| Nov 27, 2012 |
Researchers discover what keeps a cell's energy source going
|
|
(Nanowerk News) Most healthy cells rely on a complicated process to produce the fuel ATP. Knowing how ATP is produced by the cell’s energy storehouse – the mitochondria -- is important for understanding a cell’s normal state, as well as what happens when things go wrong, for example in cancer, cardiovascular disease, neurodegeneration, and many rare disorders of the mitochondria.
|
|
Two years ago, Kevin Foskett, PhD, professor of Physiology at the Perelman School of Medicine, University of Pennsylvania, and colleagues discovered that fundamental control of ATP production is an ongoing shuttle of calcium to the mitochondria from another cell compartment. They found that mitochondria rely on this transfer to make enough ATP to support normal cell metabolism.
|
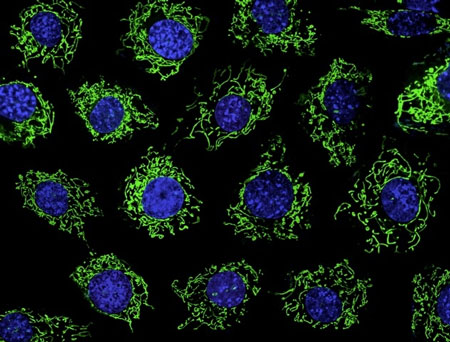 |
| Ca2+ flux across the inner mitochondrial membrane regulates cell bioenergetics, cytoplasmic Ca2+ signals and activation of cell death pathways. Ca2+ uptake from the cytoplasm is driven by the electrochemical gradient generated during ATP production, mediated by a Ca2+ ion channel called the mitochondrial Ca2+ uniporter. Molecular components of the uniporter include MCU, the Ca2+ conducting pore, and MICU1, a MCU regulator that limits Ca2+ permeation through MCU at rest. MCUR1 is as an essential third component of the uniporter complex. In the absence of MCUR1, mitochondrial Ca2+ uptake is markedly reduced, with adverse cellular consequences, including compromised cellular bioenergetics, diminished oxidative phosphorylation and activation of autophagy. The cover shows mitochondria (green) stained with the mitochondrial membrane potential indicator, rhodamine 123 in living primary mouse embryonic fibroblasts. Nuclei are stained with DAPI (blue).
|
|
Foskett’s lab and the lab of colleague Muniswamy Madesh, PhD, at Temple University, discovered last month an essential mechanism that regulates the flow of calcium into mitochondria, described in the October 26 issue of Cell ("MICU1 Is an Essential Gatekeeper for MCU-Mediated Mitochondrial Ca2+ Uptake that Regulates Cell Survival"). They found that the mitochondrial protein MICU1 is required to establish the proper level of calcium uptake under normal conditions.
|
|
In a new paper out this week in Nature Cell Biology ("MCUR1 is an essential component of mitochondrial Ca2+ uptake that regulates cellular metabolism"), the same Penn-Temple team describe a new protein and its function. Like MICU1, this new protein, MCUR1, interacts physically with MCU, the uniporter calcium ion channel within the mitochondria. Calcium uptake is driven by a voltage across the inner mitochondrial membrane and mediated by the calcium-selective ion channel called the uniporter.
|
|
“But this newly described protein, MCUR1, has the opposite role as MICU1,” notes Foskett. “It seems to be a subunit that, together with MCU, is required for a functional uniporter calcium channel.”
|
|
Many cell plasma membrane ion channels also have subunits that are required for those channels to work. Before this paper, there was no realization that this mitochondrial channel, MCU, did as well.
|
|
Maintaining the correct levels of calcium in the mitochondria plays an important role in cellular physiology: Calcium flux across the inner mitochondrial membrane regulates cell energy production and activation of cell-death pathways, for example. In MICU1’s absence mitochondria become overloaded with calcium, generating excessive amounts of reactive oxygen molecules and eventually cell death. In contrast, in the absence of MCUR1, mitochondria cannot take up enough calcium. This also has detrimental effects: the cells cannot make enough ATP and they activate autophagy, a mechanism in which cells “eat themselves” to provide sufficient nutrients for survival.
|
|
Both papers deal with the function of the uniporter, the calcium channel in the inner membrane of mitochondria that lets calcium get into the mitochondrial matrix where it can do good things like promote ATP synthesis and healthy bioenergetics, or bad things, like mitochondrial-mediated cell death, apoptosis and necrosis.
|
|
Because of these two papers, the uniporter is now recognized as a channel complex, containing -- at least -- MCU, MCUR1 and MICU1. Since the uniporter can be a therapeutic target is reperfusion injury, ischemic injury, and programmed cell death, MCUR1 and its interaction with MCU are now targets for drug development.
|

