as a very high surface, from which interesting physical and chemical features originate," says Daria Smazna, a doctoral student in the project.
|
|
The international research team led from Kiel has now managed to show that aerographite is extremely foldable.
|
 |
| A network of four-armed carbon tetrapods interconnects to form the highly-porous material aerographite. (Image: Adelung Group) (click on image to enlarge)
|
|
"In general, bulk materials like carbon or metal are not foldable, but due its special structure our carbon networks are highly flexible and mechanical stable too”, explained Professor Rainer Adelung, head of the Functional Nanomaterials Chair. You could imagine it much like a sheet of paper. "A flat sheet of paper offers no resistance, if you hold it on one side, it simply hangs down. However, if we roll it up or crumple, it attains a certain degree of stability," continued the materials scientist.
|
|
It therefore depends on the geometrical arrangement within the material. The special shape of the tetrapods made the researchers suspect that they could be folded - despite the lightness of aerographite. This is because the individual arms have very thin walls and they are hollow inside.
|
|
"This allows them to be bent at so many different places, even reversibly. They automatically go back to their original shape, without sustaining any damage," explained Mishra. "Just like an accordion, the three-dimensional object can be folded into a two-dimensional form, and then unfolded again."
|
|
The Kiel researchers imagined how aerographite behaves when it is folded - at least according to their suspicions. To characterize the material and to prove that their idea is actually true, they also had to bend the micrometer-sized objects in practice. To do so, they needed a special scanning electron microscope, which they found in Riga (Estonia).
|
|
Here, the Kiel team was already working with fellow scientists on a different project. With a nanoscale measuring needle, the colleagues there were able to grasp and bend the aerographite tetrapods. The material scientists Dr. Stefano Signetti and Prof. Pugno, co-leading author of the paper, from the Italian University of Trento, provided the final mechanical understanding and generalization, developing both the analytical and numerical models, and thus also the proof that the assumptions of the Kiel colleagues were correct.
|
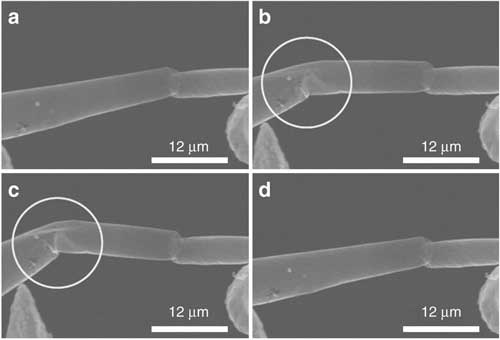 |
| A tetrapod arm in normal shape; (b) a needle touches the arm, which slowly starts to bend; (c) the arm bends significantly, before it (d) reverts back to its original shape without being damaged. (Image: Donats Erst, University of Latvia)
|
|
"Our theoretical and numerical modelling calculations provide a general understanding for the design of aerographite materials and are in very good agreement with the assumption of the Kiel researchers as well as experimental observations from Riga machine," adds Nicola Pugno, Professor of Solid and Structural Mechanics.
|
|
"The calculation method which has been developed and verified because of this international cooperation, can be applied to tetrapods in various sizes. It provides a valuable basis for investigating the properties of whole tetrapod networks and aerographite even further," elaborated Mishra.
|
|
In the long term, understanding how networks of hollow tetrapods can be folded however, we like without being damaged, could help to optimise the production of highly-porous solids such as aerogels and foams, or enable their use in tissue regeneration (so-called scaffold in medical engineering).
|



