| Apr 12, 2013 |
Nanotechnology gradient chip makes it easier to measure chemical reactivity
|
|
(Nanowerk News) Nanotechnologists at the University of Twente’s MESA+ research institute have developed a tiny chip that makes it easy to create micrometre scale gradients. Gradients are gradual transitions in specific properties, such as acidity. This newly-developed system can be used to efficiently measure the reaction kinetics of various chemical or biological reactions. The results of this research work will be published in the scientific journal Nature Communications, on 9 April ("Reactivity mapping with electrochemical gradients for monitoring reactivity at surfaces in space and time").
|
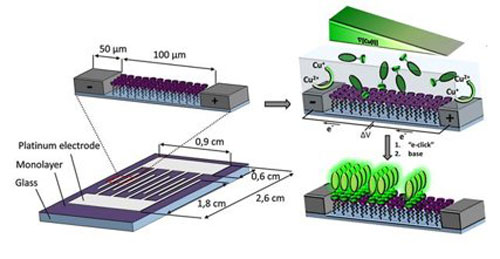 |
|
In many chemical and biological reactions, it is important to know how readily and rapidly the substances involved react with one another. For instance, it is vital to have a detailed understanding of the ideal conditions for reactions used in industrial processes. However, measuring this reactivity can often pose considerable challenges. Research workers at the University of Twente have now developed a system that allows you to easily measure the reaction kinetics of reactions that take place on surfaces.
|
|
Chemical gradient
|
|
The system consists of a reaction surface on a small glass plate, above which (in the liquid phase) the reaction can take place. The small glass plate is fitted with two electrodes, one hundred micrometres apart. By setting up an electrical potential difference (voltage) between these electrodes, a chemical gradient (such as a pH gradient) can be created above the surface. You can then determine the reaction kinetics of the reaction by measuring the speed of the reaction at various points between the electrodes. In the past, it was necessary to perform a reaction many times, under a variety of conditions. This chip allows you to test a wide range of conditions all at once.
|

