| Jan 30, 2013 |
Researchers produce new photoactive micelles with potential applications in photofunctional dyes and sensors
|
|
(Nanowerk News) A new form of micelle, which is composed of detergents with bent aromatic panels, has been created by Michito Yoshizawa and his colleagues at Tokyo Institute of Technology. Unlike traditional micelles, the new ‘aromatic micelles’ are photoactive, and capable of encapsulating dye molecules and showing unusual fluorescence in aqueous solutions ("Micelle-like Molecular Capsules with Anthracene Shells as Photoactive Hosts").
|
|
“The present micelles might be suitable for potential applications in the fields of photofunctional dyes, sensors, and materials owing to their ability to accommodate dye molecules and their efficient host-guest energy transfer in aqueous media,” explain the researchers. They also emphasise the straightforward synthesis, aqueous green chemistry and high stability of the aromatic micelles.
|
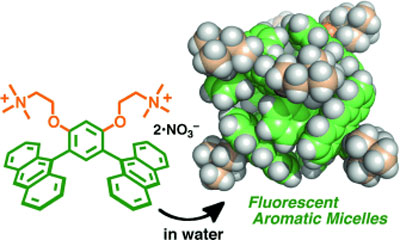 |
| Aromatic micelles: Hydrophobic and aromatic–aromatic interactions promoted the spontaneous formation of micelle-inspired molecular capsules with large aromatic shells from bent bisanthracene amphiphiles (see picture). The micellar capsules could accommodate fluorescent-dye guests in water, and the resultant nanocomposites exhibited strong fluorescence through efficient energy transfer from the host shells to the encapsulated guests. (© Wiley)
|
|
Micelles are used in a range of dissolution, separation, and preservation applications and form the basis of soap detergents. They assemble from string-like molecules in aqueous solutions as a result of different chemical components (hydrophobic and hydrophilic moieties) at either end of the strings. Michito Yoshizawa and Kei Kondo et al. replaced the hydrophobic part of the string with large aromatic panels, which are known to be photochemically active.
|
|
The new aromatic micelles form two nanometer-sized capsules that have a cavity surrounded by large aromatic panels. The selective encapsulation of fluorescent dye molecules to form photoactive guest-host complexes is the first demonstration of efficient fluorescence energy transfer from the host framework to dye ‘guest molecules’.
|
|
Background
|
|
The micelles
|
|
Micelles are typically made of string-like molecules, where one end of the string attracts water molecules (hydrophilic) and the other end repels them (hydrophobic). In aqueous solution these strings form spherical assembles like spoke on a dandelion puffball with the hydrophobic ends at the centre. These typical micelles are not photoactive, which places limitations on their potential applications.
|
|
Aromatic molecules
|
|
Aromatic molecules are carbon-based molecules with six-membered rings that have a particular type of electronic configuration, which leads to a number of specific properties. Large aromatic molecules are planar and photo- and electrochemically active, which may make them useful in applications such as liquid crystal displays. However they do not readily form discrete micelle-like assemblies.
|
|
Producing aromatic micelles
|
|
To create the aromatic micelles, the researchers made new detergents with bent aromatic panels, comprising two anthracene moieties with a spacer to connect them. The spacer was functionalized with two hydrophilic groups. The steric repulsion between the anthracene and spacer moieties gives the molecule its bent shape. With the hydrophobic bent panels and the hydrophilic groups, the detergent molecules form spherical assemblies in aqueous solution. The assembly is driven by stacking of the hydrophobic aromatic panels.
|
|
Aromatic micelle properties
|
|
The aromatic micelles had a cavity surrounded by anthracene shells with a diameter of approximately one nanometer. The shell emitted blue-green fluorescence (~500 nm). The aromatic micelles were also extremely robust. Atomic force microscopy (AFM) images showed the spherical structures persisted even after complete evaporation of water.
|
|
Dye encapsulation
|
|
The researchers demonstrated the encapsulation of two well known hydrophobic dyes, Nile red (NR) and DCM by the aromatic micelles. Stirring a suspension of the dyes in an aqueous solution of the aromatic micelles for an hour produced a clear solution. Spectroscopic observations revealed changes to the light absorption and emission properties of the micelle following encapsulation of the dyes. Strong red emission (~640 nm) from the encapsulated DCM was observed upon irradiation of the micelle-DCM complex at 370 nm. In contrast, the irradiation of DCM in the absence of the micelle at 370 nm showed almost no emission. Measurements of the fluorescence quenching profile of the anthracene panels in the micelles indicated the energy transfer efficiency as high as 97%. The work is the first demonstration of efficient fluorescent resonance energy transfer from discrete self-assembled hosts.
|
|
Next steps
|
|
The authors expect that the functionalization of the aromatic shells as well as the use of other aromatic panels will lead to new aromatic micelles with a wide range of fluorescent properties. Studies along these lines are currently in progress in their research group.
|

