| Posted: May 12, 2009 |
Smart dust biosensors |
|
(Nanowerk News) Smartdust biosensors may be smaller than a grain of sand but they have big potential, say Henry Hess, at the University of Florida, Gainesville, US, and colleagues ("'Smart dust' biosensors powered by biomolecular motors" – free access article).
|
|
A beehive is a very interesting biosensor: bees disperse from the hive, sample a large area and report their findings back to the hive. The concept of smart dust devices mimics this remote environmental monitoring process. A large number of devices dispersed over an area would be left to interact with a sample and then communicate their findings to scientists from a distance. To be economically feasible, such single-use devices have to be cheap, even cheaper than the radio-frequency identification tags currently used to track the inventory of warehouses, for example. Consequently, the devices cannot be much larger than a grain of sand, thus the name 'smart dust'.
|
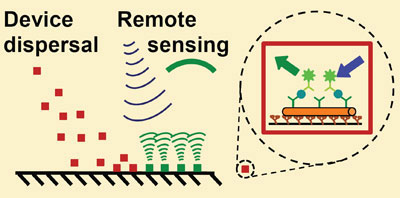 |
| With a built-in power source and driven by molecular motors, smart dust biosensors are remotely activated to detect toxins.
|
|
The design of smart dust sensors is a new frontier in the miniaturisation of lab-on-a-chip devices, since the traditional model of a passive cartridge inserted into a hand-held or desktop device has to be abandoned. The smart dust concept originated in the 1990s, but the size of the assembled microelectronic devices exceeded a cubic millimetre. In 2005, scientists suggested a bottom-up, particle-based strategy, where the spectral properties of engineered porous silicon microparticles changed as they were exposed to hydrocarbon fumes.
|
|
A third conceptual approach uses hybrid devices, combining synthetic components with active nanostructures of biological origin. This approach is promising because cells have evolved many sub-systems that are also critical in a lab-on-a-chip context. Active nanoscale transport is one such subsystem. Biomolecular motors, such as the motor protein kinesin, use energy provided by adenosine triphosphate (ATP) hydrolysis to move along the cytoskeleton and distribute a variety of cargo. Reconstituting this active transport mechanism in a smart dust device would overcome one of the bottlenecks to designing an autonomous microdevice: the pump or battery required for transport by pressure-driven fluid flow and electro-osmotic flow could be replaced by ATP-powered nanotransporters assembled from proteins.
|
|
Research efforts into the design of such nanotransporters, also called molecular shuttles, have laid the foundation for designing a complex smart dust biosensor. Scientists have developed approaches to guide molecular shuttles along predetermined paths, load them with a variety of cargo and control their activation.
|
|
|
|
At the heart of this biosensor is the double-antibody-sandwich (DAS) assay, a workhorse in biotechnology. A traditional DAS assay captures analytes with surface-adsorbed antibodies. It detects the analytes with a second type of antibody equipped with an optical tag or an enzyme catalysing a colour-changing reaction. While the traditional assay involves multiple exchanges of solution, the smart dust sensor exploits antibody-functionalised shuttles to capture the analytes, transport them to a tagging station and then move tagged analytes to a detection zone.
|
|
Designing such a biosensor not only requires some advanced molecular engineering but also the consideration of the real world challenges that the device will encounter. These challenges include storing molecular shuttles long-term and creating fluorescence signals of sufficient brightness for remote read-out.
|
|
The design, fabrication and testing of complex devices provides a focal point for the technological concepts and a proving ground for the specific solutions. Smart dust biosensors integrate the advances of the last decade in the field of nanoscale transport systems.
|

