| Jan 11, 2012 |
Metal oxide simulations could help green technology
|
|
(Nanowerk News) University of California, Davis, researchers have proposed a radical new way of thinking about the chemical reactions between water and metal oxides, the most common minerals on Earth. Their work appears in the current issue of the journal Nature Materials ("Metastable structures and isotope exchange reactions in polyoxometalate ions provide a molecular view of oxide dissolution").
|
|
The new paradigm could lead to a better understanding of corrosion and how toxic minerals leach from rocks and soil. It could also help in the development of "green" technology: new types of batteries, for example, or catalysts for splitting water to produce hydrogen fuel.
|
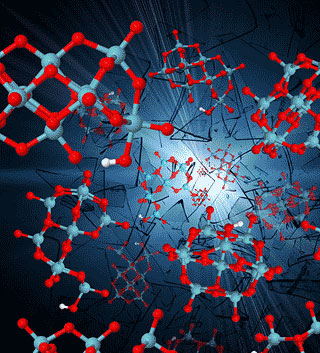 |
| Computer simulations show that metal oxides in water go through many short-lived shapes and structures. (William Casey/UC Davis graphic)
|
|
"This is a global change in how people should view these processes," said William Casey, UC Davis professor of chemistry and co-author of the study with James Rustad, a former geology professor at UC Davis who now works as a scientist at Corning Inc. in New York.
|
|
Previously, when studying the interactions of water with clusters of metal oxides, researchers tried to pick and study individual atoms to assess their reactivity. But "none of it really made sense," Rustad said.
|
|
Using computer simulations developed by Rustad, and comparing the resulting animations with lab experiments by Casey, the two found that the behavior of an atom on the surface of the cluster can be affected by an atom some distance away.
|
|
Instead of moving through a sequence of transitional forms, as had been assumed, metal oxides interacting with water fall into a variety of "metastable states" -- short-lived intermediates, the researchers found.
|
|
For example, in one of Rustad's animations, a water molecule approaches an oxygen atom on the surface of a cluster. The oxygen suddenly pulls away from another atom binding it into the middle of the cluster and leaps to the water molecule. Then the structure collapses back into place, ejecting a spare oxygen atom and incorporating the new one.
|

