| Jan 26, 2006 |
Mystery of metallic glass is cracked
|
|
(Nanowerk News) Using state-of-the-art lab techniques and powerful computer simulations, Johns Hopkins researchers have discovered how atoms pack themselves in unusual materials known as metallic glasses. Their findings should help scientists better understand the atomic scale structure of this material, which is used to make sports equipment, cell phone cases, armor-piercing projectiles and other products.
|
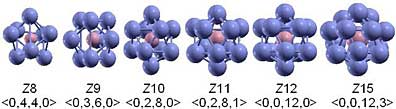 |
Johns Hopkins materials scientists determined that metallic glass atoms tend to group themselves around a central atom, forming three-dimensional shapes known as Kasper polyhedra, depicted in this illustration. (Source: Johns Hopkins University)
|
|
Despite its success as an imaging tool for nanostructures, the existing AFM system does not well reflect functionalities needed
The discovery, marking the culmination of a two-year research project, was reported in the Jan. 26, 2006 issue of the journal Nature: "Atomic packing and short-to-medium-range order in metallic glasses". The work represents a major step forward because the tools used to study traditional crystalline metals do not work well with metallic glass, and a better understanding of the material has been sorely needed.
|
|
"How the atoms pack themselves in metallic glass has been a mystery," said Howard Sheng, an associate research scientist in the Department of Materials Science and Engineering and lead author of the Nature paper. "We set out to decipher this packing information, and we were ultimately able to provide a clear description of how the atoms arrange themselves in metallic glass."
|
|
In conventional metals, atoms crystallize into uniform three-dimensional patterns known as lattices. But about a half-century ago, materials scientists learned how to make glassy metals by cooling a metallic liquid so quickly that the internal atomic configurations froze before the atoms had a chance to arrange themselves into a lattice pattern. The new material was described as amorphous, meaning its atoms seemed to be arranged in an irregular fashion without the long-range order characteristic of crystalline materials. This amorphous atomic structure is commonly found in other materials such as window glass, but it rarely occurs in metals.
|
|
Unlike window panes, metallic glasses are not transparent or easy to shatter. Many traditional metals are easy to bend out of shape because of defects (dislocations) in their crystal lattice. But metallic glasses have no crystal lattice and no such dislocations, and their disorderly arrangement of atoms gives them distinctive mechanical and magnetic properties. Metallic glasses, which are usually made of two or more metals, can display great strength, large elastic strain and toughness. Another advantage is that, like weaker plastic materials, they can easily be heated, softened and molded into complex shapes.
|
|
Despite the great potential of metallic glasses, the researchers who make them have been hampered by a scarcity of basic science knowledge about the materials. Powerful transmission electron microscopes can be used to view rows of atoms lined up in traditional metals. But when these instruments are used on a metallic glass, the resulting image is one of a scattered array of atoms, forming no obvious pattern. Because so little has been known about how atoms are arranged in metallic glasses, a number of basic materials science problems, such as how a metallic glass deforms, remain unsolved.
|
|
To help fill the knowledge gap, a team supervised by Evan Ma, a professor of materials science and
engineering at Johns Hopkins, launched a two-pronged
approach to solve the mystery of how metallic glass atoms
are arranged. "Our goal was to advance the understanding of
atomic packing in metallic glasses," Ma said. "This is a
difficult task because of the lack of long-range order in
these amorphous structures. Yet it is of fundamental
importance because it is the structure that determines
properties."
|
|
The researchers made samples of a number of binary metallic glasses, each composed of two elements, and then subjected them to high-tech lab tests to gather information about the samples´ three-dimensional atomic configurations. Some of these experiments, conducted at Oak Ridge and Brookhaven national laboratories, involved X-ray diffraction and extended X-ray absorption fine structure data taken at synchrotron X-ray sources. Other analyses, utilizing a method called reverse Monte Carlo simulations, were conducted with a computer cluster at Johns Hopkins. Independent of these lab tests, the researchers used powerful computer resources provided by the National Energy Research Scientific Computing Center to run virtual experiments aimed at uncovering the arrangement of metallic glass atoms. Results from the lab experiments and the computer trials were used to validate one another, confirming the researchers' conclusions.
|
|
One of their key findings was that metallic glass atoms do not arrange themselves in a completely random way. Instead, groups of seven to 15 atoms tend to arrange themselves around a central atom, forming three-dimensional shapes called Kasper polyhedra. Similar shapes are found in crystalline metals, but in metallic glass, the researchers said, these polyhedra are distorted and do not align themselves in long rows. In metallic glass, the polyhedra join together in unique ways as small nanometer-scale clusters. In the journal article, these structural features were described as chemical and topological short-range order and medium-range order.
|
|
The Johns Hopkins engineers also made important discoveries about how low-density spaces form among these clusters in metallic glass. These "cavities" affect the way the material forms as a glass and the mechanical properties it will possess Sheng, the lead author of the journal article, believes these discoveries will lead to significant advances in the understanding of metallic glass. "Our findings," he said, "should allow the people who make metallic glass to move closer to intelligent design techniques, developing materials with the precise mechanical characteristics needed for specific products. The discoveries also advance our understanding of materials science in general."
|
|
The metallic glass research was funded by a grant from the U.S. Department of Energy. Along with Sheng and Ma, the authors of the Nature article included Weikun Luo, a Johns Hopkins doctoral student in the Department of Materials Science and Engineering; F. M Alamgir of the National Institute of Standards and Technology; and J. M. Bai of the Oak Ridge National Laboratory.
|

