| Jan 22, 2013 |
A diffusion trap
|
|
(Nanowerk News) Over the past several years, Rong Li, Ph.D., at the Stowers Institute for Medical Research has been making crucial discoveries about the development of cell polarity—the process by which one side of a cell becomes different from the other side. Such polarity is critical for the functioning of the vast majority of cells. The outside surface of skin cells is very different from the surface inside the body, for example, while nerve cells have delicately branching dendrites on one end and axons on the other. Li's lab studies yeast cells, which form a unique polarized site, the location of the future bud.
|
|
In previous work, Li and others figured out the basic biochemical mechanism underlying cell polarity. The key regulator is a protein called Cdc42. In a non-polarized cell, Cdc42 molecules are randomly distributed around the membrane, like pebbles scattered on a beach. But some parts of the membrane contain a bit more Cdc42 than others. When the regulatory protein is activated, it stimulates the formation of a skeleton of actin filaments that guide Cdc42 in the cytoplasm towards the Cdc42 that's already in the membrane. Mathematical modeling shows that this process is able to concentrate the molecule in those areas, creating a spot—called the polar cap—where yeast cells will bud. "The model worked very well, but it was simplified," says Li.
|
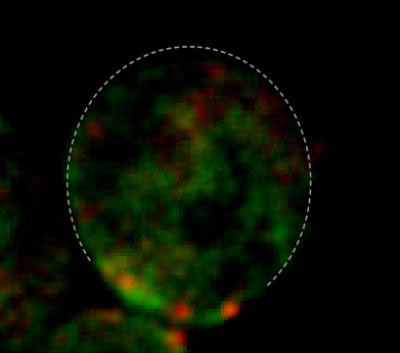 |
| This shows microdomains with enrichment of Cdc42 (green) within the polar region of a yeast cell are separate from sites of endocytosis (red). (Image: Courtesy of Research Advisor Brian Slaughter, Ph.D., Stowers Institute for Medical Research)
|
|
Now, using cutting-edge microscopy and more sophisticated mathematical modeling, Li's lab has discovered surprising new details about this process. In the January 22, 2013 issue of the online journal Nature Communications ("Non-uniform membrane diffusion enables steady-state cell polarization via vesicular trafficking"), Stowers Research Advisers Brian Slaughter, Ph.D, Jay Unruh, Ph.D., and Boris Rubinstein, Ph.D., along with Li and graduate students Arupratan Das and Sarah Smith report that the development of polarity depends upon "sticky" regions in the membrane that slow the normal rate of diffusion of Cdc42.
|
|
"We did not expect that plugging non-uniform diffusion into the mathematical simulation would make that much difference," says Slaughter, one of the paper's first authors. "But tweaking one parameter made all the difference in the world."
|
|
The findings have wide-ranging implications. The normal rate of diffusion is much faster in plant and mammalian cells than in yeast cells, suggesting that these sticky spots must play an even greater important role in animals and plants than in yeast. "Our model could apply to a lot of systems," explains Li, who is also a professor in the Department of Molecular and Integrative Physiology at the University of Kansas Medical Center. "It would be very interesting to explore this in mammalian cells."
|
|
The new work also highlights some of the unique strengths of the Stowers Institute. Unlike most biomedical research facilities, Stowers employs scientists called research advisors who act like internal freelance consultants, bringing special expertise to a variety of Stowers' labs and projects. Research Advisors Slaughter and Unruh, for instance, are experts in imaging. In this project, they used advanced microscopy to spot single photons (or particles of light) coming from molecules with fluorescent tags as those molecules moved around in living cells. That revealed the presence of the sticky spots. Unruh then modeled the "stickiness" of these spots in a complex numerical simulation while another research advisor, Boris Rubinstein, a physicist in training, brought his mathematical skills to the modeling effort in the form of an analytical model. Thanks to the capabilities at the Stowers Institute, "we can count molecules and measure their diffusion in the living cell," says Li. "Then, combining those measurements with mathematical modeling is very powerful."
|
|
The central challenge for creating polarized cells is keeping the Cdc42 molecules concentrated in one spot on the membrane. As Li's team originally discovered, a small excess of Cdc42 in one area stimulates the formation of actin filaments within the cell that then bring more Cdc42 to the spot—even as molecules already in the membrane are diffusing away. Imagine that region of the membrane is a prison and the Cdc42 molecules are prisoners, says Li: "The prisoners escape from prison and the police catch them and put them back." For polarity to develop, recaptures have to happen faster than escapes.
|
|
What complicates this picture is that for the particular mechanism of cell polarization being studied, the Cdc42 that is brought to the cell membrane along the actin filaments is not in the form of free-floating molecules. Instead, the Cdc42 is embedded in the membranes of vesicles, which are small bubbles inside cells. When vesicles reach the cell membrane, the vesicle and cell membranes join together in a process called exocytosis. As a result, Cdc42 from the vesicle membrane ends up in the cell membrane. However, in an opposite process, called endocytosis, sections of cell membrane can bulge inward, forming a new vesicle that pinches off and floats into the cell. That process thus can snatch Cdc42 molecules out of the cell membrane to recycle them back to the polar cap.
|
|
But building a polar cap requires vesicles with lots of Cdc42 in their membranes. As the vesicles fuse with the cell membrane, those with high Cdc42 concentrations quickly get used up. Soon, the remaining vesicles have so little Cdc42 that when they get inserted in the cell membrane, the concentration of Cdc42 drops. The result: polarization can't be stably maintained, as shown by a model developed by researchers in the Lew lab at Duke University. So Li and her team set out to figure out what is really happening in cells. Using advanced microscopy, the researchers looked at the rate of movement of Cdc42 at the sites of exocytosis and endocytosis on the cell membrane.
|
|
The painstaking experimental work turned up two crucial new details. First, the process of endocytosis, which theoretically could remove Cdc42 from the developing polar cap, occurs in small regions adjacent to, but not overlapping with, areas where Cdc42 is delivered. More important, the section of the membrane where Cdc42 accumulates gets "sticky", trapping the master polarity regulator and keeping it from rapidly diffusing away. When Li's team put these details—the diffusion trap and the targeted endocytosis—into the mathematical model, "the model turned out to work beautifully," says Li.
|

